Robert KARAS
Ion Exchange Electrolysis Continuous Mining
( Salt ElectroMining )
Ion Exchange Electrolysis Continuous Mining
( Salt ElectroMining )
SEM TECH // ROWOW LLC
irowow@gmail.com
(407) 724-8687
https://www.sciencemadness.org/talk/viewthread.php?tid=161515#pid705578
New mining technology with closed loop membrane electrolysis [ PDF ]
https://www.youtube.com/watch?v=UMI_ITPgirI
New mining technology with closed loop membrane electrolysis
The video goes further in detail but its a open sourced mining technology I developed with our in house membranes. Its closed loop and eco friendly. I call it SEM TECH (salt electro mining) because it uses saltwater and electricity to mine... Very creative ya ya :P
The two active chemicals are sodium chlorate and hcl. The sodium chlorates are very powerful oxidizers capable of dissolving noble metals like rhodium and iridium. This is similar to people extracting PGMS from catalytic converters using bleach and hcl
So the loop goes as this, positive side acids get regenerated through a cation exchange membrane. The acid leaches and dissolves the heavy metals. The leached solution gets pumped into the negative side to be reduced and plate off the metals as a powder. This powder gets collected at the bottom of a cone tank. The negative depleted solution then goes back to the positive side to regenerate and repeat the cycle. Very simple
But implications are huge. Rare earth elements, precious metals, critical minerals. 53 out of 60 critical minerals can get concentrated in a single step. Most mining processes require high grade ore because the chemicals otherwise cost more than the material they extract. But this process generates the acids on site in situ. Furthermore the process can use cheap inputs like salt. Because the process is closed loop we get 99% recovery rates. There is no losses to occur. Cyanide leaching for example is 50% losses, so is froth flotation and even shaker tables suck at 20% efficiency. Our losses are with only a little left dissolved in the solution as electro plating cant remove 100% of ions. But the solution is simple by adding more ore we just keep adding ions solving that issue. The benefits go on and on. From testing ive done we are extracting at around $50 worth of electricity (at $0.15 per kwh) per ton of ore. And unlike other technologies that only can focus on gold/silver, we can extract the rest of the platinum group metals.
You may ask ok but what do we do with the concentrate powder. This power is in a economically concentrate form to be refined through various traditional methods. But thats where we step in again. We can run this powder through various selective solutions to separate individual or groups of metals and again through the same principle, dissolve, reduce, regenerate the acid. For example with acetic/nitric solution we can dissolve the base metals leaving behind gold platinum etc behind (of course silver and palladium also gets separated in this case with the base metals, but we can use different solutions for that in multiple stages)
The technology is just the start and I see a potential were we electro chemically separate each element in a single solution by simply flowing it through multiple cells. The technology has huge potential
https://goldrefiningforum.com/threads/update-on-my-open-source-salt-water-electro-mining-technology-gold-rhodium-platinum-rare-earths-etc.36831/
Update on my open source salt water electro mining technology, gold rhodium platinum rare earths etc
I started this project 2 years ago and open sourced it with a forum post here a year and a half ago. Heres a update on the technology and this past year has drastically expanded my view on the technology being developed realizing it has is world changing. This technology is going to be the standard. Industries either transition to using ion exchange membranes or get left behind. If you have any questions ill be more than glad to answer!
Yes its very much scalable. I have it as a 50 amp cell currently as a standard units. But my small scale testing are tiny cups at 1-2 amps. Any size you like to the thousands of amps. They can be wired in series also so technically can run directly off 120v with a bridge rectifier/capacitor setup. Each cell is 5-6 volts.
For the refining industry we have some very unique and creative ideas, but the fundamental basic "CRU" (continuous refining unit) we have built and used basically uses different acids for its selective dissolving properties. For example, once you have a dory bar, powder, etc, you can run it through a CRU unit with acetic/nitric acid. This dissolves most base metals (lead iron copper etc) and leaves the precious metals behind (except silver/palladium). I already tested this on a bar that had vanadium and was a pain to dissolve in nitric. It just wouldn't touch it. But through the CRU, I was able to continuously dissolve it completely automated. I didnt have to bother with fresh acids etc. The system regenerates the acid, leaches the material, pumps the leached solution to the negative side to plate off the metals, then the depleted solution flows back into the positive side to regenerate and repeat the cycle. This is all possible due to the ion exchange membrane between the negative and positive acting as a chemical diode, forcing reactions to go one way. Theres still room for improvement and optimization but as the technology stands it works and is economical. Mostly about scaling up to production, which costs alot of money, hopefully this grant will help me with that. I dedicated the past 2 years on this technology and have gotten so far on so little.
I have many units built and will attach pictures of my current design. It uses hot swappable electrodes and membranes, very easy to change out. The cost to refine per ton of material ends up around $50-400 a ton of iron. As a comparison, you would need 3.5 tons of nitric acid to dissolve that amount of iron. The cost benefits is immense. I have such a large range as my calculated costs are $50 per ton from a small test, but as a engineer I like to give lots of extra cushion just in case, which still makes it insanely game changing. Additionally with fire assay refining this has a huge benefit in being able to recover the lead into a precipitate powder for further reuse. I can go on and on.
That post I made a long time ago about recovering the gold is how I ended up down this journey and developed this process. Electro plating allowed me to recover but then I didn't like the cost of nitric acid so was searching for a alternative oxidizer and found sodium chlorates, which is how PGMS are extracted from catalytic converters. My CMU (continuous mining unit) uses HCL and Sodium chlorate, both chemicals derived from saltwater and electricity, and can dissolve noble metals like rhodium which aqua regia wont touch, all at room temperature.
Additionally if you look at my shorts I have tons of videos on the precipitate powder being produced from my system. I have a professional lab grade desktop xrf rigaku NED DE spectrometer verifying my results. (handheld XRF is a scam)
Do you digest the scrap to selectively dissolve out the non precious metals and what form does it dissolve from, bar, shot, or atomized particles?
Depending on which system used. I have currently two units, the CMU which uses HCL and Sodium Chlorate to dissolve pretty much everything. This is targeting for general extraction and recovery from ore, e waste, mining waste, etc. The intention is to concentrate down the heavy metals/rare earths/precious metals from rocks. This creates a metal powder.
The CRU uses more selective acids to separate out groups of metals for further refinement. It drastically reduced the amount of work, steps, and acid used in traditional refining and reduction.
https://www.youtube.com/watch?v=RdYTBQ42KMU
Safety and dangers of salt electro mining technology and chemistry in general
https://www.youtube.com/watch?v=CuFFl4NJDOw
Proof of Function: Our Ion Exchange Membrane Works (Cell Assembly + Test)
https://www.youtube.com/watch?v=c3tNXDlgE2M
DIY Low-Cost Ion Exchange Membrane: Full Build Guide & Open Source Dedication
Rowow details a DIY ion exchange membrane creation, using readily available materials and open-sourcing the design. Learn how to build these membranes for under $1/sq yd, from initial grinding to final application methods. Explore various techniques, including ball milling and spray application, suitable for diverse scales.
https://github.com/Rowow1/Open-sourced-off-the-shelf-ion-exchange-membrane
Open source off the Shelf Ion Exchange Membrane
https://rowow.net/
Introducing our open sourced electro chemistry technology to extract precious metals and rare earth metals using closed loop electrolysis techniques while also cleaning up the environment of heavy metal and other pollutions
https://rowow.net/methods/
Continuous Treatment
Acidic waste gets fed into the negative side where the heavy metals gets plated off, first platinum group metals and rare earth elements, then gold, then silver, and so forth down the electro voltage potential depending on each solution chemistry. Lead arsenic mercury and other heavy metals can be plated out of the solution electrochemically
Continuous Leaching
A completely continuous electro chemical leaching system that regenerates the acid for further leaching while simultaneously plating off the precious metals leached in the solution first then the heavy metals and other base metals as long as desired to run the system. This method converts toxic ore or e waste into valuable concentrate and rocks safe for use in gardens
Continuous Refining
Electrolysis of slag, metal bars and many other materials can be refined by plating off the base metals on the negative electrode and leaving behind precious metal precipitate that is inert and insoluble by the electrolytic solution. Without the complexity of common refining techniques and expensive chemicals and waste, this solutions allows for a continuous automated refining system with minimal to no additional maintenance.
https://www.gofundme.com/f/semtech
Open-Source Membrane for Sustainable Mining
Total fund raising attempt is at 10 million dollars. However the first important purchase is a VACUUM FILTRATION CONTINUOUS DRUM to start testing out a pilot scale automated ton per day system. This costs approximately $25,000 after tarrifs and shipping.
I've spent years developing a revolutionary ion exchange membrane using off-the-shelf ingredients like water softener resin and PVC cement. This homogeneous membrane is extremely affordable to make, yet it performs like commercial versions that cost much more. It's durable in extreme conditions (pH 0, high ORP) and enables closed-loop processes with zero toxic waste.
My primary focus right now is the mining industry: using it in Salt Electro Mining (SEM TECH) to extract precious metals and rare earths from ore, tailings, or e-waste while regenerating acids and oxidizers on-site. It cleans up legacy mine waste, recovers critical minerals domestically, and eliminates toxic ponds—real environmental impact.
I've already filed a non-provisional patent (App. No. 19/531,984) and dedicated the technology to the public domain under CC0—anyone can build, use, or improve it freely. The full recipe, build guide, and files are open-sourced on GitHub.
https://www.youtube.com/watch?v=luulTI1RKHE
DIY Ion-Exchange Membranes from Hardware Store Items // NightHawkInLight Extra
In this video I talk to Robert Karas of @Rowow (https://rowow.net/) who has invented and open sourced an ion exchange membrane that can be made for as little as $1 per square yard from common items. These membranes unlock some truly remarkable chemistry, from highly efficient metal ore processing, to dirt cheap flow battery energy storage, split output hydrogen electrolysis, high efficiency chemical production, methanol fuel cells, and the ability to run all these technologies on variable power sources like solar without efficiency loss. We talk about all this and more in this conversation about affordable ion exchange membranes.
Low-Cost
Homogeneous Ion Exchange Membrane Formed from Off-the-Shelf
Precursors and Manufacturing Process Thereof
Abstract -- A low-cost homogeneous ion exchange membrane is formed from pulverized commercial pre-functionalized ion exchange resin particles dispersed in a PVC or CPVC matrix. The membrane is manufactured by pulverizing resin beads into powder, mixing with dissolved PVC/CPVC in solvent to create a glue-like mixture, applying to a surface, and drying to form a peelable film. The process avoids post-functionalization, enabling costs less than $1 per square foot, high selectivity, durability in harsh conditions, and easy integration into electrochemical cells for applications including mining, energy storage, and chemical processing.
Background of the Invention
[0001] The present invention relates to ion exchange membranes, particularly homogeneous cation and anion exchange membranes for electrochemical applications, including but not limited to electrolysis cells, redox flow batteries, mining processes (e.g., acid regeneration and metal leaching), water treatment, and chemical production.
[0002] Ion exchange membranes enable selective transport of cations or anions under an applied electric field, facilitating direct electrochemical control of reactions with minimal waste. Applications span energy storage (e.g., redox flow batteries), resource recovery (e.g., precious metal extraction), agriculture (e.g., pH control in hydroponics), waste management, and general chemical synthesis.
[0003] Conventional ion exchange membranes are typically produced using specialized precursors and multi-step functionalization processes, such as sulfonation or amination of polymer backbones, often requiring hazardous chemicals (e.g., chlorosulfonic acid) or complex post-treatments. These methods result in high manufacturing costs (often hundreds of dollars per square foot), limited accessibility, and challenges in scalability or integration into electrolysis cells. Commercial membranes are expensive and not readily available to researchers or hobbyists, while makeshift alternatives (e.g., clay pots) exhibit poor selectivity, high resistivity, and mechanical/chemical limitations.
[0004] Prior art, such as US2948637A, relies on sulfonating agents to functionalize polymers, while US4851100A describes coating existing membranes to form bipolar structures rather than standalone homogeneous membranes. Other approaches involve melt-processing, additives, or post-functionalization, which add complexity and cost.
[0005] There remains a need for low-cost, homogeneous ion exchange membranes using readily available, off-the-shelf ingredients, with simple manufacturing that avoids post-functionalization and enables easy integration into cells
Brief Summary of the Invention
[0006] The present invention provides a homogeneous ion exchange membrane comprising pulverized commercial ion exchange resin particles dispersed in a polyvinyl chloride (PVC) or chlorinated polyvinyl chloride (CPVC) matrix, formed from a solvent-based mixture and dried into a film. The membrane utilizes pre-functionalized resins (e.g., strong/weak cation or anion exchange resins from water softeners), achieving high ion selectivity, low resistivity, and chemical durability at a cost less than $1 per square foot (or yard in thin applications).
[0007] The manufacturing process involves: (1) pulverizing commercial resin beads into fine powder; (2) mixing the powder with dissolved PVC/CPVC in a compatible solvent to form a homogeneous glue; and (3) applying the mixture to a surface, drying, and peeling to form the membrane. No post-functionalization, heating, or cross-linking is required.
[0008] Advantages include accessibility using household equipment (e.g., blender), compatibility with PVC/CPVC cements for cell sealing, tunable properties (e.g., resin type/ratio for selectivity), and applications in harsh environments (e.g., pH 0, high oxidizing potential).
Brief Description of the Drawings
FIG. 1 is a flowchart illustrating the manufacturing process for the homogeneous ion exchange membrane according to embodiments of the present invention, including optional dry and wet pulverizing paths, water removal options, and the mixing, application, and formation steps.
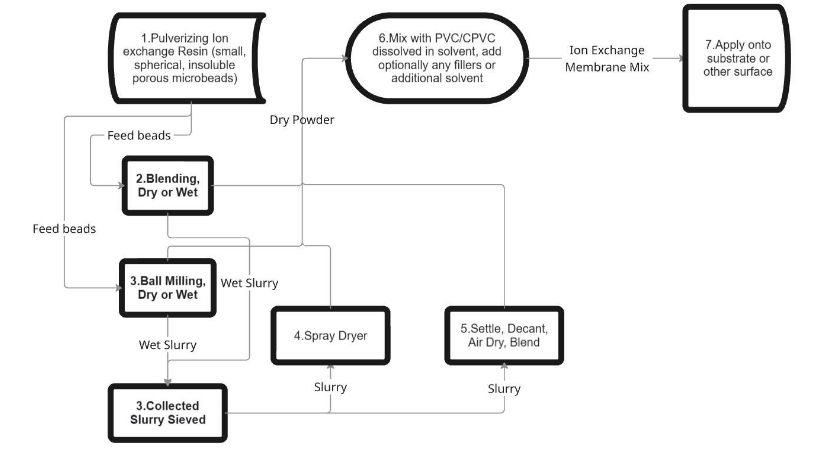
Detailed Description of the Invention
[0009]
The invention provides a homogeneous ion exchange membrane and
method of manufacture using off-the-shelf precursors. The
membrane is homogeneous, meaning uniform distribution of ion
exchange functionality throughout the structure without
distinct layers or heterogeneous particles.
[0010] The membrane comprises: (a) pulverized particles of commercial ion exchange resin (pre-functionalized, e.g., strong acid cation, weak acid cation, strong base anion, weak base anion, or mixtures thereof); and (b) a binder matrix of PVC or CPVC with a solvent.
[0011] As illustrated in FIG. 1, the overall manufacturing process begins with commercial ion exchange resins, which are typically spherical beads (small, insoluble, porous micro-beads used, for example, in water softening). These beads are pulverized into fine powder using mechanical shearing methods such as grinding (step 2, dry or wet), ball milling (step 3, dry or wet), or other grinding techniques. Wet processes may include water to assist lubrication. If water is introduced, it is removed to obtain dry powder or a controlled slurry before further processing.
[0012] Water removal from slurries can be accomplished by spray drying (step 4), settling/decanting followed by air drying and blending (step 5), or sieving collected slurries. These options are shown in the branched paths of FIG. 1. The resulting dry powder or suitably prepared material is then mixed with a solution of PVC or CPVC dissolved in a compatible solvent (e.g., tetrahydrofuran (THF), cyclohexanone, methyl ethyl ketone) (step 6). Preferred ratio: 10-70% resin powder by volume (typically ~50% for balanced conductivity and mechanical strength); polymer-to-solvent ratio ~3:7 (adjustable for desired viscosity). Optional additives (e.g., fiberglass, fumed silica, sand) may be included for structural enhancement. Additional solvent can be added to reduce viscosity for spraying or other application methods.
[0013] The homogeneous glue-like mixture (referred to as the "Ion Exchange Membrane Mix" in FIG. 1) is applied (step 7) to a non-adhesive, solvent-resistant surface (e.g., polypropylene sheet) or directly onto a substrate (e.g., polypropylene felt for reinforcement), cell frame, or electrode by spreading, extruding, spraying, pouring, or other suitable means. The applied mixture dries to form a peelable film or permanent coating, with thickness tunable by the application technique. Uneven drying is prevented (e.g., using an enclosed environment). The membrane may be partially dried to avoid cracking or warping before peeling or further handling.
[0014] Embodiments include: cation-selective membranes (using cation resins); anion-selective (using anion resins); specialized ion resins for selectivity (e.g., rare earths, platinum group metals); thin spray films (<$1/sq yard); coatings on electrodes or microporous ion selective separator (e.g., for lithium separation to block out larger sodium ions).
[0015] Best mode: Use water softener cation exchange resin beads, pulverize in a high-speed blender (<120 seconds), mix 50% powder with PVC in THF (3:7 polymer:solvent), spread on polypropylene sheet, dry partially to avoid cracking, peel. Result: flexible/rigid, watertight membrane with commercial-level conductivity/selectivity.
[0016] Testing in harsh conditions (acidic/alkaline mining solutions, high ORP) shows durability over months, low energy loss, and electro-osmosis. Cost: fraction of commercial membranes.
Claims
A homogeneous ion exchange membrane comprising: pulverized particles of a commercial pre-functionalized ion exchange resin dispersed in a matrix of polyvinyl chloride (PVC) or chlorinated polyvinyl chloride (CPVC).
The membrane of claim 1, wherein the ion exchange resin is selected from strong acid cation exchange resin, weak acid cation exchange resin, strong base anion exchange resin, weak base anion exchange resin, or mixtures thereof.
The membrane of claim 1, wherein the resin particles are pulverized to a particle size less than 200 microns.
The membrane of claim 1, wherein the resin particles comprise 10-70% by volume of the membrane.
A method of manufacturing a homogeneous ion exchange membrane, comprising:
a) pulverizing beads of a commercial pre-functionalized ion exchange resin into a fine powder;
b) mixing the powder with a solution of polyvinyl chloride (PVC) or chlorinated polyvinyl chloride (CPVC) in a solvent to form a homogeneous mixture; and
c) applying the mixture to a surface and drying to form a membrane composite.
The method of claim 5, wherein pulverizing is performed using a mechanical shearing device selected from a blender, grinder, or ball mill.
The method of claim 5, wherein the solvent is selected from tetrahydrofuran, cyclohexanone, or methyl ethyl ketone.
The method of claim 5, further comprising removing water from the powder prior to mixing if introduced during pulverizing.
The method of claim 5, wherein applying comprises spreading, spraying, extruding, or pouring.
The method of claim 5, wherein the membrane is peeled from the surface after partial drying or left as a permanent coating.
Sources: Capillary Electrolysis [ PDF ]