GcMAF
[ Vitamin D binding protein macrophage activating factor ]
[ Vitamin D binding protein macrophage activating factor ]
Wikipedia :Gc-MAF
YouTube : GcMAF: THE AMAZING ANSWER FOR YOUR HEALTH
Drbradstreet.org : Dr Jeff Bradstreet
Michael Miller : Anti-vaccine doctor behind ‘dangerous’ autism therapy found dead. Family cries foul
YouTube : The real reason Holistic Doctors are being killed and vanishing!
Richard Presser : Is this the real reason Holistic Doctors are being killed and vanishing?
Informahealthcare.com : Vitamin D binding protein
Kat Arney : “Cancer cured for good?” – Gc-MAF and the miracle cure
RetractionWatch : Yet another study of widely touted cancer “cure” retracted
Gcmaf.eu / YouTube : Cancer cells destroyed by First Immune GcMAF
anticancerfund.org : Warning: GcMAF has not been properly studied in clinical trials...
David M. Mosser : The many faces of macrophage activation
Nobuto Yamamoto, et al. : Retracted: Immunotherapy of HIV-infected patients with Gc protein-derived macrophage activating factor (GcMAF)
Nobuto Yamamoto : Retracted: Immunotherapy of metastatic breast cancer patients with vitamin D-binding protein-derived macrophage activating factor
Efranat : Clinical Trials
Nobuto Yamamoto : Bio
Patents & Applications
Pharmocracy in action :
https://en.wikipedia.org/wiki/Gc-MAF
Gc-MAF
Gc-MAF or Gc protein-derived macrophage activating factor is an immunomodulatory protein that, by affecting the immune system, may play a role in various diseases.[1]
In 2008 claims were made that Gc-MAF can provide a permanent cure for cancer and HIV. These claims have been the subject of much criticism[2] and are not supported by scientific evidence. The papers supporting the claims have since been retracted by the journals in which they were published.[3] Consumers have been warned about illegal marketing of the substance over the internet.[4]
Description
Biochemically, Gc-MAF results from sequential deglycosylation of the vitamin D-binding protein (the Gc protein), which is naturally promoted by lymphocytes (B and T cells).[1] The resulting protein may be a macrophage activating factor (MAF).[1] MAFs are lymphokines that control the expression of antigens on the surface of macrophages, and one of their functions is to make macrophages become cytotoxic to tumors.[5] Gc-MAF may play a role in various diseases.[1]
Therapeutic claims
GcMAF has not been properly studied in clinical trials and its laboratory results still need to be confirmed independently. So far, all claims on the efficacy of this product have no solid scientific basis. Its marketing is illegal; therefore there is no controlled guarantee on the quality of the product for human consumption sold over the internet.
Public warning issued by the Anticancer Fund[4]
Gc-MAF has been promoted as a cure for some cancers,[2] HIV[6] and other diseases. The integrity of the research, conducted by Nobuto Yamamoto and colleagues, that originally prompted claims regarding cancer and HIV has been questioned.[2][4] Cancer Research UK has warned the public about spurious claims of clinical benefits, misleadingly based on reduced levels of the alpha-N-acetylgalactosaminidase enzyme (also known as nagalase), whose production might be increased in many cancers.[2] Nagalase is an enzyme present in normal cells and its use to diagnose or follow-up the diseases claimed to be cured by GcMAF has not been validated. Nagalase deficiency, however, is associated to a rare congenital metabolic disorder called Schindler/Kanzaki disease.
Three out of four of the original studies authored by Yamamoto (published between 2007 and 2009) were retracted by the scientific journals in which they were published in 2014, officially due to irregularities in the way ethical approval was granted.[7][8][6][9]
The Belgian Anticancer Fund has communicated serious concerns to other journals that published studies on Gc-MAF by Yamamoto and colleagues.[4] They have also warned consumers of illegal marketing of Gc-MAF, over the internet and elsewhere.[4][10]
As of May 2014 there was one Phase I clinical trial registered to evaluate Gc-MAF. This trial only aims to evaluate the safety of this product for human consumption, efficacy is not yet being studied. The product used in this trial is not available out of the study and the companies commercializing Gc-MAF over the internet are not involved. No results are yet available.[11]
1) Malik, Suneil; Fu, Lei; Juras, David James; Karmali, Mohamed; Wong, Betty Y. L.; Gozdzik, Agnes; Cole, David E. C. (January–February 2013). "Common variants of the vitamin D binding protein gene and adverse health outcomes". Critical Reviews in Clinical Laboratory Sciences 50 (1): 1–22. http://informahealthcare.com/doi/full/10.3109/10408363.2012.750262
doi:10.3109/10408363.2012.750262. PMC 3613945. PMID 23427793.
2) Arney, Kat (3 December 2008). "'Cancer cured for good?' – Gc-MAF and the miracle cure (revised 25 July 2014)". Cancer Research UK.
http://informahealthcare.com/doi/full/10.3109/10408363.2012.750262
3) "Tracking retractions as a window into the scientific process Yet another study of widely touted cancer “cure” retracted". Retraction Watch.
http://retractionwatch.com/2014/10/10/yet-another-study-of-widely-touted-cancer-cure-retracted/
4) "GCMAF". Anticancer Fund. 24 July 2014.
http://www.anticancerfund.org/therapies/gcmaf
5) Mosser, David M. (February 2003). "The many faces of macrophage activation". Journal of Leukocyte Biology 73 (2): 209–212. https://en.wikipedia.org/wiki/Journal_of_Leukocyte_Biology
doi:10.1189/jlb.0602325. PMID 12554797.
6) Yamamoto, Nobuto; Ushijima, Naofumi; Koga, Yoshihiko (January 2009). " (Retracted): Immunotherapy of HIV-infected patients with Gc protein-derived macrophage activating factor (GcMAF)". Journal of Medical Virology 81 (1): 16–26.
https://en.wikipedia.org/wiki/Journal_of_Medical_Virology
doi:10.1002/jmv.21376. PMID 19031451
7) Yamamoto, Nobuto; Suyama, Hirofumi; Yamamoto, Nobuyuki; Ushijima, Naofumi (15 January 2008). " (Retracted): Immunotherapy of metastatic breast cancer patients with vitamin D-binding protein-derived macrophage activating factor (GcMAF)". International Journal of Cancer 122 (2): 461–467.
doi:10.1002/ijc.23107.
8) ) Yamamoto, N.; Suyama, H.; Nakazato, H.; Yamamoto, N.; Koga, Y. (2014). "Retraction Note to: Immunotherapy of metastatic colorectal cancer with vitamin D-binding protein-derived macrophage-activating factor, GcMAF". Cancer Immunology, Immunotherapy 63 (12): 1349. doi:10.1007/s00262-014-1616-x.
"Retraction". International Journal of Cancer 135 (6): 1509. 15 September 2014. doi:10.1002/ijc.29014.
9) "Retraction". International Journal of Cancer 135 (6): 1509. 15 September 2014.
doi:10.1002/ijc.29014
10) Emmanuel Borloz (19 June 2015). "Clinique privée sous enquête pénale après cinq morts Enquête" (in French). 24 Heures.
http://www.24heures.ch/vaud-regions/lausanne-region/clinique-privee-vaudoise-enquete-penale-cinq-morts/story/12349881
11) "Safety Study of GcMAF (Globulin Component Macrophage Activating Factor) in Subjects With Advanced Solid Tumors". U.S. National Institutes of Health.
https://clinicaltrials.gov/ct2/show/NCT02052492?term=gcmaf&rank=1
https://www.youtube.com/watch?v=KqMohmjJ4mg
GcMAF:
THE AMAZING ANSWER FOR YOUR HEALTH
Huge amounts of sci-tech info exposing the vaccine-autism connection :
http://drbradstreet.org/page/8/
Huge amounts of sci-tech info exposing the vaccine-autism connection :
http://drbradstreet.org/page/8/
Dr
Jeff Bradstreet, MD, MD(H), FAAFP
Hope for complex health issues: (678) 288-9222 (NEW NUMBER) Accepting New Patients

http://www.washingtonpost.com/news/morning-mix/wp/2015/06/29/anti-vaccine-doctor-behind-dangerous-autism-therapy-found-dead-family-cries-foul/
Anti-vaccine
doctor behind ‘dangerous’ autism therapy found dead.
Family cries foul
by
Michael E. Miller
by
Michael E. Miller
https://www.youtube.com/watch?v=cALgIHETMDU&feature=youtu.be
Explosive:
The real reason Holistic Doctors are being killed and
vanishing!
http://www.richardpresser.com/wordpress/is-this-the-real-reason-holistic-doctors-are-being-killed-and-vanishing/
July 24, 2015
Is
this the real reason Holistic Doctors are being killed and
vanishing?
by
Richard Presser
by
Richard Presser
Firstly, let me say that I do not have the knowledge to assess this material from a scientific perspective, but it rings a lot of bells for me, and there are some who read my posts who will know.
It is clear that several holistic doctors have died recently, and another was announced today. Is there a link between some or all of them?
It’s difficult to be certain, but when the breadcrumbs begin to line up, a trail begins to emerge.
So let me cut to the chase of what this article says:
Human GcMAF, otherwise known as Vitamin D binding protein macrophage activating factor, holds great promise in the treatment of various illnesses including cancer, autism, chronic fatigue and possibly Parkinson’s. Since 1990, 59 research papers have been published on GcMAF, 20 of these pertaining to the treatment of cancer. 46 of these papers can be accessed through the GcMAF web site.
GcMAF is a vital part of our immune system which does not work without it; and is part of our blood. GcMAF stimulates the macrophage element of the immune system to destroy cancer cells. It also blocks the supply of nutrients to cancer cells by stopping blood vessel development to the site (anti-angiogenesis). Cancer cells are weakened and starved, making them more vulnerable to attack by the GcMAF stimulated macrophage system. Research has shown macrophage activation and stopping diseased blood vessel development can also help in various neurological diseases such as Parkinson’s, Alzheimer’s, rheumatoid arthritis, inflammatory conditions, and diabetic retinopathy…
…In the past months Dr. Bradstreet has become interested in nagalese (also spelled nagalase in this document), which he describes as an enzyme “produced by cancer cells and viruses.” He thinks it unlikely that children with autism have undiagnosed cancers, and thus suspicion falls on a viral etiology. Dr. Bradstreet writes, “Viruses make the nagalese enzyme as part of their attachment proteins. It serves to get the virus into the cell and also decreases the body’s immune reaction to the virus-thereby increasing the odds of viral survival.”
Further on Dr. Bradstreet writes, “It is reasonable and likely that the nature of the immune dysfunction and the frequently observed autoimmune problems in autism are mediated by persistent, unresolved viral infections.” He claims to have tested approximately 400 children with autism for the viral marker, nagalese, and found that nearly 80% have significantly elevated levels. He hopes to publish soon on this study and believes this information “is one of the most important developments in the clinical treatment of children on the spectrum that I have experienced in the last 15 years.”…
…Nonetheless, his son’s case helped convince Bradstreet that vaccines caused autism. He took his message to the highest levels of government. Twice he testified about the supposed link between vaccines and autism before the U.S. House of Representatives.
“He was a very happy, well connected child prior to his MMR at approximately 12 months of age,” Bradstreet told representatives in 2002, presenting copies of his son’s various tests. “Matthew completely lost about 2 months after his MMR vaccine.”
From his clinic in Buford, Ga., Bradstreet treated patients from around the world, many who sought him out online. Desperate parents seeking answers for their children’s maladies would write to him on his blog, begging him for help…
…In the case of autism, Dr. James Bradstreet has so far treated 1,100 patients with GcMAF with an 85% response rate. His results show a bell curve response with 15% of the patients showing total eradication of symptoms and 15% showing no response.
In addition, experimental and clinical evidence confirms that GcMAF shows multiple powerful anti-cancer effects that have significant therapeutical impact on most tumors including breast, prostate, and kidney. GcMAF is created in the body by the release of two sugar molecules from a GcProtein molecule…
…In conclusion, GcMAF restores the energetic balance in the cell. Cancer cells driven by sugar metabolism become healthy oxygen driven cells, so tumor cells no longer behave as parasitic organisms. GcMAF stimulates macrophages to consume the cancer cells and cells invaded by viruses. This stimulation of the immune system and the anti-angiogenetic effect surrounding the tumor is beneficial in cancer and several neurological disorders like autism, chronic fatigue, Parkinson’s, and Alzheimer’s, and it is available to the general public… (not any more – the company has been shut down)
…After discussing her options the patient wanted to try GcMAF therapy prior to considering more radiation or chemotherapy. After 6 weeks of GcMAF 100ng/week subcutaneous injections (much like a shot of insulin) her repeat nagalase test returned at 2.10 (a 50% reduction). All of her other tumor markers remain negative and she is taking the dose of Vitamin D3 required to optimize her blood levels (9000 iu/day). It is too soon for her PET to be repeated but we will follow this soon to determine the course of the bone metastasis. The nagalase test may be a more sensitive marker for tumor burden than other more accepted blood tests. GcMAF given via simple patient administered once weekly injections is clearly able to reduce the nagalase level dramatically over a short period of time. In previous published studies, nagalase response to GcMAF was correlated with reduction and eventual elimination of cancer. This is an encouragement to us all and I will keep you posted on the patient’s progress.”…
…With compromised immune activation, increased nagalase cuts off the conversion to GcMAF – result is a deglycosylated Gc protein that cannot activate macrophages.
If you have increased nagalase, you have less GcMAF and your Gc protein is not effectively transferred into GcMAF.
Nagalase is part of the gp120 enzyme in HIV. HERV’s or other viruses active in cells may produce nagalase.
Several intestinal bacteria are producers of nagalase. Editor’s Note: I found this connection to be quite interesting; the gut is big.
Similar to HIV, CFS patients have many infections and reactivate endogenous herpes viruses – EBV, CMV, HHV-6, HSV-1, as well as Herpes 7.
Healthy controls have very low nagalase enzyme activity. Normal people do have some, but it should be very low. There is a clear difference in those with pathology…
What is Nagalase?
Nagalase is a protein made by all cancer cells and viruses (HIV, hepatitis B, hepatitis C, influenza, herpes, Epstein-Barr virus, and others). Its formal, official chemical name is alpha-N-acetylgalactosaminidase, but this is such a tongue-twisting mouthful of a moniker that we usually just call it “Nagalase.” (Sometimes, when I want to impress friends with my brilliance, I’ll say the entire word real fast: “alpha-N-acetylgalactosaminidase.” I have found that it’s important to practice beforehand if one doesn’t want to embarrass oneself.)
Why is Nagalase important?
Nagalase causes immunodeficiency. Nagalase blocks production of GcMAF, thus preventing the immune system from doing its job. Without an active immune system, cancer and viral infections can grow unchecked.
As an extremely sensitive marker for all cancers, Nagalase provides a powerful system for early detection.
Serial Nagalase testing provides a reliable and accurate method for tracking the results of any therapeutic regimen for cancer, AIDS, or other chronic viral infection.
Nagalase proves that cancer cells break all the rules
Normal healthy cells cooperate with one another in a concerted effort to further the good of all. Cancer cells refuse to play ball. Their disdainful attitude toward the rest of our cellular community is appalling. For example, these cellular scofflaws ignore clear messages to stop growing and spreading and encroaching on their neighbor’s space. How would you like it if your neighbor moved his fence over into your backyard?
Of all the rules cancer cells break, none is more alarming than the production of Nagalase, the evil enzyme that completely hog-ties the immune system army’s ability to stop cancer cells.
Virus particles also make Nagalase. Their goal is the same as that of the cancer cells: survival by incapacitating their number one enemy: the immune system.
Nagalase precision
Like a stealth bomber, the Nagalase enzyme synthesized in and released from a cancer cell or a virus particle pinpoints the GcMAF production facilities on the surface of your T and B lymphocytes and then wipes them out with an incredibly precise bomb. How precise? Let me put it this way: Nagalase locates and attacks one specific two-electron bond located at, and only at, the 420th amino acid position on a huge protein molecule (DBP), one of tens of thousands of proteins, each containing millions of electrons. This is like selectively taking out a park bench in a major city from six thousand miles away. More astonishing, if that is possible, Nagalase never misses its target. There is no collateral damage.
As you already know, GcMAF is a cell-signaling glycoprotein that talks to macrophages, enabling them to rapidly find, attack, and kill viruses and cancer cells. By activating macrophages, GcMAF triggers a cascade that activates the entire immune system. Blockage of GcMAF production by Nagalase brings all this wonderful anti-cancer and anti-viral immune activity to a screeching halt, allowing cancer and infections to spread.
What does Nagalase actually do? How does it destroy immune functioning and deactivate macrophages?
Once synthesized and released into nearby tissue or into the bloodstream, Nagalase, like that drill sergeant at boot camp, shouts harsh commands at the vitamin D binding protein (DBP) that is about to be turned into GcMAF. Nagalase demands that DBP not, under any circumstances, attach itself to a specific sugar molecule (galactosamine). If DBP has already grabbed (i.e., connected to, using a two-electron, “covalent” bond) a galactosamine sugar molecule, it is commanded to immediately let go. “Leave galactosamine alone, or you’ll be in big trouble!!!” is the Nagalase sergeant’s command. We’ll probably never know whether or not, on some deeper level, DBP knows that Nagalase’s motives are dastardly—but it doesn’t really matter: DBP will definitely always obey. Like the army private, the DBP literally has no choice. Because of the way hierarchies work in cellular biology, proteins must do the bidding of their enzymes. The enzymes, like Nagalase, are the drill sergeant and the proteins, like DBP, are the privates. That’s just the way it is. Obeying the drill sergeant’s command means DBP can’t do its assigned task, that of becoming GcMAF. It is rendered useless. For DBP, on a molecular level, life no longer has meaning…
End of quotes. These quotes are not in the same order in the document they are taken from.
So where does nagalase come from in infants?
Although this article does not spell it out, this video claims that nagalase is INTENTIONALLY included in vaccines. Perhaps this is one reason for the vaccine/autism correlation. It also fits with the broader array of health issues flowing from vaccination.
There are many links to follow here for those interested; however, it appears most if not all of the doctors who have recently “died” were connected to treatment practices utilising GcMAF to defeat the effects of nagalase.
I would appreciate feedback from those qualified to assess this. To me, it has all the hallmarks of a bombshell.
https://www.youtube.com/watch?v=0v3IA2Hj1TA&feature=youtu.be
Dr.Bradstreet Search Warrant: the promising drug GcMAF was their target!!
http://scienceblogs.com/insolence/files/2015/07/BradstreetSearchWarrant.pdf
http://informahealthcare.com/doi/full/10.3109/10408363.2012.750262
ABSTRACT
The vitamin D binding protein (DBP) is the major plasma carrier for vitamin D and its metabolites, but it is also an actin scavenger, and is the precursor to the immunomodulatory protein, Gc-MAF. Two missense variants of the DBP gene – rs7041 encoding Asp432Glu and rs4588 encoding Thr436Lys – change the amino acid sequence and alter the protein function. They are common enough to generate population-wide constitutive differences in vitamin D status, based on assay of the serum metabolite, 25-hydroxyvitamin D (25OHD). Whether these variants also influence the role of vitamin D in an immunologic milieu is not known. However, the issue is relevant, given the immunomodulatory effects of DBP and the role of protracted innate immune-related inflammation in response to tissue injury or repeated infection. Indeed, DBP and vitamin D may jointly or independently contribute to a variety of adverse health outcomes unrelated to classical notions of their function in bone and mineral metabolism. This review summarizes the reports to date of associations between DBP variants, and various chronic and infectious diseases. The available information leads us to conclude that DBP variants are a significant and common genetic factor in some common disorders, and therefore, are worthy of closer attention. In view of the heightened interest in vitamin D as a public health target, well-designed studies that look simultaneously at vitamin D and its carrier in relation to genotypes and adverse health outcome should be encouraged.
http://scienceblog.cancerresearchuk.org/2008/12/03/cancer-cured-for-good-gc-maf-and-the-miracle-cure/
December 3, 2008
“Cancer cured for good?” – Gc-MAF and the miracle cure
by
Kat Arney
Note: This post has been updated as several research papers about Gc-MAF have been retracted. We will continue to update this post as more information becomes available. Last update 09/10/14
As an organisation dedicated to beating cancer, we have a deep-rooted interest in any new research developments that could lead to new, more effective treatments for the disease.
So when we received an enquiry from a supporter about an article entitled “Cancer cured for good” by Bill Sardi and Timothy Hubbell* we were intrigued. The article talks about research by Nobuto Yamamoto in the US, looking at a protein called Gc-MAF (aka GcMAF). His published studies appear to show that injections of very small amounts of Gc-MAF can “cure” people with breast, bowel and prostate cancer.
According to the article, “It works 100% of the time to eradicate cancer completely, and cancer does not recur even years later.” Could this be the so-called ‘cure for cancer’ that we’ve been searching for all these years?
Sadly – as with so many things in life – if it sounds too good to be true it probably is. Let’s explore a bit further.
What’s the idea behind it?
Dr Yamamoto studies the immune system – the highly complex network of cells that helps to keep us healthy. The cells of the immune system – white blood cells – fight bacterial and viral infections because they can recognise and attack these ‘foreign’ invaders . But they’re not so good at tackling cancer, since tumours grow from our own cells and have clever mechanisms to ’cloak’ them from immune attack.
Macrophages (meaning “big eaters” in Greek) are an important type of white blood cell. They patrol the body, eating up foreign invaders and dead cells. They also help to alert other immune cells to the presence of infections.
Macrophages can be stirred into action by a small sugar-coated protein (glycoprotein) called Gc-MAF, short for Gc Macrophage Activating Factor, which is produced by the body. But the production of Gc-MAF is blocked by an enzyme called Nagalase (alpha-N-acetylgalactosaminidase), produced by many cancers. This is one of the mechanisms that helps tumours evade the immune system.
Yamamoto’s theory is that injecting cancer patients with Gc-MAF should activate their macrophages to fight the cancer. He tested it back in 1997 in a paper published in the journal Cancer Research, showing that injecting Gc-MAF into mice transplanted with cancer cells could improve their survival from around 16 days to around 35.
But the treatment did not ‘cure’ the cancer, as the cancer cells continued to multiply, eventually killing the mice.
Clinical trials
Fast-forward a few years, to the publication of three papers detailing the results of clinical trials of Gc-MAF carried out by Yamamoto, testing the treatment on patients with breast, bowel and prostate cancer.
Note: The breast cancer paper (Yamamoto et al Int J Cancer 2008) has now been retracted, due to various concerns with the work. Read more on the RetractionWatch blog. [Updated KA 25/07/14] The bowel cancer paper (Yamamoto et al Cancer Immunology Immunotherapy 2008) has also now been retracted. This letter details some of the concerns about the work. [Updated KA 09/10/14]
The results appear to be startling – all the patients on the trials are ‘cured’ of cancer. Surely this is an amazing breakthrough?
Put bluntly, no it isn’t. There are significant scientific problems with the trials. For a start, all the studies are very small, involving fewer than twenty patients in each – rather than the thousands needed to make the sort of claims mentioned above.
Next, all the patients involved had received standard treatment for their cancer, including surgery, chemotherapy and/or radiotherapy. This is a somewhat unorthodox design for a trial of this kind, because it makes it very difficult to tell if any successes are due to the new drug, or the more conventional treatments.
On top of this, the researchers didn’t actually monitor the progress of tumours in the patients, and provide no clinical information about them. Instead they opt to measure levels of Nagalase in the blood, rather than looking at any standard established markers for cancer.
For example, in the case of the breast cancer patients, there is no detail about their “TNM” (tumour, node, metastasis) status. This is a standard measure of how far a patient’s cancer has spread, and is used to calculate the likelihood that it will return.
Furthermore, the researchers didn’t do any tests to show that injected Gc-MAF was actually activating macrophages in the patients’ blood, or even working in the way that they expect. There is no information about levels of cytokines – the proteins produced by immune cells when they are activated – or analysis of the patients’ immune cells.
Perhaps most significantly, there are no controls – untreated patients for comparison – and the studies only followed the patients for a few years. We have no way of telling whether their cancers were growing again, or had been successfully treated, and whether this was due to Gc-MAF or the other treatment they had received.
Given that 80 per cent of all women with breast cancer survive for at least 5 years, an uncontrolled study showing that 16 women of unknown TNM status survive for at least 4 years is no great shakes, scientifically speaking.
Further problems
Another telling point is the type of journal in which the research was published. If this research was truly groundbreaking, and pointed the way to a cure for cancer, then the research would likely be found in top-tier ’high-impact’ medical journals journals like The Lancet, The New England Journal of Medicine or the Journal of the American Medical Association.
And finally, virtually all the references in the papers are to other papers published by Yamamoto and his team. If Gc-MAF was indeed a promising candidate for a successful cancer treatment, you’d expect plenty of other research to show the same thing. Scientists are usually quick to spot promising, emerging fields of research and jump on the bandwagon.
The poor quality of scientific papers supporting GcMAF is discussed here on the Scholarly Open Access blog. [Updated KA 25/07/14]
Is there hope?
Although this particular approach isn’t all it’s hyped up to be, harnessing the power of immune system could be a very potent way to treat cancer. We’ve blogged many times already on high-quality research into immunotherapy (for example here, here, here and here)
And many Cancer Research UK-funded scientists are also working in this field. For example, Professor Fran Balkwill and her team are working on ways to trick macrophages and other immune cells into attacking cancer cells.
In 2014, researchers in Israel started a small-scale early-stage clinical trial looking at the dosage and safety of GcMAF in cancer patients. The full protocol and further information are available on the Clinicaltrials.gov register. [Updated KA 25/07/14]
To sum up
The advent of the internet has led to a wild proliferation of stories of ‘miracle cures’ for cancer – virtually all of which are based on shaky (or zero) science.
Some companies are selling Gc-MAF for use by cancer patients. This treatment is not approved or licensed in the UK for treating cancer or any 0ther disease. Given that there is no solid scientific evidence to show that the treatment is safe or effective, we would not recommend that cancer patients use it. [Updated KA 25/07/14]
Cancer is an extremely complex disease. In fact, it is more than 200 distinct diseases, each requiring different treatment. And the success of treatment depends on many things, including the genetic make-up of the tumour, the stage of diagnosis, and how aggressive the cancer is.
To suggest that there is a ‘magic bullet’ that cures all cancers is simplistic in the extreme.
http://retractionwatch.com/2014/10/10/yet-another-study-of-widely-touted-cancer-cure-retracted/
Retraction Watch
Yet
another study of widely touted cancer “cure” retracted
A third study of GcMAF, a protein being used to treat a variety of conditions from AIDS to autism to cancer, all without the blessing of health agencies, has been retracted.
Here’s the notice in Cancer Immunology, Immunotherapy for “Immunotherapy of metastatic colorectal cancer with vitamin D-binding protein-derived macrophage-activating factor, GcMAF:
"This article has been retracted by the Journal’s Editors-in-Chief in conjunction with the Publisher (Springer) due to irregularities in the Institutional Review Board documentation."
The paper has been cited 28 times, according to Thomson Scientific’s Web of Knowledge.
As we wrote in another post about Nobutu Yamamoto’s work, we’ve noticed that retractions for IRB documentation problems are often a bit like jailing Al Capone on tax evasion: They’re the easiest charges to prove, but they’re likely the least of a study’s problems.
A website, GcMAF.eu, continues to hawk the results of treatment:
The results from all the diseases we list are astonishing, but in late stage cancer the clinics achieve an average of 25% tumour reduction per week. (We get that reduction with pancreatic cancer too.)
The other two retractions for Yamamoto were in the International Journal of Cancer and the Journal of Medical Virology.
https://gcmaf.se/
Here is a time lapse video of the 8th assay we do in our laboratory - our GcMAF activates macrophages that eat cancers cells. We are probably the only people in the world with this technology.
Watch the video to see what happens to cancer cells when GcMAF is added without macrophages. This again is a world first, and again it has been done in our laboratory. Within just 4 weeks a research abstract paper on our results has already been accepted for publication at this year’s Immunology Conference in California.
https://youtu.be/D1WZrnCcH24
https://www.youtube.com/watch?t=119&v=D1WZrnCcH24
Cancer
cells destroyed by First Immune GcMAF (gcmaf.eu)
http://www.anticancerfund.org/therapies/gcmaf
Warning: GcMAF has not been properly studied in clinical trials and its laboratory results still need to be confirmed independently. So far, all claims on the efficacy of this product have no solid scientific basis. Its marketing is illegal; and the activities of Immuno Biotech Ltd. (www.gcmaf.se formerly www.gcmaf.eu) formerly with contact address in Brussels are currently under investigation by different European regulatory authorities. The factory where GcMAF was being manufactured in the UK, was closed because it was found that the product was not being produced under Good Manufacturing Practice (GMP) standards. There were concerns on the sterility of the products and the equipment used to produce it. The blood plasma used to manufacture this product should not be injected to humans nor should it be used to produce drugs. Not only did UK authorities shut down the GcMAF factory, but importation of the product has also been banned to guarantee the wellbeing of patients.
Immuno Biotech Ltd. opened three centers (www.immunocentre.eu), one in Switzerland and two purportedly in Germany and The Netherlands. The Swiss center was closed down by Swiss authorities earlier this year.
In contrast to the statements made by David Noakes from Immuno Biotech Ltd., GcMAF needs to be investigated in randomized clinical trials and it is currently being investigated in a registered clinical trial that complies with the established guidelines for the first time. This trial is a phase I carried out by a company independent of Noakes’s operation (NCT02052492).
GcMAF is a protein claimed to cure cancer and other ailments, but no proper clinical studies have been ever performed to confirm this claim.
Three clinical studies, by Nobuto Yamamoto et al., are referred as the initial evidence that GcMAF can cure cancer. However, after a thorough review of this work and discussion with experts, many flaws have been identified. Specially the use of an invalid endpoint to evaluate treatment response: the measurement of an enzyme in blood called Nagalase. After months of trying to get additional information on the patients and scientists involved in this research we came to the conclusion that these data should not be relied on since there are important issues in the methodology and procedures. The same group has also presented their results to different scientific conferences and we could confirm that one co-author’s participation was denied by the person himself, while we could not contact others besides Nobuto Yamamoto. The editors of the journals where Yamamoto et al. published their cancer-related articles were informed on the irregularities and flaws on this research. The articles claiming that GcMAF is effective to treat breast and colorectal cancer have been retracted, as reported here and here. Unfortunately due to lack of interest from the Editorial Board of the Translational Oncology Journal on discussing the flaws and irregularities in the prostate cancer article, it is still part of the official literature.
Other groups have reported their results treating cancer patients with GcMAF, but they used the same invalid methods to measure the response to treatment than Yamamoto, Nagalase test in blood. In recent articles they based their conclusions on ultrasound imaging, which is specifically advised against by the RECIST criteria as a way to measure tumor response. These groups are also involved in the illegal marketing of GcMAF. So far, we could not find any evidence that their products are being manufactured according to Good Manufacturing Practices (GMP) guidelines and have been properly tested for safety for human consumption according to the Good Clinical Practices (GCP) guidelines.
http://www.jleukbio.org/content/73/2/209.short
The
many faces of macrophage activation
by
David M. Mosser
David M. Mosser
INTRODUCTION
It used to be easy. In the old days (~8 years ago), activated macrophages were simply defined as cells that secreted inflammatory mediators and killed intracellular pathogens. Things are becoming progressively more complicated in the world of leukocyte biology. Activated macrophages may be a more heterogenous group of cells than originally appreciated, with different physiologies and performing distinct immunological functions. The first hint of this heterogeneity came with the characterization of the “alternatively activated macrophage” [1]. The exposure of macrophages to interleukin (IL)-4 or glucocorticoids induced a population of cells that up-regulated certain phagocytic receptors but failed to produce nitrogen radicals [2] and as a result, were relatively poor at killing intracellular pathogens. Recent studies have shown that these alternatively activated cells produce several components involved in the synthesis of the extracellular matrix (ECM) [3], suggesting their primary role may be involved in tissue repair rather than microbial killing. It turns out that the name alternatively activated macrophage may be unfortunate for a few reasons. First, although these cells express some markers of activation, they have not been exposed to the classical, activating stimuli, interferon-? (IFN-?) and lipopolysaccharide (LPS). Second, and more importantly, the name implies that this is the only other way to activate a macrophage. Recent studies suggest that this may not be the case. Exposure of macrophages to classical activating signals in the presence of immunoglobulin G (IgG) immune complexes induced the production of a cell type that was fundamentally different from the classically activated macrophage. These cells generated large amounts of IL-10 and as a result, were potent inhibitors of acute inflammatory responses to bacterial endotoxin [4]. These activated macrophages have been called type 2-activated macrophages [5] because of their ability to induce T helper cell …
http://onlinelibrary.wiley.com/doi/10.1002/jmv.21376/abstract
Retracted:
Immunotherapy of HIV-infected patients with Gc
protein-derived macrophage activating factor (GcMAF)
Nobuto Yamamoto, Naofumi Ushijima, Yoshihiko Koga
Nobuto Yamamoto, Naofumi Ushijima, Yoshihiko Koga
Abstract
The above article, published online on 21 Nov 2008 Wiley Online Library (wileyonlinelibrary.com), has been retracted by agreement between Dr. Ari Zuckerman, Editor-in-Chief, Journal of Medical Virology and Wiley Periodicals, Inc. due to irregularities in the documentation for institutional review board approval.
http://onlinelibrary.wiley.com/doi/10.1002/ijc.23107/abstract
Retracted:
Immunotherapy of metastatic breast cancer patients with
vitamin D-binding protein-derived macrophage activating
factor (GcMAF)
Nobuto Yamamoto, Hirofumi Suyama, Nobuyuki Yamamoto, Naofumi Ushijima
Nobuto Yamamoto, Hirofumi Suyama, Nobuyuki Yamamoto, Naofumi Ushijima
Abstract
The above article from the International Journal of Cancer, published online on 12 October 2007 in Wiley Online Library and in Volume 122, Issue 2, pp 461–467, has been retracted by agreement between the journal Editor-in-Chief Peter Lichter and Wiley Periodicals, Inc. due to irregularities in the documentation for institutional review board approval.
[ Excerpt ]
https://clinicaltrials.gov/ct2/show/NCT02052492?term=gcmaf&rank=1
Safety
Study of GcMAF (Globulin Component Macrophage Activating
Factor) in Subjects With Advanced Solid Tumors
Activated macrophages, present in excess during natural inflammatory responses, bear the potential to kill and eradicate cancer cells. Macrophage activation has been demonstrated to require the serum-borne vitamin D binding protein (known as Gc protein), as well as B and T lymphocytes. However, in various cancer patients the Gc protein is deglycosylated by serum a-N-acetyl galactosaminidase (Nagalase) secreted from cancer cells. This deglycosylated Gc protein, lacking the N-acetylgalactosamine monosaccharide, cannot be converted to its form of Macrophage Activating Factor, leading to immunosuppression rather than Macrophage activation against cancer cells.
Efranat has developed cancer immunotherapy based on Macrophage Activating Factor produced from natural Gc protein extracted from FDA approved healthy human plasma.
In this phase I study, the treatment is given as Intramuscular, once-weekly injection of GcMAF, for two courses, while each course is comprised of 4 injections.
The investigational treatment is expected to enhance immune response, thereby, improve patient's well being, quality of life and disease control.
Primary objectives:
To determine the safety and tolerability of GcMAF and to define the Maximal Tolerated Dose (MTD)
To identify the Dose Limiting Toxicity (DLT) of GcMAF
Secondary objectives:
To determine the 'Recommended Phase 2 Dose' (RP2D) based on MTD data, immunological and pharmacodynamic markers
.
To explore the preliminary efficacy of GcMAF in advanced solid tumors, considering the 'Immune-related Response Criteria' (irRC), the 'Response Evaluation Criteria in Solid Tumors' (RECIST) and blood levels of tumor-related markers known to reflect tumor burden.
Exploratory objectives:
To assess levels of immune-related factors in peripheral blood, reflecting induced immunological activities.
To immunohistochemically assess and compare tumor derived tissue samples Pre and post treatment. To analyze the infiltration of different population of cells into the tumor bed...
Detailed Description:
Part 1:
Eligible subjects will be assigned, successively in order of accrual, to one of the three cohorts, to receive intramuscular (IM) injections of GcMAF, once weekly for two courses of treatment. Each course will consist of 4 injections with one week intervals (total: 8 weeks of treatment).
Dose escalation will only proceed in the absence of dose-limiting toxicity (DLT) during course 1. For this purpose, each cohort will only begin its first course of GcMAF when the cohort preceding it has successfully completed its first 4-week treatment course without any signs of DLT. During this first course, should 1/3 patients experience DLT, dose escalation for the next cohort will not be authorized; the next cohort will receive the same dose as the one preceding it. If 2 patients or more of all patients treated with a given dose develop DLT, dose escalation will be halted and no more patients will be treated at the DLT dose. The value of MTD will be defined as the GcMAF dose below the dose at which DLT was seen for at least 2 subjects. Upon determination of the MTD, an additional cohort will be opened (confirmatory cohort) and treated with two courses of that dose.
Part 2:
Once MTD is established, or, when the last cohort completes its first course of treatment without an established MTD, an additional cohort will be opened in order to treat up to additional subjects with a selected dose of GcMAF already confirmed to be safe in part 1 of the study.
Continued treatment after completion of course 2 will be as per the investigator's discretion. Patient follow-up will continue for 12 months after start of treatment (Day 1). Patients for whom progressive disease (PD) is observed prior to completion of those two courses will be followed up only until PD...

Nobuto Yamamoto
Prof Nobuto Yamamoto is the pioneer and discoverer of the GcMAF glycoprotein and has conducted over 30 years of extensive research on its anti-cancer traits. Dr. Yamamoto has been a visiting Scientist at Fox Chase Cancer Center in Philadelphia where he studied the genetic evolution of bacterial viruses (1959-1961) He has been a scientist at Biological Standards NIH (1962-1964) and became Chief of Virology and Genetics of The Fels Institute at the Temple University School of Medicine in Philadelphia where he served from 1964 up till 1980 and again from 1990 till 1994. In 1980 Dr. Yamamoto was appointed Professor of Microbiology and Immunology at Hahnemann University School of Medicine, Philadelphia. (1980-1990) In this role he continued his research on immunology which emphasized mechanism of macrophage activation and discovered GcMAF.In 1994 he founded the Socrates Institute for Therapeutic Immunology, Philadelphia (1994 to date) where he continues to study the therapeutic efficacy of GcMAF for a variety of cancers and HIV. And in 2009 became one of the founders and board member of Efranat.
[ NOTE : The following article touts the work of Dr Burzynski re: Antineoplastons vs Cancer. Dr B. has however brought much of his problem upon himself because ( according to the FDA ) of his :
Enrollment of subjects into antineoplaston study protocols prior to the protocol-specified interval following prior chemotherapy and/or radiation therapy.
Failure to report all serious adverse events (SAEs) and adverse events (AEs) to the agency and/or IRB.
Failure to follow proper informed consent procedures.
Failure to maintain adequate drug accountability records.
Discrepancies between case report forms and source documents.
Failure to keep a copy of the study protocol and informed consent form.
Failure to receive and/or require progress reports from the principal investigator for the study.
Failure to receive and/or require a final report from the principal investigator for the study prior to removal from the IRB’s active list of studies.
Failure to assure that FDA approval was obtained by the principal investigator for the study prior to the treatment of a patient under a special exception.
Approval of special exceptions via expedited review.
The IRB approved research without determining that the following criteria were met: That risks to subjects were minimized and that risks to subjects were reasonable in relation to anticipated benefits, if any, to subjects, and the importance of the knowledge that may be expected to result.
The IRB failed to prepare, maintain, and follow written procedures for conducting its initial and continuing review of research.
The IRB failed to ensure that informed consent would be sought from each prospective subject or the subject’s legally authorized representative.
The IRB failed to ensure that no member participated in the initial or continuing review of a project in which the member had a conflicting interest.
The IRB failed to conduct continuing reviews.
And :
The IRB failed to follow FDA regulations regarding expedited review procedures [21 CFR 56.110(b)].
The IRB approved research without determining that the following criteria were met: risks to subjects were minimized [21 CFR 56.111(a)(1)]; risks to subjects were reasonable in relation to anticipated benefits, if any, to subjects, and the importance of the knowledge that may be expected to result [21 CFR 56.111(a)(2)].
The IRB failed to determine at the time of initial review that studies involving children are in compliance with 21 CFR part 50, subpart D, Additional Safeguards for Children in Clinical Investigations [21 CFR 56.109(h)]. This is a repeat violation from our 2010 inspection.
The IRB failed to prepare, maintain, and follow written procedures and maintain adequate documentation governing the functions and operations of the IRB [21 CFR 56.108(a), 21 CFR 56.108(b), and 21 CFR 56.115(a)(6)].
For more information -- http://www.rexresearch.com/burzynski/burzynski.htm ]
[ Excerpt ]
http://anh-europe.org/news/how-maverick-cancer-treatments-are-suppressed-by-the-mainstream
How
Maverick Cancer Treatments are Suppressed by the
Mainstream
Gc-MAF: antineoplastons mark II?
...Back in 1993, Nobuto Yamamoto, then working at Temple University School of Medicine in Philadelphia, PA, USA, first described a remarkable molecule. His paper reported the conversion of vitamin D3 binding protein (DBP, known in humans as Gc) into a potent macrophage-activating factor (MAF), known as Gc-MAF. Macrophages are a key part of the human immune system with two roles: to engulf and destroy pathogens and cellular debris, and to recruit other immune cells to respond to the pathogen.
Macrophages are crucial to both innate, or non-specific, immunity and adaptive, or specific, immunity. Under normal circumstances, Gc-MAF is upregulated when the immune system detects a threat, ‘activating’ macrophages so that they single-mindedly pursue pathogens. Cancer cells, a prime target of macrophages, are clever little critters that secrete an enzyme – alpha-N-acetylgalactosaminidase or nagalase – that inactivates Gc-MAF, thus preventing macrophages from becoming activated and protecting cancer cells. Administration of Gc-MAF is proposed to bypass nagalase, stimulating macrophages to become activated and attack tumour cells. In other words, it’s a potent immunomodulator, rather like antineoplastons.
So why haven’t most of us heard of it?
Unlike antineoplastons, however, Gc-MAF hasn’t had the benefit of a single patent owner – as a natural molecule, it cannot be patented without being modified – with the will and resources to push it under the noses of the public and health authorities. Dr Yamamoto has run small human trials in breast, prostate and colorectal cancers, with promising results. However, he is by no means alone, as David Noakes is at pains to point out: “There’s better research than Dr Yamamoto’s out there these days, and it’s all listed on our website,” he says.
David Noakes might just be the person to bring Gc-MAF into the mainstream. He’s the CEO of Immuno Biotech Ltd. and spokesperson for First Immune Gc-MAF, a project he describes as, “PhD and BSc biochemists and biomedical scientists...with external doctors, oncologists and scientists who kindly provide advice, committed to bringing some of the increasing number of published but relatively unused medical cures to as many people as we can.” At the moment, Noakes and his colleagues are supplying Gc-MAF to 30 countries where it is legal, via a network of “around 300” doctors. Their Gc-MAF is made to extremely high standards, and is being used in ongoing clinical research by Noakes’ collaborators and others. Their ultimate goal is to, “Build the case that GcMAF is effective for various illnesses, which will help to make it available to the public”.
An uphill struggle
Noakes has no illusions about the struggle he faces, as he explained to ANH-Intl. “Doctors, cancer doctors especially, know what they’re up against in countries like the US and UK. In all of the US, there’s only one doctor – Jeffrey Bradstreet, MD – who’s so far been prepared to put his head above the parapet about Gc-MAF. Many more take a cautious but pragmatic attitude; they might say to their patients, “Get it, but please don’t tell me!””
Getting nowhere in Guernsey
Recently, Noakes asked the Guernsey authorities whether they would allow First Immune Gc-MAF to produce the product in peace, in their own laboratory, hoping to take advantage of the island’s historically liberal attitude and exemption from EU laws. “We even offered to treat Guernsey kids with autism or cancer with Gc-MAF, for free – and anyone with infectious diseases for one-third of the normal cost. But they said no!” he reports in disgust. “Guernsey has even declared Gc-MAF an illegal drug, unlike anywhere else in the world. It’s come to something when Dubai is a better environment for the production and distribution of a potentially life-saving treatment than Guernsey, the UK or USA, but there you go.”...
PATENTS
& PATENT APPLICATIONS


WO2014199373
COMPOSITIONS COMPRISING GC- MACROPHAGE ACTIVATING FACTOR AND USES THEREOF
RU2198218
METHOD OF VITAMIN D3-BINDING PROTEIN (PROTEIN GC) CLONING...
FIELD:
molecular biology, protein engineering, medicine. SUBSTANCE:
invention relates to strong factors of macrophages activation.
Vitamin D3-binding protein (Gc-protein) and its small domain
(about 1/5 of Gc-peptide) that is known as domain III also) is
cloned using baculovirus vector. The cloned Gc-protein and
cloned peptide domain III (Cd) are treated with immobilized
beta-galactosidase and sialidase and factors of activation of
macrophages GcMAFc and CdMAF, respectively, are prepared. These
cloned factors of activation of macrophages and GcMAF can be
used as adjuvants in immunization and vaccination. Invention
allows to treat sicknesses, for example, osteopetrosis. EFFECT:
improved method of cloning and preparing, valuable medicinal
properties.METHOD OF VITAMIN D3-BINDING PROTEIN (PROTEIN GC) CLONING...
US2014030215
/ JP2014511857 / CN103547280 / WO2012137199
Macrophage activating factor for pharmaceutical compositions
The present invention
relates to pharmaceutical compositions comprising macrophage
activating factor (MAF) and method of producing same,
particularly to MAF compositions essentially devoid of
glycosidase enzymes. The compositions of the present invention
and pharmaceutical compositions comprising same are particularly
suitable for intravenous administration. Thus according to one
aspect, the present invention provides a composition
comprising,Gc protein-derived macrophage activating factor
(GcMAF), wherein the composition is essentially devoid of
glycosidase enzymes.Macrophage activating factor for pharmaceutical compositions
Vitamin D-based complexes for use as supplements
GB2515347
Vitamin D-based complexes for use as supplements
Vitamin D based
preparations are disclosed comprising a complex of
de-glycosylated vitamin D-binding protein (also called GcMAF),
vitamin D3 or its analogues, and at least on unsaturated fatty
acid. The preparations may include an aqueous alcoholic saline
solvent, and the unsaturated fatty acid can be oleic acid or
eicosapentaenoic acid. Orally administrable compositions
comprising the aforementioned preparations encapsulated in
liposomes are also disclosed, along with methods for the
production of the aforementioned preparationsVitamin D-based complexes for use as supplements
US2013129670
MACROPHAGE ACTIVATING FACTOR FOR USE IN THE TREATMENT OF CHRONIC FATIGUE SYNDROME (CFS) AND CFS-RELATED DISEASES AND DISORDERS
The present invention
relates to Macrophage Activating Factors such as GcMAF and
compositions thereof, for use in the treatment of a patient
suffering from CFS/ME and/or XMRV infection.MACROPHAGE ACTIVATING FACTOR FOR USE IN THE TREATMENT OF CHRONIC FATIGUE SYNDROME (CFS) AND CFS-RELATED DISEASES AND DISORDERS
US2011123591
Tumoricidal, bactericidal, or viricidal macrophage activation
The activation of
macrophages and methods for treating cancer, bacterial pathogens
and viral pathogens are disclosed. In particular, Gc protein is
converted to Gc-macrophage activating factor (GcMAF), in vivo or
ex vivo. The GcMAF activates macrophages which can then target
cancer cells, bacterial pathogens and/or viral pathogens.
Alternatively, macrophages are activated by contacting them, in
vivo or ex vivo, with GcMAF. Optionally, nagalase is inactivated
in a patient receiving the present macrophage activating
treatment by contacting the patient's blood with a
Nagalase-binding ligand immobilized on an inert medium.Tumoricidal, bactericidal, or viricidal macrophage activation
WO2012029954
PROCESS FOR PRODUCTION OF NOVEL DEGALACTOSYLATED GC GLOBULIN
This invention
provides a Gc globulin derivative that can be easily produced
from Gc globulin and can be used as GcMAF. This invention also
provides degalactosylated Gc globulin obtained by processing Gc
globulin with -galactosidase.PROCESS FOR PRODUCTION OF NOVEL DEGALACTOSYLATED GC GLOBULIN
WO2016162867
COMBINATION THERAPY OF MACROPHAGE ACTIVATING FACTOR AND PD-1 SIGNALING INHIBITORS
Inventor: SHAHAR
MICHAL, et al.COMBINATION THERAPY OF MACROPHAGE ACTIVATING FACTOR AND PD-1 SIGNALING INHIBITORS
The present invention relates to methods for treating cancer or infectious diseases comprising administering to a subject an agent that reduces or inhibits signal transduction mediated by PD-1 in combination with Gc protein derived macrophage activating factor (GcMAF). Particularly, the present invention provides methods of treating cancer comprising administering to a subject an anti-PD-1 antibody and GcMAF.
US2011123591
TUMORICIDAL BACTERICIDAL OR VIRICIDAL MACROPHAGE ACTIVATION
Inventor: KNEZEVICH
CHARLES, et al.TUMORICIDAL BACTERICIDAL OR VIRICIDAL MACROPHAGE ACTIVATION
The activation of macrophages and methods for treating cancer bacterial pathogens and viral pathogens are disclosed. In particular Gc protein is converted to Gc-macrophage activating factor (GcMAF) in vivo or ex vivo. The GcMAF activates macrophages which can then target cancer cells bacterial pathogens and/or viral pathogens. Alternatively macrophages are activated by contacting them in vivo or ex vivo with GcMAF. Optionally nagalase is inactivated in a patient receiving the present macrophage activating treatment by contacting the patient’s blood with a Nagalase-binding ligand immobilized on an inert medium.
US2015361151
MACROPHAGE ACTIVATING FACTOR FOR PHARMACEUTICAL COMPOSITIONS
Inventor(s): YAMAMOTO
NOBUTOMACROPHAGE ACTIVATING FACTOR FOR PHARMACEUTICAL COMPOSITIONS
The present invention relates to pharmaceutical compositions comprising macrophage activating factor (MAF) and method of producing same, particularly to MAF compositions essentially devoid of glycosidase enzymes. The compositions of the present invention and pharmaceutical compositions comprising same are particularly suitable for intravenous administration.
US
5712104 / US6410269
Preparation of potent macrophage activating factors derived from cloned vitamin D binding protein...
Preparation of potent macrophage activating factors derived from cloned vitamin D binding protein...
Nobuto
Yamamoto
Vitamin D-binding protein (Gc protein) and its small domain (approximately [1/5] of the Gc peptide also known as domain III) were cloned via a baculovirus vector. The cloned Gc protein and the cloned domain (Cd) peptide were treated with immobilized beta-galactosidase and sialidase to yield macrophage activating factors, GcMAFc and CdMAF, respectively. These cloned macrophage activating factors and GcMAF are to be used for therapy of cancer, HIV-infection and osteopetrosis, and may also be used as adjuvants for immunization and vaccination.
FIELD OF THE INVENTION
[0001] This invention relates to potent macrophage activating factors, prepared by oligosaccharide digestion of the cloned vitamin D binding protein (Gc protein) and the cloned Gc protein domain III, and the use of these macrophage activating factors for various cancer, HIV-infection and osteopetrosis, and as adjuvants for immunization and vaccination.
TABLE OF TERMS
[0002] Gc protein Vitamin D3-binding protein
[0003] MAF macrophage activating factor
[0004] GcMAF Gc protein-derived macrophage activating protein
[0005] GcMAFc cloned Gc protein-derived macrophage activating factor
[0006] Gc domain III domain III region of Gc protein
[0007] CdMAF cloned domain III-derived macrophage activating factor
SUMMARY OF THE INVENTION
[0008] Vitamin D-binding protein (Gc protein) and its small domain (approximately [1/5] of the Gc peptide also known as domain III) were cloned via a baculovirus vector. The cloned Gc protein and the cloned domain (Cd) peptide were treated with immobilized [beta]-galactosidase and sialidase to yield macrophage activating factors, GcMAFc and CdMAF, respectively. These cloned macrophage activating factors and GcMAF are to be used for therapy of cancer, HIV-infection and osteopetrosis, and may also be used as adjuvants for immunization and vaccination.
DESCRIPTION OF THE DRAWINGS
[0009] Other objects and many attendant features of this invention will become readily appreciated as the same becomes better understood by reference to the following detailed description when considered in connection with the accompanying drawings wherein:
[0010] FIG. 1a is a schematic illustration of the formation of macrophage activating factor (MAF).
[0011] FIG. 1b is a schematic illustration of the deglycosylation of Gc protein in a cancer or HIV-infected patient's blood stream.
[0012] FIG. 2 shows the correlation between plasma [alpha]-N-acetylgalactosaminidase activity and tumor burden (total cell counts) in the peritoneal cavity of Ehrlich ascites tumor.
[0013] FIG. 3 shows the amino acid sequence of cloned GcMAF which is SEQ ID NO:1 which is the entire cloned Gc protein.
[0014] FIG. 4 shows the construction of the DNA fragment encoding the leader sequence of EcoRi fragment E1 and domain III regions of the Gc protein; A, the entire cDNA for Gc protein; B, the construct to be inserted into the non-fusion vector; the shaded area indicates the compressed regions of about 1,000 base pairs (bp).
[0015] FIG. 5 shows the 89 amino acid sequence, SEQ ID NO:2, of the cloned domain III (CdMAF1), using the non-fusion vector.
[0016] FIG. 6 shows the baculovirus fusion vector for cloning the domain III of Gc protein.
[0017] FIG. 7 shows the 94 amino acid sequence, SEQ ID NO:3, of the cloned domain III (CdMAF2), using the fusion vector.
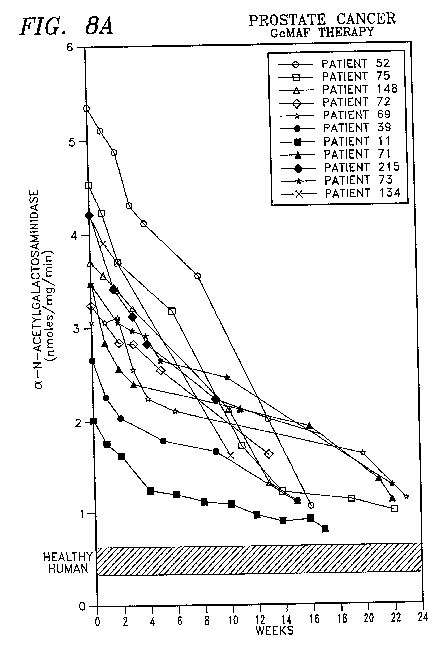
[0018] FIG. 8A shows the therapeutic effect of GcMAF in accordance with the present invention on adult persons suffering from prostate cancer.
[0019] FIG. 8B shows the therapeutic effect of GcMAF in accordance with the present invention on adult persons suffering from breast cancer.
[0020] FIG. 8C shows the therapeutic effect of GcMAF in accordance with the present invention on adult persons suffering from colon cancer.
[0021] FIG. 8D shows the therapeutic effect of GcMAF in accordance with the present invention on adult persons suffering from leukemia.










BACKGROUND OF THE INVENTION
[0022] A. Inflammatory Response Results in Activation of Macrophages
[0023] Inflammation results in the activation of macrophages. Inflamed lesions release lysophospholipids. The administration into mice of small doses (5-20 [mu]g/mouse) of lysophosphatidylcholine (lyso-Pc) and other lysophospholipids induced a greatly enhanced phagocytic and superoxide generating capacity of macrophages (Ngwenya and Yamamoto, Proc. Soc. Exp. Biol. Med. 193:118, 1990; Yamamoto et al, Inf. Imm. 61:5388, 1993; Yamamoto et al., Inflammation. 18:311, 1994).
[0024] This macrophage activation requires participation of B and T lymphocytes and serum vitamin D binding protein (DBP; human DBP is known as Gc protein). In vitro activation of mouse peritoneal macrophages by lyso-Pc requires the step-wise modification of Gc protein by 3-galactosidase of Iyso-Pc-treated B cells and sialidase of T cells to generate the macrophage activating factor (MAF), a protein with N-acetylgalactosamine as the remaining sugar moiety (FIG. 1a (Yamamoto et al., Proc. Natl. Acad. Sci. USA. 88:8539, 1991; Yamamoto et al., J. Immunol. 151:2794, 1993; Naraparaju and Yamamoto, Immunol. Lett. 43:143, 1994). Thus, Gc protein is a precursor for MAF.
[0025] Incubation of Gc protein with immobilized [beta]-galactosidase and sialidase generates a remarkably high titered MAF (GcMAF) (Yamamoto et al., Proc. Natl. Acad. Sci. USA. 88:8539, 1991; Yamamoto et al., J. Immunol. 151:2794, 1993; Naraparaju and Yamamoto, Immunol. Lett. 43:143, 1994; U.S. Pat. No. 51,177,002). Administration of a minute amount (10 pg/mouse; 100 ng/human) of GcMAF resulted in greatly enhanced phagocytic and super-oxide generating capacities of macrophages.
[0026] When peripheral blood monocytesimacrophages (designated as macrophages hereafter) of 258 cancer patients bearing various types of cancer were treated in vitro with 100 pg GcMAF/ml, macrophages of all cancer patients were activated for phagocytic and superoxide generating capacity. This observation indicates that cancer patient macrophages are capable of being activated. However, the MAF precursor activity of plasma Gc protein was lost or reduced in approximately 70% of this cancer patient population. Loss of the MAF precursor activity prevents generation of MAF. Therefore, macrophage activation cannot develop in certain cancer patients. Since macrophage activation is the first step in the immune development cascade, such cancer patients become immunosuppressed. This may explain at least in part why cancer patients die from overwhelming infection. Lost or reduced precursor activity of Gc protein was found to be due to deglycosylation of plasma Gc protein by [alpha]-N-acetylgalactosaminidase detected in cancer patient blood stream. Deglycosylated Gc protein cannot be converted to MAF (FIG. 1b.
[0027] Similarly, when peripheral blood macrophages of 160 HIV-infected/AIDS patients were treated in vitro with 100 pg GcMAF/ml, macrophages of all patients were activated for phagocytic and superoxide generating capacity. However, the MAF precursor activity of plasma Gc protein was low in approximately 35% of the HIV-infected patient population. As in cancer patients, these patients' plasma Gc protein is deglycosylated by [alpha]-N-acetylgalactosaminidase detected in HIV-infected patients.
[0028] Both cancer and HIV-infected patients having severely decreased precursor activity of plasma Gc protein carried large amounts of [alpha]-N-acetylgalactosaminidase while patients having moderately decreased precursor activity had moderate levels of plasma [alpha]-N-acetylgalactosaminidase activities. Patients with high precursor activity, including asymptomatic HIV-infected patients, had low but significant levels of plasma [alpha]-N-acetylgalactosaminidase activity. Since a large amount (260 [mu]g/ml) of Gc protein exists in the blood stream, a low level of the enzyme does not affect the precursor activity. Nevertheless, [alpha]-N-acetylgalactosaminidase activity was found in plasmas of all cancer and HIV-infected patients and had an inverse correlation with the precursor activity of their plasma Gc protein (Yamamoto et al., AIDS Res. Human Ret. 11:1373, 1995). Thus, increase in patient plasma [alpha]-N-acetylgalactosaminidase activity is responsible for decrease in the precursor activity of plasma Gc protein. These observations lead us to propose that plasma [alpha]-N-acetylgalactosaminidase plays a role in immunosuppression in cancer and HIV-infected/AIDS patients.
[0029] B. The Origin of Immunosuppression
[0030] The source of the plasma [alpha]-N-acetylgalactosaminidase in cancer patients appeared to be cancerous cells. High [alpha]-N-acetylgalactosaminidase activities were detected in tumor tissue homogenates of various organs, including eleven different tumor tissues including 4 lung, 3 breast, 3 colon and 1 cervix tumors, though the [alpha]-N-acetylgalactosaminidase activity varied from 15.9 to 50.8 nmoles/mg/min. Surgical removal of malignant lesions in human cancer results in subtle decrease of plasma [alpha]-N-acetylgalactosaminidase activity with concomitant increase in the precursor activity, particularly if malignant cells are localized.
[0031] In a preclinical mouse tumor model, BALB/c mice were transplanted with 5*10<5 >Ehrlich ascites tumor cells/mice into peritoneal cavity and analyzed for serum [alpha]-N-acetylgalactosaminidase activity. When plasma enzyme level were measured as transplanted Ehrlich ascites tumor grew in mouse peritoneal cavity, the enzyme activity was directly proportional to tumor burden as shown in FIG. 2. This was also confirmed with nude mouse transplanted with KB cells (human oral squamous cell carcinoma cell line). Serum [alpha]-N-acetylgalactosaminidase activity increased as tumor size (measured by weight) of the solid tumor increased. Thus, I have been using plasma [alpha]-N-acetylgalactosaminidase activity as a prognostic index to monitor the progress of therapy.
[0032] Radiation therapy of human cancer decreased plasma [alpha]-N-acetylgalactosaminidase activity with a concomitant increase of precursor activity. This implies that radiation therapy decreases the number of cancerous cells capable of secreting [alpha]-N-acetylgalactosaminidase. These results also confirmed that plasma [alpha]-N-acetylgalactosaminidase activity has an inverse correlation with the MAF precursor activity of Gc protein. Even after surgical removal of tumor lesions in cancer patients, most post-operative patients carried significant amounts of [alpha]-N-acetylgalactosaminidase activity in their blood stream. The remnant cancerous lesions in these post-operative patients cannot be detectable by any other procedures, e.g., X-ray, scintigraphy, etc. I have been using this most sensitive enzyme assay as prognostic index during the course of GcMAF therapy for treating cancer.
[0033] HIV-infected cells appeared to secrete [alpha]-N-acetylgalactosaminidase. When peripheral blood mononuclear cells (PBMC) of HIV-infected patients were cultured and treated with mitomycin as a provirus inducing agent (Sato et al., Arch. Virol. 54:333, 1977), [alpha]-N-acetylgalactosaminidase was secreted into culture media. These results led us to suggest that [alpha]-N-acetylgalactosaminidase is a virus coded product. In fact, HIV-envelope protein gp120 appears to carry the [alpha]-N-acetylgalactosaminidase activity.
[0034] C. A Defect in Macrophage Activation Cascade Manifests Osteopetrosis
[0035] An inflammation-primed macrophage activation cascade has been defined as a major process leading to the production of macrophage activating factor. Activation of other phagocytes such as osteoclasts shares the macrophage activation cascade (Yamamoto et al., J. Immunol. 152:5100, 1994). Thus, a defect in the macrophage activation cascade results in lack of activation in osteoclasts.
[0036] Autosomal recessive osteopetrosis is characterized by an excess accumulation of bone throughout the skeleton as a result of dysfunctional osteoclasts, resulting in reduced bone resorption (Marks, Clin. Orthop. 189:239, 1984). In animal models of osteopetrosis, depending on the degree of osteoclast dysfunction, marrow cavity development and tooth eruption are either delayed or more commonly absent (Marks, Am. J. Med. Genet. 34:43, 1989). In human infantile osteopetrosis, death occurs within the first decade of life usually overwhelming infection (Reeves, Pediatrics. 64:202,1979), indicating immunosuppression. Accumulated evidence suggests that deficient or dysfunctional osteoclasts in osteopetrotic animals are often accompanied by deficiencies or dysfunctions of macrophages. The studies of the present inventor on the activation of both osteoclasts and macrophages in the osteopetrotic mutations revealed that osteoclasts and macrophages can be activated by a common signaling factor, the macrophage activating factor and that a defect in [beta]-galactosidase of B cells incapacitates the generation process of macrophage activating factor (Yamamoto et al., J. Immunol. 152:5100, 1994). Since GcMAF and its cloned derivatives bypass the function of lymphocytes and Gc protein and act directly on macrophages and osteoclasts, administration of these factors into osteopetrotic hosts should rectify the bone disorder. In fact the present inventor has recently found that four administrations of purified cloned human macrophage activating factor (GcMAFc) (100 pg/week) to the p mutant mice beginning at birth for four weeks resulted in the activation of both macrophages and -osteoclasts and subsequent resorption of the excess skeletal matrix.
[0037] D. Therapeutic Application of GcMAF and its Cloned Derivatives on Cancer
[0038] Despite defects in the macrophage activation cascade in cancer, HIV-infected and osteopetrotic patients, GcMAF bypasses the functions of lymphocytes and Gc protein and acts directly on macrophages (or osteoclasts) for activation. Macrophages have a potential to eliminate cancerous cells and HIV-infected cells when activated. When cancer patients were treated with 100 ng GcMAF/patient weekly for several months, GcMAF showed remarkable curative effects on a variety of human cancer indiscriminately.
[0039] Instead of obtaining of GcMAF from human blood source, it can be obtained from the cloned Gc protein or its small domain responsible for macrophage activation. The cloning Gc protein require an eukaryotic vector/host capable of the glycosylation of the cloned products. The Gc protein having a molecular weight of 52,000 and 458 amino acid residues) is a multi-functional protein and carries three distinct domains (Cooke and Haddad, Endocrine Rev., 10:294,1989).
[0040] Domain I interacts with vitamin D while domain III interacts with actin (Haddad et al., Biochem., 31:7174, 1992). Chemically and proteolytically fragmented Gc enabled me to indicate that the smallest domain, domain ill, contains an essential peptide for macrophage activation. Accordingly, I cloned both Gc protein and the entire domain III peptide, by the use of a baculovirus vector and an insect host, and treated them with the immobilized [beta]-galactosidase and sialidase to yield potent macrophage activating factors, designated GcMAFc and CdMAF, respectively. Like GcMAF, these cloned GcMAFc and CdMAF appear to have curative effects on cancer.
[0041] E. A Potent Adjuvant Activity of GcMAF for Immunization with Antigens or Vaccines
[0042] Macrophages are antigen presenting cells. Macrophages activated by GcMAF rapidly phagocytize target antigens or cells and presented the processed antigens to antibody producing cells. I observed a rapid development of a large amount of antibody secreting cells immediately (1 to 4 days) after inoculation of small amount of GcMAF (100 pg/mouse) and sheep erythrocytes (SRBC). This finding indicates that GcMAF and its cloned derivatives, GcMAFc and CdMAF, should serve as potent adjuvants for immunization and vaccination.
DESCRIPTION OF THE METHODS FOR GENE CLONING FOR MACROPHAGE ACTIVATING FACTORS
[0043] A. Cloning of the cDNA of Gc Protein into an Insect Virus.
[0044] A full length cDNA encoding the human Gc protein was isolated from a human liver cDNA library in bacteriophage [lambda]gt11 (Clontech, Palo Alto, Calif.) by the use of pico Blue(TM) immunoscreening kit available from Stratagene of La Jolla, Calif. The baculoviral expression system in the insect cells takes advantages of several facts about the polyhedron protein: (a) it is expressed to very high levels in infected cells where it constitutes more than half of the total cellular protein late in the infection cycle; (b) it is nonessential for infection or replication of the virus, meaning that the recombinant virus does not require any helper function; (c) viruses lacking polyhedron gene have distinct plaque morphology from viruses containing the cloned gene; and d) unlike bacterial cells, the insect cell efficiently glycosylate the cloned gene products.
[0045] One of the beauties of this expression system is a visual screen allowing recombinant viruses to be distinguished and quantified. The polyhedron protein is produced at very high levels in the nuclei of infected cells late in the viral infection cycle. Accumulated polyhedron protein forms occlusion bodies that also contain embedded virus particles. These occlusion bodies, up to 15 [mu]m in size, are highly refractile, giving them a bright shiny appearance that is readily visualized under a light microscope. Cells infected with recombinant viruses lack occlusion bodies. To distinguish recombinant virus from wild-type virus, the transfection supernatant (recombinant containing virus lysate) is plaqued onto a monolayer of insect cells. Plaques are then screened under a light microscope for the presence (indicative of wild-type virus) or absence (indicative of recombinant virus) of occlusion bodies.
[0046] Unlike bacterial expression systems, the baculovirus-based system is an eukaryotic expression system and thus uses many of the protein modification, processing such as glycosylation, and transport reactions present in higher eukaryotic cells. In addition, the baculoviral expression system uses a helper-independent virus that can be propagated to high titers in insect cells adapted for growth in suspension cultures, making it possible to obtain large amounts of recombinant protein with relative ease. The majority of the overproduced protein remains soluble in insect cells by contrast with the insoluble proteins often obtained from bacteria. Furthermore, the viral genome is large (130 kbp) and thus can accommodate large segments of foreign DNA. Finally, baculoviruses are noninfectious to vertebrates, and their promoters have been shown to be inactive in mammalian cells (Carbonell et al., J. Virol. 56:153, 1985), which gives them a possible advantage over other systems when expressing oncogenes or potentially toxic proteins.
[0047] 1) Choice of Baculoviral Vector.
[0048] All available baculoviral vectors are pUC-based and confer ampicillin resistance. Each contains the polyhedron gene promoter, variable lengths of polyhedron coding sequence, and insertion site(s) for cloning the foreign gene of interest flanked by viral sequences that lie 5' to the promoter and 3' to the foreign gene insert. These flanking sequences facilitate homologous recombination between the vector and wild-type baculoviral DNA (Ausubel et al., Current Protocols in Mol. Biol. 1990). The major consideration when choosing the appropriate baculoviral expression vector is whether to express the recombinant as a fusion or non-fusion protein. Since glycosylation of Gc peptide requires a leader signal sequence for transfer of the peptide into the endoplasmic reticulum, the cDNA containing initiation codon (-16 Met) through the leader sequence to the +1 amino acid (leu) of the native Gc protein should be introduced to non-fusion vector with a polylinker carrying the EcoRI site, pLV1393 (Invitrogen, San Diego, Calif.).
[0049] During partial digestion of the cDNA for Go protein in [lambda]gt11 with EcoRI enzyme, a full length Gc cDNA with EcoRi termini was isolated electrophoretically, mixed with EcoRI-cut pVL1393, and ligated with T4 ligase. This construct in correct orientation should express the entire Gc peptide, a total of 458 amino acids (FIG. 3). To obtain the correct construction, competent E. coli HB101 cells were transformed with pVL vector and selected for transformants on Luria broth agar plates containing ampicillin (LB/ampicillin plates). The DNA was prepared for the sequencing procedure to determine which colony contains the insert or gene with proper reading orientation, by first searching for the 3' poly A stretch. The clones with 3' ply A (from the poly A tail of mRNA) were then sequenced from the 5' end to confirm the correct orientation of the full length DNA for the Gc peptide.
[0050] 2) Co-transfection of Insect Cells with the Cloned Plasmid DNA and Wild-type Viral DNA
[0051] A monolayer (2.5*10<6 >cells in each of 25-cm<2 >flasks) of Spodoptera frugiperda (Sf9) cells was co-transfected with a cloned plasmid (vector) DNA (2 [mu]g) and a wild-type (AcMNPV) baculoviral DNA (10 [mu]g) in 950 [mu]l transfection buffer (Ausubel et al., In Curr Protocols in Mol. Biol. 1990). When the cells were cultured for 4 or 5 days, the transfection supernatant contained recombinant viruses.
[0052] 3) Identification of Recombinant Baculovirus
[0053] The co-transfection lysates were diluted 10<4>, 10<5 >or 10<6 >and plated on Sf9 cells for cultivation for 4 to 6 days. After the plaques were well formed, plaques containing occlusion-negative cells were identified at a frequency of 1.3%. Several putative recombinant viral plaques were isolated and twice re-plaqued for purification. Pure recombinant viral plaque clones were isolated.
[0054] B. Analysis of Protein of Interest from Recombinant Baculovirus
[0055] 1) Preparation of Recombinant Virus Lysate
[0056] An insect cell Sf9 monolayer (2.5*10<6 >cells per 25-cm<2 >flask) was infected with a recombinant virus clone and cultured in 5 ml GIBCO serum-free medium (from GIBCO Biochemicals, Rockville, Md.) or medium supplemented with 0.1% egg albumin to avoid contamination of serum bovine vitamin D binding protein. The culture flasks were incubated at 27[deg.] C. and monitored daily for signs of infection. After 4 to 5 days, the cells were harvested by gently dislodging them from the flask and the cells and culture medium were transferred to centrifuge tubes and centrifuged for 10 min at 1000* g, 4[deg.] C. To maximize infection for recombinant protein production, Sf9 cells were grown in a 100-ml spinner suspension culture flask with 50 ml complete medium up to about 2*10<6 >cells/ml. The cells were harvested, centrifuged at 1000* g for 10 min and re-suspended in 10 to 20 ml serum-free medium containing recombinant virus at a multiplicity of infection (MOI) of 10. After 1 hour of incubation at room temperature, the infected cells were transferred to a 200-ml spinner flask containing 100 ml serum-free medium and incubated for 40 hr. More than 40% of the protein secreted was the protein of interest. The protein in the supernatant was isolated.
[0057] 2) Qualitative Estimation of the Protein of Interest
[0058] Coomassie Blue staining of the SDS-polyacrylamide gel, loading 20 to 40 [mu]g total cell protein per lane, was to estimate quantity of expressed protein. Because the samples contain cellular proteins, the recombinant protein was readily detected by comparison with uninfected cellular proteins.
[0059] 3) Enzymatic Conversion of the Cloned Gc Protein to Macrophage Activating Factor (GcMAFc).
[0060] The cloned Gc protein (2 [mu]g) with a molecular weight of 52,000 and 458 amino acid residues (FIG. 3) was isolated by electroeluter and treated with immobilized [beta]-galactosidase and sialidase. The resultant cloned macrophage activating factor (GcMAFc) was added to mouse and human macrophages and assayed for phagocytic and superoxide generating capacity. Incubation of macrophages with 10 pg GcMAFc/ml for 3 hours resulted in a 5-fold increased phagocytic and a 15-fold increase in the superoxide generating capacity of macrophages.
[0061] C. Subcloning of a Domain Required for Macrophage Activation
[0062] I. Cloning Procedure I: Non-fusion Vector.
[0063] 1) Cloning the Domain Responsible for Macrophage Activation (CdMAF)
[0064] The entire cDNA sequence for Gc protein in [lambda]gt11, including 76 bp of the upstream 5' flanking region and 204 bp of the 3' flanking stretch, was fragmented by EcoRi to yield four restriction fragments designated E1, 120; E2, 314; E3, 482; and E4, 748 bp, respectively. Each was cloned into the EcoRI site of the plasmid pSP65 from Promega (Madison, Wis.) by the method of Cooke and David (J. Clin. Invest., 76 2420, 1985). Although I found that a region less than one half of the domain III was found to be responsible for macrophage activation, small segments less than 40 amino acid residues cannot be expressed in the insect cells. Moreover, short peptides are rapidly degraded by proteases in human plasma, and thus are not clinically useful. Accordingly, the entire domain III (approximately 80 amino acid residues) should be subcloned into an insect virus where I anticipate the efficient production and glycosylation of the peptide in the infected cells.
[0065] 2) Subcloning cDNA Fragment into the Polyhedron Gene of Baculovirus.
[0066] Since the glycosylation of a peptide requires a leader signal sequence for transfer of the peptide into the endoplasmic reticulum, the DNA segment of E1 containing the initiation codon (-16 Met) through the leader sequence to the +1 amino acid (Leu) of the native Gc protein should be introduced into the vector. Because this segment carries the initiation codon for the Gc protein, non-fusion vector, pVL1393 (Invitrogen, San Diego, Calif.) was used. A segment containing the initiation codon-leader sequence of the cDNA clone E1 and a segment coding for 85 C-terminal amino acids (the entire domain III plus 3' non-coding stretch) of the cDNA clone E4 were ligated together and cloned into the EcoRI site of the insect virus pVL vector. To achieve this construct, both E1 and E4 DNA were fragmented with HaeIII to yield two fragments each; E1hl (87 bp), E1hs (33 bp) and E4hs (298 bp), E4hl (450 bp), respectively. Both the larger fragments E1hl and E4hl were isolated electrophoretically, mixed with EcoRI-cut pVL, and ligated with T4 ligase, as shown in FIG. 4. This construct in correct orientation should express the entire domain III, a total of 89 amino acids, including the 4 amino acids of E1hl, also referred to herein as CdMAF1 as shown in FIG. 5. To obtain the correct construction, competent E. coli HB101 cells are transformed with pVL vector and selected for transformants on LB/ampicillin plates. DNA was prepared for sequencing procedures to determine which colony contains the construct with proper reading orientation by first searching for the 3' poly dA stretch. Those clones with 3' poly dA (from the poly A tail of mRNA) were then sequenced from the 5' end to confirm correct orientation of the E1hl fragment. I found that the vector contains the entire construct (domain III) in the correct orientation.
[0067] 3) Isolation of Recombinant Baculovirus, Purification of the Cloned Domain Peptide (Cd) and Enzymatic Generation of the Cloned Macrophage Activating Factor (CdMAF)
[0068] Monolayers (2.5*10<6 >cells in each of 25-cm<2 >flasks) of Spodoptera frugiperda (Sf9) cells were co-transfected with cloned plasmid DNA (2 [mu]g) and wild-type (AcMNPV) baculoviral DNA (10 [mu]g) in 950 [mu]l transfection buffer. Recombinant baculovirus plaques were isolated and used for production of the Gc domain III peptide in insect cells. This cloned domain with a molecular weight (MW) of 10,000 and 89 amino acids as shown in FIG. 5, was purified electrophoretically. Two [mu]g of the cloned domain (Cd) peptide was treated with immobilized [beta]3-galactosidase and sialidase to yield a cloned macrophage activating factor, designated as CdMAF1.
[0069] II. Cloning Procedure II: Fusion Vector.
[0070] 1) Cloning the Domain Responsible for Macrophage Activation (CdMAF)
[0071] A baculovirus fusion vector, pPbac vector (Stratagene, La Jolla, Calif.), contains human placental alkaline phosphatase secretory signal sequences that direct the nascent cloned peptide chain toward the secretory pathway of the cells leading to secretion into culture media. The signal sequence is cleaved off by signal-sequence peptidase as the nascent cloned peptide is channeled toward the secretory pathway of the host insect cells leading to secretion of the cloned domain (Cd) peptide. FIG. 6 depicts that the vector carries the stuffer fragment for gene substitution and lacZ gene for identification of the gene insertion.
[0072] The stuffer fragment of pPbac vector was excised by digesting the vector DNA with restriction enzymes Smal and BamHI and was removed by electroelution. The E4 cDNA fragment of the Gc protein was digested with HaeIII and BamHI, yielding a fragment practically the same as E4hl (see FIG. 4). This fragment was mixed with the above pPbac vector and ligated with T4 ligase. This strategy not only fixes the orientation of ligation but also fuses the fragment with the reading frame. The E. coli DH5aF' cells were transformed with the reaction mixture. The cloned DNA insert was isolated from a number of colonies after digestion with HaeIII and BamHI. The insert was confirmed by sequencing. The sequence confirmed the correct orientation.
[0073] 2) Isolation of Recombinant Baculovirus by Transfection of Sf9 Insect Cells with Wild Type Baculovirus and the Cloned DNA Insert.
[0074] For transfection of insect cells (Spodoptera frugiperda, Sf9), linear wild type (AcMNPV) baculoviral DNA and insectin liposomes (Invitrogen, San Diego, Calif.) have been used. Liposome-mediated transfection of insect cells is the most efficient transfection method available. For transfection to a monolayer of Sf9 cells (2*10<6>) in a 60 mm dish a mixture of the following was gently added:
[0075] 3 [mu]g cloned plasmid DNA
[0076] 10 [mu]l linear wild type baculovirus (AcMNPV) DNA (0.1 [mu]g/[mu]l)
[0077] 1 ml medium
[0078] 29 [mu]l insectin liposomes
[0079] The dishes were incubated at room temperature for 4 hours with slow rocking. After transfection, the 1 ml of medium was added and incubated at 27[deg.] C. in a humidified environment for 48 hours. The resultant transfection lysate was plaque assayed. Purification of recombinant virus, isolation of the cloned domain peptide (Cd) and enzymatic generation of the cloned macrophage activating factor designated CdMAF2 were described in the Cloning Procedure I. This CdMAF is composed of 94 amino acid residues as shown in FIG. 7, including 9 amino acids from the fusion vector and is referred to herein as CdMAF2. Although CdMAF2 has five amino acids more than the CdMAF1 peptide derived from the non-fusion vector, they exhibited the same biological activities.
Supporting Observations
[0080] A. Effects of Cloned Macrophage Activating Factors, GcMAFc and CdMAF on Cultured Phagocytes (Macrophages and Osteoclasts).
[0081] The three hour treatment of human macrophages and osteoclasts with picogram quantities (pg) of the cloned macrophage activating factors, GcMAFc and CdMAF, resulted in a greatly enhanced superoxide generating capacity of the phagocytes as shown in Table 1. The levels of the phagocyte activation are similar to those of macrophage activation by GcMAF (Yamamoto et al., AIDS Res. Human Ret. 11:1373, 1995).
TABLE 1
Activation of phagocytes by in vitro treatment with GcMAF and its cloned derivatives.
nmole of superoxide produced/min/10<6 >phagocytes
Conc. Mouse peritoneal Human
pg/ml Human macrophages* macrophages osteoclasts
GcMAFc
0 0.07 0.06 0.03
10 3.20 3.46 2.56
100 5.18 5.08 4.22
CdMAF
0 0.01 0.02 0.08
10 2.96 2.87 2.43
100 4.26 4.53 4.09
*Peripheral blood monocytes/macrophages of cancer patients. Similar results were also observed when those of HIV-infected patients were used.
[0082] B. Activation of Mouse Peritoneal Macrophages by Administration of Cloned Macrophage Activating Factors, GcMAFc and CdMAF.
[0083] One day post-administration of a picogram quantity (10 and 100 pg/mouse) of GcMAFc or CdMAF to BALB/c mice, peritoneal macrophages were isolated and assayed for superoxide generating capacity. As shown in Table 2, the macrophages were efficiently activated. These results are similar to those of macrophage activation with GcMAF (Naraparaju and Yamamoto, Immunol. Lett. 43:143, 1994; Yamamoto et al., AIDS Res. Human Ret. 11:1373,1995).
TABLE 2
Activation of mouse peritoneal macrophages by administration of cloned GcMAF derivatives.
Dosage nmole of superoxide produced/min/10<6 >phagocytes
pg/mouse Mouse peritoneal macrophages
0 0.05
10 3.18
100 5.23
CdMAF
0 0.03
10 2.54
100 4.23
[0084] C. Therapeutic Effects of GcMAF, GcMAFc or CdMAF on Tumor Bearing Mice and Osteopetrotic Mice.
[0085] 1) Therapeutic Effects of GcMAF, GcMAFc or CdMAF on Ehrlich Ascites Tumor Bearing Mice.
[0086] When BALB/c mice were administered with GcMAF, GcMAFc or CdMAF (100 pg/mouse) and received 10<5 >Ehrlich ascites tumor cells/mouse, they survived for at least 5 weeks. All the control mice received only the ascites tumor and died in approximately 14 days. When mice were administered with an additional 100 pg GcMAF/mouse 4 days post-transplantation, the tumor cells were completely eliminated (Table 3).
[0087] When mice were transplanted with 10<5 >Ehrlich ascites tumor cells/mouse and treated twice with GcMAF, GcMAFc or CdMAF (100 pg/mouse) at 4 days and 8 days post-transplantation, all treated mouse groups survived over 65 days while the untreated 8 mouse groups all died at approximately 13 days (Groups 4 through 9 of Table 3).
TABLE 3
Therapeutic effects of GcMAF and cloned derivatives on mice bearing Ehrlich ascites tumor.
No. of Post-transplantation No. of mice
Group mice treatment survived/period
Group 1.
6 mice untreated control 6 mice/13 ± 3 days
10 mice day 0 100 pg GcMAF/mouse 10 mice/36 ± 7 days
Group 2.
6 mice untreated control 6 mice/14 ± 4 days
10 mice day 0 100 pg GcMAFc/mouse 10 mice/35 ± 6 days
Group 3.
6 mice untreated control 6 mice/14 ± 5 days
10 mice day 0 100 pg CdMAF/mouse 10 mice/34 ± 3 days
Group 4.
8 mice untreated control 8 mice/15 ± 5 days
12 mice day 0 100 pg GcMAF/mouse
day 4 100 pg GcMAF/mouse 12 mice/>65 days
Group 5.
8 mice untreated control 8 mice/14 ± 5 days
12 mice day 0 100 pg GcMAFc/mouse
day 4 100 pg GcMAFc/mouse 12 mice/>65 days
Group 6.
8 mice untreated control 8 mice/14 ± 5 days
12 mice day 0 100 pg CdMAF/mouse
day 4 100 pg CdMAF/mouse 12 mice/>65 days
Group 7.
8 mice untreated control 8 mice/14 ± 4 days
8 mice day 4 100 pg GcMAF/mouse
day 8 100 pg GcMAF/mouse 8 mice/>65 days
Group 8.
8 mice untreated control 8 mice/13 ± 3 days
8 mice day 4 100 pg GcMAFc/mouse
day 8 100 pg GcMAFc/mouse 8 mice/>65 days
Group 9.
8 mice untreated control 8 mice/13 ± 5 days
8 mice day 4 100 pg CdMAF/mouse
day 8 100 pg CdMAF/mouse 8 mice/>65 days
[0088] With respect to the results of Table 3, GcMAF was administered intraperitoneally for Groups 1 through 6, and GcMAF was administered intramuscularly (systemically) for Groups 7 through 9; mice in all groups received 105 tumor cells/mouse.
[0089] 2) Therapeutic Effects of GcMAF and Cloned GcMAF Derivatives (GcMAFc and CdMAF) On Osteopetrotic Mice.
[0090] Administration of GcMAFc or CdMAF to new born litters of osteopetrotic op/op mouse was performed by the weekly injection of 100 picograms for four weeks beginning from a day after birth. Mice were sacrificed at 28 days. The tibiae were removed from the treated and untreated control mice, longitudinally bisected, and examined under a dissecting microscope to measure the size of the bone marrow cavity. The cavity size was expressed as a percentage of the distance between the epiphyseal plates of the tibia. The untreated mouse group formed bone marrow with 30% of the total length of tibia. The treated mouse group experienced a 20% increased bone marrow formation over that of the untreated mouse group. This increased bone marrow cavity formation is an indication of osteoclast activation and increased osteoclastic bone resorption.
[0091] D. Therapeutic Effects of GcMAF, GcMAFc and CdMAF on Human Cancer and Virus Infected Patients.
[0092] 1. Cancer Patients: Therapeutic Effect of GcMAF on Prostate, Breast and Colon Cancer and Adult Leukemia Patients.
[0093] The administration of GcMAF (100 and 500 ng/human) to healthy volunteers resulted in the greatly enhanced activation of macrophages as measured by the 7-fold enhanced phagocytic capacity and the 15-fold superoxide generating capacity of macrophages. The administration of GcMAF showed no signs of any side effects to the recipients. Administration of various doses (100 pg to 10 ng/mouse) to a number of mice produced neither ill effects nor histological changes in various organs including liver, lung, kidney, spleen, brain, etc. When patients with various types of cancer were treated with GcMAF (100 ng/week), remarkable curative effects on various types of cancer were observed. The therapeutic efficacy of GcMAF on patients bearing various types of cancers was assessed by tumor specific serum [alpha]-N-acetylgalactosaminidase activity because the serum enzyme level is proportional to the total amount of cancerous cells (tumor burden). Curative effects of GcMAF on prostate, breast and colon cancer and leukemia are illustrated in FIGS. 8A to 8D. After 25 weekly administrations of 100 ng GcMAF the majority (>90%) of prostate and breast cancer patients exhibited insignificantly low levels of the serum enzyme. A similar result was also observed after 35 GcMAF administrations to colon cancer patients. Similar curative effects of GcMAF on lung, liver, stomach, brain, bladder, kidney, uterus, ovarian, larynx, esophagus, oral and skin cancers were observed. Thus, GcMAF appeared to be effective on a variety of cancers indiscriminately. However, GcMAF showed no evidence of side effects in patients after more than 6 months of therapy. This was also confirmed by blood cell counts profile, liver and kidney functions, etc.
[0094] When GcMAFc (100 ng/week) and CdMAF (100 ng/week) were administered to two prostate cancer patients each, curative effects similar to those of GcMAF were observed.
[0095] 2. Virus Infected Patients
[0096] Treatment of peripheral blood macrophages of HIV-infected/AIDS patients with 100 pg GcMAF/ml resulted in a greatly enhanced macrophage activation (Yamamoto et al., AIDS Res. Human Ret. 11:1373, 1995). HIV-infected patients carry anti-HIV antibodies. HIV-infected cells express the viral antigens on the cell surface. Thus, macrophages have a potential to eliminate the infected cells via Fc-receptor mediated cell-killing/ingestion when activated.
[0097] Similarly, treatment of peripheral blood macrophages of patents chronically infected with Epstein-Barr virus (EBV) and with herpes zoster with 100 ng GcMAF/ml resulted in a greatly enhanced macrophage activation. Like HIV, EBV infects lymphocytes (B cells). Since these enveloped viruses code for [alpha]-N-acetylgalactosaminidase and infected cells secrete it into blood stream. Thus this enzyme activity in patient sera can be used as a prognostic index during therapy. After approximately 25 administrations of GcMAF (100 ng/week) to patients chronically infected with EBV and with herpes zoster, the enzyme activity decreased to that of healthy control levels. When GcMAFc (100 ng/week) and CdMAF (100 ng/week) were administered to EBV-infected patients, curative effects similar to those of GcMAF were observed.
[0098] E. Adjuvant Activities of GcMAF, GcMAFc and CdMAF for Immunization and Vaccinations.
[0099] 1. Rapid Increase of the Number of Antibody Secreting Cells (PFC) in Mice after Administration of GcMAF and Sheep Erythrocytes.
[0100] BALB/c mice were inoculated with SRBC 6 hours after the intraperitoneal administration of 50 pg GcMAF/mouse. At various intervals (1-5 days) after immunization, IgM-antibody secreting cells in the spleen were determined using the Jerne plaque assay (Jerne et al., Cell-bound antibodies, Wistar Institute Press, 1963). One day post-administration of GcMAF and SRBC produced 1.35*10<4 >PFC/spleen. Two days after administration of GcMAF and SRBC, the number of antibody secreting cells had increased to 8.2*10<4 >PFC/spleen. By the 4th day the number of antibody secreting cells reached the maximal level (about 23.6*10<4 >PFC/spleen), as shown in Table 4. In contrast, mice that received an injection of SRBC alone produced about 3.8*10<4 >PFC/spleen, 4 days after SRBC-injection.
[0101] To ascertain the dose response, mice were injected with SRBC 6 hours after administration of various doses of GcMAF ranging from 1 to 50 pg/mouse. On the 4th day post-administration of GcMAF and SRBC, the number of antibody secreting cells per spleen was determined by the Jerne plaque assay. On the 4th day post-administration there was a commensurate increase in the number of plaque forming cells as the concentration of GcMAF was increased above 1 pg per mouse. At a GcMAF dose of 5, 10 and 50 pg/mouse, I detected 12.6*10<4>, 20.2*10<4 >and 24.3*10<4 >PFC/spleen, respectively.
TABLE 4
Time course studies on development of cells secreting antibody against sheep erythrocytes (SRBC) in BALB/c mice after administration of GcMAF and SRBC
After SRBC
immunization Antibody secreting cells/spleen (*10<4>)
(days) SRBC only GcMAF + SRBC
1 0.01 ± 0.002 1.35 ± 0.21
2 0.08 ± 0.02 8.28 ± 1.26
3 1.18 ± 0.42 14.42 ± 2.32
4 3.86 ± 0.95 23.68 ± 6.05
5 2.15 ± 0.63 18.63 ± 3.43
<a>Mice were inoculated with SRBC (10<8 >cells) 6 hr after administration of GcMAF (50 pg/mouse). The number of plaques (IgM secreting cells) was quantified microscopically on various days post-SRBC injection. The number of plaque-forming cells (PFC) per spleen is expressed as the mean value of triplicate assays ± SEM.
[0102] Without further elaboration the foregoing will so fully illustrate my invention that others may, by applying current or future knowledge, adapt the same for use under various conditions of service.
REFERENCES CITED
[0103] The following references are cited and their entire text is incorporated fully herein as are all references set forth above in the specification.
U.S. Patent Documents
[0104] U.S. Pat. Nos. 5,177,001, 5,177,002 and 5,326,749 (Yamamoto).
Other Publications
[0105] 1. Jerne, N. K., Nordin, A. A. and Henry, C., The agar plaque technique for recognizing antibody producing cells. In Amos and Koprowski (eds). Cell-bound Antibody. Wistar Institute Press, Philadelphia, PA (1963).
[0106] 2. Sato, M., Tanaka, H., Yamada, T. and Yamamoto, N., Persistent infection of BHK/WI-2 cells with rubella virus and characterization of rubella variants. Arch. Virology 54:333-343 (1977).
[0107] 3. Reeves, J. D., August, C. S., Humbert, J. R., Weston, W. L, Host defense in infantile osteopetrosis. Pediatrics. 64:202-(1979).
[0108] 4. Marks, S. C., Jr., Congenital osteopetrotic mutations as probes of the origin, structure and function of osteoclasts. Clin. Orthop. 189:239-(1984).
[0109] 5. Carbonell, L. F., Klowden, M. J. and Miller, L. K., Baculovirus-mediated expression of bacterial genes in dipteran and mammalian cells. J. Virol. 56:153-160 (1985).
[0110] 6. Ngwenya, B. Z., and Yamamoto, N., Activation of peritoneal macrophages by lysophosphatidylcholine. Biochem. Biophys. Acta 839: 9-15 (1985).
[0111] 7. Cooke, N. E. and Haddad, J. G., Vitamin D binding protein (Gc-globulin). Endocrine Rev. 10:294-307 (1989).
[0112] 8. Marks, S. C., Jr., Osteoclast biology: Lessons from mammalian mutations. Am. J. Med. Genet. 34:43-54 (1989).
[0113] 9. Ngwenya, B. Z. and Yamamoto, N., Contribution of lysophosphatidylcholine treated nonadherent cells to mechanism of macrophage stimulation. Proc. Soc. Exp. Biol. Med. 193:118-124 (1990).
[0114] 10. Ausubel, F. A., Brent, R., Kingston, R. E., Moore, D. D., Seidman, J. G., Smith, J. A. and Struhl, K. (eds.), Expression of proteins in insect cells using baculoviral vectors. Current Protocols in Molecular Biology. Sections 16.8.1-16.11.7. Greene Publishing and Wiley-Interscience, New York (1990).
[0115] 11. Yagi, F., Eckhardt, A. E. and Goldstein I. J., Glycosidases of Ehrlich ascites tumor cells and ascitic fluid-purification and substrate specificity of [alpha]-N-acetylgalactosaminidase and [alpha]-galactosidase: comparison with coffee bean [alpha]-galactosidase. Arch. Biochem. Biophys. 280:61-67 (1990).
[0116] 12. Yamamoto, N. and Homma, S., Vitamin D3 binding protein (group-specific component, Gc) is a precursor for the macrophage activating signal from lysophosphatidylcholine-treated lymphocytes. Proc. Natl. Acad. Sci. USA. 88:8539-8543 (1991).
[0117] 13. Cooke, N. E. and David, E. V., Serum vitamin D-binding protein is a third member of the albumin and alpha-fetoprotein gene family. J. Clin. Invest. 76:2420-2424 (1985).
[0118] 14. Haddad, J. G., Hu, Y. Z., Kowalski, M. A., Laramore, C., Ray, K., Robzyk, P. and Cooke, N. E., Identification of the sterol- and actin-binding domains of plasma vitamin D binding protein (Gc-globulin). Biochemistry 31:71747181 (1992).
[0119] 15. Yamamoto, N. and Kumashiro, R., Conversion of vitamin D3 binding protein (Group-specific component) to a macrophage activating factor by stepwise action of [beta]-galactosidase of B cells and sialidase of T cells. J. Immunol. 151:27-94-2902 (1993).
[0120] 16. Homma, S., Yamamoto, M. and Yamamoto, N., Vitamin D binding protein (group-specific component, Gc) is the sole serum protein required for macrophage activation after treatment of peritoneal cells with lysophosphatidylcholine. Immunol. Cell Biol. 71:249-257 (1993).
[0121] 17. Yamamoto, N., Kumashiro, R., Yamamoto, M., Willett, N. P. and Undsay, D. D., Regulation of inflammation-primed activation of macrophages by two serum factors, vitamin D3-binding protein and albumin. Inf. Imm. 61:5388-5391 (1993).
[0122] 18. Yamamoto, N., Lindsay, D. D., Naraparaju, V. R., Irelalnd, R. A. and Popoff, S. M., A defect in the inflammation-primed macrophage activation cascade in osteopetrotic (op) rats. J. Immunol. 152:5100-5107 (1994).
[0123] 19. Yamamoto, N., Willett, N. P. and Lindsay, D. D., Participation of serum proteins in the inflammation-primed activation of macrophages. Inflammation. 18:311-322 (1994).
[0124] 20. Naraparaju, V. R. and Yamamoto, N., Roles of [beta]-galactosidase of B lymphocytes and sialidase of T lymphocytes in inflammation-primed activation of macrophages. Immunol. Lett. 43:143-148 (1994).
[0125] 21. Yamamoto, N., Naraparaju, V. R. and Srinivasula, S. M., Structural modification of serum vitamin D3-binding protein and immunosuppression in HIV-infected patients. AIDS Res. Human Ret. 11:1373-1378 (1995).