Paul D. ROBBINS, et al.
Fisetin vs Aging
Fisetin vs Aging
http://www.wikipedia.org
Fisetin


Fisetin, is a plant polyphenol from the flavonoid group. It can be found in many plants, where it serves as a colouring agent. It is also found in many fruits and vegetables, such as strawberries, apples, persimmons, onions and cucumbers.
Molar mass: 286.2363 g/mol
Formula: C15H10O6
Melting point: 626°F (330°C)
ChEMBL Id: 31574
ChemSpider ID: 4444933
PubChem CID: 5281614
https://www.sciencedaily.com/releases/2018/10/181002114024.htm
Researchers
have discovered how to slow aging
Natural product found to reduce the level of damaged cells in the body, caused by aging
Natural product found to reduce the level of damaged cells in the body, caused by aging
Summary: Previous research showed it was possible to reduce the burden of damaged cells, termed senescent cells, and extend lifespan and improve health, even when treatment was initiated late in life.
They now have shown that treatment of aged mice with the natural product Fisetin, found in many fruits and vegetables, also has significant positive effects on health and lifespan.
Previous research published earlier this year in Nature Medicine involving University of Minnesota Medical School faculty Paul D. Robbins and Laura J. Niedernhofer and Mayo Clinic investigators James L.
Kirkland and Tamara Tchkonia, showed it was possible to reduce the burden of damaged cells, termed senescent cells, and extend lifespan and improve health, even when treatment was initiated late in life.
They now have shown that treatment of aged mice with the natural product Fisetin, found in many fruits and vegetables, also has significant positive effects on health and lifespan.
As people age, they accumulate damaged cells. When the cells get to a certain level of damage they go through an aging process of their own, called cellular senescence. The cells also release inflammatory factors that tell the immune system to clear those damaged cells. A younger person's immune system is healthy and is able to clear the damaged cells. But as people age, they aren't cleared as effectively. Thus they begin to accumulate, cause low level inflammation and release enzymes that can degrade the tissue.
Robbins and fellow researchers found a natural product, called Fisetin, reduces the level of these damaged cells in the body. They found this by treating mice towards the end of life with this compound and see improvement in health and lifespan. The paper, "Fisetin is a senotherapeutic that extends health and lifespan," was recently published in EBioMedicine.
"These results suggest that we can extend the period of health, termed healthspan, even towards the end of life," said Robbins. "But there are still many questions to address, including the right dosage, for example."
One question they can now answer, however, is why haven't they done this before? There were always key limitations when it came to figuring out how a drug will act on different tissues, different cells in an aging body. Researchers didn't have a way to identify if a treatment was actually attacking the particular cells that are senescent, until now.
Under the guidance of Edgar Arriaga, a professor in the Department of Chemistry in the College of Science and Engineering at the University of Minnesota, the team used mass cytometry, or CyTOF, technology and applied it for the first time in aging research, which is unique to the University of Minnesota.
"In addition to showing that the drug works, this is the first demonstration that shows the effects of the drug on specific subsets of these damaged cells within a given tissue." Robbins said.
https://linkinghub.elsevier.com/retrieve/pii/S2352396418303736
EBioMedicine, 2018;
DOI: 10.1016/j.ebiom.2018.09.015
Fisetin
is a senotherapeutic that extends health and lifespan.
EBioMedicine, 2018; DOI: 10.1016/j.ebiom.2018.09.015
Matthew J. Yousefzadeh, et al.
AbstractMatthew J. Yousefzadeh, et al.
Background
Senescence is a tumor suppressor mechanism activated in stressed cells to prevent replication of damaged DNA. Senescent cells have been demonstrated to play a causal role in driving aging and age-related diseases using genetic and pharmacologic approaches. We previously demonstrated that the combination of dasatinib and the flavonoid quercetin is a potent senolytic improving numerous age-related conditions including frailty, osteoporosis and cardiovascular disease. The goal of this study was to identify flavonoids with more potent senolytic activity.
Methods
A panel of flavonoid polyphenols was screened for senolytic activity using senescent murine and human fibroblasts, driven by oxidative and genotoxic stress, respectively. The top senotherapeutic flavonoid was tested in mice modeling a progeroid syndrome carrying a p16INK4a-luciferase reporter and aged wild-type mice to determine the effects of fisetin on senescence markers, age-related histopathology, disease markers, health span and lifespan. Human adipose tissue explants were used to determine if results translated.
Findings
Of the 10 flavonoids tested, fisetin was the most potent senolytic. Acute or intermittent treatment of progeroid and old mice with fisetin reduced senescence markers in multiple tissues, consistent with a hit-and-run senolytic mechanism. Fisetin reduced senescence in a subset of cells in murine and human adipose tissue, demonstrating cell-type specificity. Administration of fisetin to wild-type mice late in life restored tissue homeostasis, reduced age-related pathology, and extended median and maximum lifespan.
Interpretation
The natural product fisetin has senotherapeutic activity in mice and in human tissues. Late life intervention was sufficient to yield a potent health benefit. These characteristics suggest the feasibility to translation to human clinical studies.
PATENTS
: FISETIN EXTRACTION
Method for extracting fisetin from toxicodendron succedaneum
CN108003125
The invention discloses a method for extracting fisetin from toxicodendron succedaneum. The method comprises the following operation steps: (1) taking toxicodendron succedaneum branches, crushing the toxicodendron succedaneum branches with a crusher, adding a sodium hydroxide solution, soaking for 1-2 h, taking out the soaked toxicodendron succedaneum branches, performing drying till the water content of the toxicodendron succedaneum branches is 10-15%; (2) adding ethyl alcohol into the dried toxicodendron succedaneum branches; performing heating for a reaction; performing extraction for 1.5-2.5 h at 180-220 DEG C, stopping heating, performing cooling to the room temperature, taking out a substance obtained after the reaction, performing filtration, and taking the filtrate; and (3) concentrating the filtrate obtained in step (2) till the total liquid content of the filtrate is 23-25 wt% to obtain a concentrated solution, adding water into the concentrated solution according to the massratio 1:(1.55-1.59) of the concentrated solution to water, and separating out fisetin. The method can effectively extract fisetin, the extracted fisetin is good in quality and high in purity, and theextraction ratio can be up to 19%; and furthermore, the method is easy and simple to operate, suitable for massive industrial production and safe in extraction process.
COSMETIC SKIN WHITENING COMPOSITIONS CONTAINING EXTRACTS DERIVED FROM IN VITRO PROPAGATED HYPERICUM HIRSUTUM
EP3135341
The present invention relates to extract containing active substances and use of extract as active ingredient for skin whitening cosmetic formulations. A skin whitening cosmetic active ingredient based on extract derived from in vitro cultivated Hypericum hirsutum by 50% ethanol extraction is proposed. Invention relates specifically to the plant material derived from in vitro shoot cultures with no growth regulators added. The offered ingredient contains Kaempferol-3-O-glucoside, Kaempferol-7-O-glucoside, Fisetin, Chlorogenic acid, 3-Caffeoylquinic acid and Umbelliferone. Invention proposes use of extract, which exhibits anti-proliferative effect on melanocytes and is not highly cytotoxic in concentration range of 1 -3 %, as active ingredient in skin whitening cosmetic formulations. The proposed concentration range for use in cosmetic formulations is 0.5-2% (v/v)
Method for extracting fisetin from boxwood
CN102924419
The invention provides a method for extracting fisetin from boxwood. The method comprises that boxwood as a raw material is subjected to crushing, extraction and crystallization to produce fisetin crystals. The method has the advantages that processes are simple and easy; equipment requirements are low and only simple extraction condensation equipment is adopted; extracted fisetin has the purity of 98%; a fisetin yield is greatly improved and 15 to 20kg of fisetin can be extracted from 1T of the raw material having the content of 1 to 2%; an active ingredient loss is less in the processes; and compared with the prior art, the method can improve a yield about 4-5 times.
Vernonia anthelmintica flavone components, preparation method and application thereof
CN102526153
The invention relates to vernonia anthelmintica flavone components, a preparation method and application thereof. The flavone components are fisetin, butein, 7,8,3',4'-tetrahydroxy flavone, 5,7,8,3',4'-pentahydroxy chalcone, 6,8,3',5'-tetrahydroxy-dihydroflavone, liquiritigenin and isoliquiritigenin, which are prepared from plant vernonia anthelmintica through extraction, separation and purification. Application of each flavone component in preparation of a medicament for treating leucoderma provides a new medicament choice for treating leucoderma.
Fisetin extraction method
CN102442987
The invention relates to a fisetin extraction method. The process method comprises the following steps: crushing dried branches and leaves of smoke trees; adding 10-20 times of saturated limewater solution, and soaking and extracting at normal temperature; filtering the extracting solution, regulating the pH value to 7, and adding a macroporous resin column for adsorption; eluting with a 60-70% ethanol solution; recovering ethanol in the eluent while depressurizing, standing for crystallization, and filtering out coarse crystals; dissolving in a 5% sodium carbonate solution; sequentially leaching with n-butyl alcohol and ethyl acetate; removing the organic phase, regulating the water phase with hydrochloric acid until the pH value is 4-5, and standing for precipitation; and recrystallizing the precipitate, and drying to obtain the product. The fisetin extraction method has the advantages of easy acquisition of raw materials, simple process operation and low production cost, and is applicable to industrial production.
https://www.ncbi.nlm.nih.gov/pubmed/28575152
J Gerontol A Biol Sci Med Sci. 2018 Mar 2;73(3):299-307. doi: 10.1093/gerona/glx104.
Fisetin
Reduces the Impact of Aging on Behavior and Physiology in
the Rapidly Aging SAMP8 Mouse.
Currais A, Farrokhi C, Dargusch R, Armando A, Quehenberger O, Schubert D, Maher P.
Currais A, Farrokhi C, Dargusch R, Armando A, Quehenberger O, Schubert D, Maher P.
Abstract
Alzheimer's disease (AD) is rarely addressed in the context of aging even though there is an overlap in pathology. We previously used a phenotypic screening platform based on old age-associated brain toxicities to identify the flavonol fisetin as a potential therapeutic for AD and other age-related neurodegenerative diseases. Based on earlier results with fisetin in transgenic AD mice, we hypothesized that fisetin would be effective against brain aging and cognitive dysfunction in rapidly aging senescence-accelerated prone 8 (SAMP8) mice, a model for sporadic AD and dementia. An integrative approach was used to correlate protein expression and metabolite levels in the brain with cognition. It was found that fisetin reduced cognitive deficits in old SAMP8 mice while restoring multiple markers associated with impaired synaptic function, stress, and inflammation. These results provide further evidence for the potential benefits of fisetin for the treatment of age-related neurodegenerative diseases.
Aging (Albany NY). 2017 Mar 8;9(3):955-963. doi: 10.18632/aging.101202.
New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463.
Zhu Y1, Doornebal EJ1,2, Pirtskhalava T1, Giorgadze N1, Wentworth M3, Fuhrmann-Stroissnigg H4, Niedernhofer LJ4, Robbins PD4, Tchkonia T1, Kirkland JL1.
Abstract
Senescent cells accumulate with aging and at sites of pathology in multiple chronic diseases. Senolytics are drugs that selectively promote apoptosis of senescent cells by temporarily disabling the pro-survival pathways that enable senescent cells to resist the pro-apoptotic, pro-inflammatory factors that they themselves secrete. Reducing senescent cell burden by genetic approaches or by administering senolytics delays or alleviates multiple age- and disease-related adverse phenotypes in preclinical models. Reported senolytics include dasatinib, quercetin, navitoclax (ABT263), and piperlongumine. Here we report that fisetin, a naturally-occurring flavone with low toxicity, and A1331852 and A1155463, selective BCL-XL inhibitors that may have less hematological toxicity than the less specific BCL-2 family inhibitor navitoclax, are senolytic. Fisetin selectively induces apoptosis in senescent but not proliferating human umbilical vein endothelial cells (HUVECs). It is not senolytic in senescent IMR90 cells, a human lung fibroblast strain, or primary human preadipocytes. A1331852 and A1155463 are senolytic in HUVECs and IMR90 cells, but not preadipocytes. These agents may be better candidates for eventual translation into clinical interventions than some existing senolytics, such as navitoclax, which is associated with hematological toxicity.
https://www.selfhacked.com/blog/15-science-based-health-benefits-fisetin-1-favorite/
15
Science-Based Fisetin Health Benefits + Natural Sources
Health Benefits of Fisetin
1) Fisetin is Good For Your Brain.. Encourages New Brain Growth... Fisetin Improves Memory... Protects Against Brain Degeneration... Decreases Brain Damage After Stroke... Fisetin Minimizes Brain Damage From Injury... Fisetin is Neuroprotective
2) Fisetin May Treat Depression
3) Fisetin Has Anti-Inflammatory Properties
4) Fisetin May Prevent and Treat Cancer
5) Fisetin Improves Blood Flow & Lowers Blood Pressure
6) Fisetin May Help Treat Diabetes
7) Fisetin May Extend Lifespan
8) Fisetin May Lower Body Weight
9) Fisetin Lowers Pain
10) Fisetin Protects Bone
11) Fisetin Protects Skin From Sun Damage
12) Fisetin Prevents Toxicity
13) Fisetin Helps Maintain Energy Levels
14) Fisetin Can Treat Infections
15) Fisetin is a Mast Cell Inhibitor and Can Help Histamine Intolerance
http://www.naturalblaze.com/2016/02/powerful-nutrient-in-cucumbers-prevents-memory-loss.html
February 16, 2016
Powerful
Nutrient in Cucumbers Prevents Memory Loss
by
Dena Schmidt
by
Dena Schmidt
The results of a recent study are offering new hope that avoiding memory loss related to aging as well as Alzheimer’s disease could be as simple as eating more cucumbers.
Many older adults resign themselves to memory loss as part of the aging process. However, a study out of the the Salk Institute for Biological Studies has shown that this doesn’t have to be the case. The health benefits of cucumbers are many, and one of them seems to be better memory and even the prevention of Alzheimer’s disease.
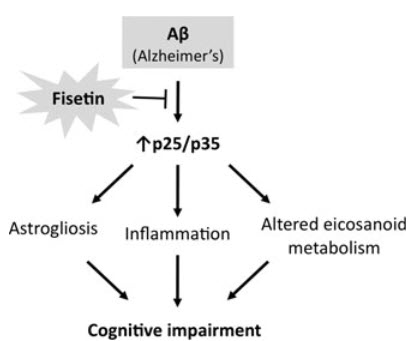
Researchers working with mice that normally developed the symptoms of Alzheimer’s (including memory loss) discovered that a daily dose of a flavonol called fisetin prevented these and other related impairments. This improvement occurred despite the continued formation of amyloid plaques, the brain proteins commonly blamed for Alzheimer’s.
The compound fisetin is found in numerous vegetables and fruits but is especially concentrated in strawberries and cucumbers. This flavonol is quite effective in stopping memory loss in mice and holds hope for humans as well...
Fisetin works by switching on a cellular pathway associated with the process of retrieving memories in the brain. Over a decade ago, other researchers discovered the compound fisetin assists in protecting the neurons of the brain from aging and its associated effects. It was found that this potent compound has both anti-inflammatory and antioxidant effects on brain cells..
http://www.salk.edu/news-release/natural-plant-compound-prevents-alzheimers-disease-in-mice/
January 27, 2014
Natural
plant compound prevents Alzheimer's disease in mice
A daily dose of the antioxidant fisetin keeps mice—even those with genetic mutations linked to Alzheimer's—from experiencing memory and learning deficits as they age.
A chemical that’s found in fruits and vegetables from strawberries to cucumbers appears to stop memory loss that accompanies Alzheimer’s disease in mice, scientists at the Salk Institute for Biological Studies have discovered. In experiments on mice that normally develop Alzheimer’s symptoms less than a year after birth, a daily dose of the compound—a flavonol called fisetin—prevented the progressive memory and learning impairments. The drug, however, did not alter the formation of amyloid plaques in the brain, accumulations of proteins which are commonly blamed for Alzheimer’s disease. The new finding suggests a way to treat Alzheimer’s symptoms independently of targeting amyloid plaques.
“We had already shown that in normal animals, fisetin can improve memory,” says Pamela Maher, a senior staff scientist in Salk’s Cellular Neurobiology Laboratory who led the new study. “What we showed here is that it also can have an effect on animals prone to Alzheimer’s.”
More than a decade ago, Maher discovered that fisetin helps protect neurons in the brain from the effects of aging. She and her colleagues have since—in both isolated cell cultures and mouse studies—probed how the compound has both antioxidant and anti-inflammatory effects on cells in the brain. Most recently, they found that fisetin turns on a cellular pathway known to be involved in memory.
“What we realized is that fisetin has a number of properties that we thought might be beneficial when it comes to Alzheimer’s,” says Maher.
So Maher — who works with Dave Schubert, the head of the Cellular Neurobiology Lab — turned to a strain of mice that have mutations in two genes linked to Alzheimer’s disease. The researchers took a subset of these mice and, when they were only three months old, began adding fisetin to their food. As the mice aged, the researchers tested their memory and learning skills with water mazes. By nine months of age, mice that hadn’t received fisetin began performing more poorly in the mazes. Mice that had gotten a daily dose of the compound, however, performed as well as normal mice, at both nine months and a year old.
“Even as the disease would have been progressing, the fisetin was able to continue preventing symptoms,” Maher says.
In collaboration with scientists at the University of California, San Diego, Maher’s team next tested the levels of different molecules in the brains of mice that had received doses of fisetin and those that hadn’t. In mice with Alzheimer’s symptoms, they found, pathways involved in cellular inflammation were turned on. In the animals that had taken fisetin, those pathways were dampened and anti-inflammatory molecules were present instead. One protein in particular—known as p35—was blocked from being cleaved into a shorter version when fisetin was taken. The shortened version of p35 is known to turn on and off many other molecular pathways. The results were published December 17, 2013, in the journal Aging Cell.
Studies on isolated tissue had hinted that fisetin might also decrease the number of amyloid plaques in Alzheimer’s affected brains. However, that observation didn’t hold up in the mice studies. “Fisetin didn’t affect the plaques,” says Maher. “It seems to act on other pathways that haven’t been seriously investigated in the past as therapeutic targets.”
Next, Maher’s team hopes to understand more of the molecular details on how fisetin affects memory, including whether there are targets other than p35.
“It may be that compounds like this that have more than one target are most effective at treating Alzheimer’s disease,” says Maher, “because it’s a complex disease where there are a lot of things going wrong.”
They also aim to develop new studies to look at how the timing of fisetin doses affect its influence on Alzheimer’s.
“The model that we used here was a preventive model,” explains Maher. “We started the mice on the drugs before they had any memory loss. But obviously human patients don’t go to the doctor until they are already having memory problems.” So the next step in moving the discovery toward the clinic, she says, is to test whether fisetin can reverse declines in memory once they have already appeared.
Other researchers on the paper were Antonio Currais, Marguerite Prior, Richard Dargusch, Jennifer Ehren, and David Schubert of the Salk Institute and Aaron Armando and Oswald Quehenberger of the University of California at San Diego...
MEDICAL
FOOD FOR COGNITIVE DECLINE.
US9254280
Nutraceutical
formulations that improve cognitive function in adults diagnosed
with neurodegenerative disease are provided, which comprise a
combination of fisetin and DHA.US9254280
BACKGROUND OF THE INVENTION
Alzheimer's disease (AD) is a degenerative disorder of the brain. It is the leading cause of dementia in elderly persons. AD patients have increased problems with memory loss and intellectual functions which progress to the point where they cannot function as normal individuals. With the loss of intellectual skills the patients exhibit personality changes, socially inappropriate actions and schizophrenia. AD is devastating for both victims and their families, for currently there is no effective palliative or preventive treatment for the inevitable neurodegeneration.
At a macroscopic level, the brains of AD patients are usually smaller, sometimes weighing less than 1,000 grams. At a microscopic level, the histopathological hallmarks of AD include neurofibrillary tangles (NFT), neuritic plaques, and degeneration of neurons. AD patients exhibit degeneration of nerve cells in the frontal and temporal cortex of the cerebral cortex, pyramidal neurons of the hippocampus, neurons in the medial, medial central, and cortical nuclei of the amygdala, noradrenergic neurons in the locus coeruleus, and the neurons in the basal forebrain cholinergic system. Loss of neurons in the cholinergic system leads to a consistent deficit in cholinergic presynaptic markers in AD.
The microtubule-associated protein known as Tau has been implicated in Alzheimers disease etiology. Tau binds to microtubules and assists with their formation and stabilization. However when tau is hyperphosphorylated, it is unable to bind and the microtubules become unstable and begin disintegrating. The unbound tau clumps together in formations called neurofibrillary tangles. More explicitly, intracellular lesions known as pretangles develop when tau is phosphorylated excessively and on improper amino acid residues. These lesions, over time, develop into filamentous neurofibrilary tangles (NFTs) which interfere with numerous intracellular functions.
Three different maturation states of NFT have been defined using anti-tau and anti-ubiquitin immunostaining. At stage 0 there are morphologically normal pyramidal cells showing diffuse or fine granular cytoplasmic staining with anti-tau. At stage 1 some delicate elongate inclusions are stained by tau antibodies; stage 2 is represented by the classic NFT demonstration with anti-tau staining; stage 3 is exemplified by ghost tangles where the host neuron has died, which are characterized by a reduced anti-tau but marked anti-ubiquitin immunostaining. It has been shown that the degree of cognitive impairment in diseases such as AD significantly correlates with the presence of neurofibrillary tangles.
The development of agents that can decrease cognitive decline, which may be a tauopathy, is of great interest for clinical and research purposes.
PUBLICATIONS
Cole and Frautschy, The Journal of Nutrition, first published as doi: 10.3945/jn.109.113910. Green et al. (2007) J. Neurosci. 27 (16) 4385-95; Maher (2010) In Micronutrients and Brain Health, L. Parker, H. Sies, M. Eggersdorfer, and E. Cardenas, eds. (Boca Raton, Fla., CRC Press), pp. 189-206, Genes Nutr. 2009 Sep. 10; Akaishi et al. (2008) Neurosci Lett. 444(3):280-5; Maher (2008) Arch Biochem Biophys. 476(2):139-44; Maher et al. (2006) P.N.A.S. 103(44):16568-73; Maher (2006) Free Radic Res. 40(10):1105-11.
SUMMARY OF THE INVENTION
The present invention is based, at least in part, on the discovery of novel nutraceutical formulations that improve cognitive function in adults diagnosed with neurodegenerative disease. Diseases of interest include those associated with beta amyloid and tau/tangle pathology, which may include cognitive decline with aging or Alzheimer's disease, frontal temporal dementia, Pick's, and progressive supranuclear palsy. The compositions of the invention provide a combined dose of docosahexaenoic acid (DHA) and fisetin effective in slowing or treating cognitive decline.
The novel formulations include a combination of fisetin and DHA, which unexpectedly behaves synergistically. Accordingly, the formulations of the invention provide a novel therapeutic approach for improving cognition, for example in aging adults and in adults diagnosed with neurodegenerative disease. The invention features, in a first aspect, methods of improving cognitive performance in a subject, comprising administering to the subject a nutraceutical formulation or composition of the invention. In an exemplary embodiment, the subject has been diagnosed with Alzheimer's Disease.
Other features and advantages of the invention will be apparent from the following detailed description, and from the claims.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1. Morris Water Maze results.
FIG. 2. Retention in the probe test.
FIG. 3. Western analysis of proteins.
FIG. 4. Fisetin and DHA in N9 microglia cells.
DEFINITIONS
The compositions of the present invention can be formulated according to known methods to prepare pharmaceutically and nutraceutically useful compositions, whereby these materials, or their functional derivatives, are combined in admixture with a pharmaceutically or nutraceutically acceptable carrier vehicle. Suitable pharmaceutical vehicles and their formulation, inclusive of other human proteins, e.g., human serum albumin, are described, for example, in Remington's Pharmaceutical Sciences (16.sup.th ed., Osol, A. ed., Mack Easton Pa. (1980)). In order to form a pharmaceutically acceptable composition suitable for effective administration, such compositions will contain an effective amount of the above-described compounds together with a suitable amount of carrier vehicle.
An effective amount varies depending upon the health and physical condition of the subject to be treated, the taxonomic group of subjects to be treated (e.g. human, nonhuman primate, etc.), the capacity of the subject's nervous system, the degree of protection desired, the treating doctor's assessment of the medical situation, the condition to be treated or prevented, and other relevant factors.
Before the present compositions and methods are described in further detail, it is to be understood that this invention is not limited to particular methods described, as such may, of course, vary. It is also to be understood that unless stated otherwise, the terminology used herein is for the purpose of describing particular embodiments only, and is not intended to be limiting.
Where a range of values is provided, it is understood that each intervening value, to the tenth of the unit of the lower limit unless the context clearly dictates otherwise, between the upper and lower limit of that range and any other stated or intervening value in that stated range, is encompassed within the invention. The upper and lower limits of these smaller ranges may independently be included in the smaller ranges, subject to any specifically excluded limit in the stated range.
The publications discussed herein are provided solely for their disclosure prior to the filing date of the present application. Nothing herein is to be construed as an admission that the present invention is not entitled to antedate such publication by virtue of prior invention. Further, the dates of publication provided may be different from the actual publication dates, which may need to be independently confirmed.
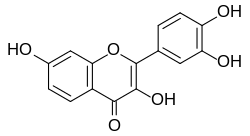
Fisetin (2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4-one) is a yellow flavonoid polyphenol present at very low quantities in many foods, notably strawberries. Like resveratrol, it has been reported as a sirtuin-activating compound. Like many other polyphenols, it has pleiotropic antioxidant and anti-inflammatory activities (see, for example, Geraets et al. (2009) Biochemical and Biophysical Research Communications 382 (3): 598-603).
DHA (Docosahexaenoic acid; all-cis-docosa-4,7,10,13,16,19-hexa-enoic acid) is an omega-3 fatty acid that is a primary structural component of the human cerebral cortex, sperm, testicles and retina. It can be synthesized from alpha-linolenic acid or obtained directly from fish oil.
Cold-water oceanic fish oils are rich in DHA. Most of the DHA in fish and multi-cellular organisms with access to cold-water oceanic foods originates from photosynthetic and heterotrophic microalgae, and becomes increasingly concentrated in organisms the further they are up the food chain. DHA is also commercially manufactured from microalgae; Crypthecodinium cohnii and another of the genus Schizochytrium. In humans, DHA is either obtained from the diet or synthesized from eicosapentaenoic acid (EPA, 20:5, ω-3) via docosapentaenoic acid (DPA, 22:5 ω-3) as an intermediate.
DHA is the most abundant omega-3 fatty acid in the brain and retina. DHA comprises 40% of the polyunsaturated fatty acids in the brain and 60% of the PUFAs in the retina. Fifty percent of the weight of a neuron's plasma membrane is composed of DHA. DHA modulates the carrier-mediated transport of choline, glycine, and taurine, the function of delayed rectifier potassium channels, and the response of rhodopsin contained in the synaptic vesicles, among many other functions. DHA deficiency is associated with cognitive decline (see Lukiw et al. (2005) J Clin Invest. 115 (10): 2774-83).
Preliminary studies indicated that DHA can slow the progression of Alzhiemer's disease in mice. However, the first large-scale human trials showed that DHA alone did not slow decline of mental function in elderly people with mild to moderate Alzheimer's disease (Quinn et al. (2010) JAMA 304 (17): 1903-11). These trials were part of a large U.S. National Institutes of Health (NIH) intervention study to evaluate DHA in Alzheimer's disease.
Detailed Description Of The Embodiments
The present invention is based, at least in part, on the discovery of novel nutraceutical formulations which improve cognitive function, e.g. in adults diagnosed with neurodegenerative disease, such as early-stage and mid/late-stage Alzheimer's Disease. The development of non-prescription nutraceutical formulations is highly desirable as both a preventative measure, as well as to augment any pharmacological treatment approaches. Such nutraceutical formulations also are useful for normal subjects, e.g., normal adults seeking to improve cognitive function.
The nutraceutical formulations described herein contain components that synergistically provide neuroprotection against undesirable inflammation and development of filamentous neurofibrilary tangles (NFTs) which interfere with numerous intracellular functions. Co-administration of the components of the nutraceutical formulations maintained and/or improved cognitive performance over an extended period of time in the animal model 3×AD transgenic mice, indicating that the combination of components is useful in treating and/or reducing the symptoms associated with AD and other taouopathies.
The formulations can be used or administered alone, or together in combination with other nutraceutical or pharmaceutical compositions. Nutraceutical or pharmaceutical compositions suitable for administration in combination with the formulations of the invention include nutraceutical or pharmaceutical compositions effective in improving cognition or reducing symptoms associated with a neurological disorder (e.g., Alzheimer's disease and other taouopathies). The compositions of the invention may be administered to a mammal, e.g. a mouse, including mouse models for neurologic disease, a human, etc.
In some preferred embodiments, the formulation is administered orally. In an alternative embodiment, the formulation is administered parenterally. In a further embodiment of these aspects, the formulation is administered as a unit dosage form. A combination product of the invention can be defined based on the weights of the two agents per dosage unit. On a weight basis, the ratio of fisetin to DHA is usually from about 20:1 to about 1:20; from about 10:1 to about 1:10; from about 5:1 to about 1:5; from about 2:1 to about 1:2; and in some embodiments is about 1:6, about 1:10, about 1:15 or about 1:20.
In an exemplary embodiment, the unit dose of the formulation comprises DHA in an amount of at least about 25 mg, at least about 100 mg, at least about 500 mg, at least about 1 g, at least 2 g, at least about 5 g, or at least about 10 g. The unit dose of the formulation may comprise fisetin in an amount of at least about 10 mg, at least about 50 mg, at least about 100 mg, at least about 250 mg, at least about 500 mg, at least about 1 g, at least about 2 g, at least about 5 g, or at least about 10 g. Nutraceutical formulations can be prepared using methods known in the art. Optionally the nutraceutical formulation further comprises one or more anti-oxidants in addition to the fisetin and DHA, e.g. ascorbic acid, coenzyme Q, etc.
In an exemplary embodiment, the unit dose of the formulation comprises DHA in an amount of at least about 25 mg, at least about 100 mg, at least about 500 mg, at least about 1 g, at least 2 g, at least about 5 g, or at least about 10 g. The unit dose of the formulation may comprise fisetin in an amount of at least about 10 mg, at least about 50 mg, at least about 100 mg, at least about 250 mg, at least about 500 mg, at least about 1 g, at least about 2 g, at least about 5 g, or at least about 10 g. Nutraceutical formulations can be prepared using methods known in the art. Additional information regarding nutraceuticals can be found at the Natural Products Association.
Table 1 sets forth certain preferred embodiments of the instant nutraceutical composition with respect to the amounts of DHA and fisetin present in the composition. Such composition, which preferably is for human oral consumption, is envisioned for daily consumption to treat an existing cognitive disorder (e.g., AD) or improve cognition in a “normal” subject (i.e., a subject who has an as yet undiagnosed cognitive disorder or a subject (preferably over the age of 60) who has no cognitive disorder). Additional delivery regimens for these compositions include twice per day, once every two days, once every three days, and once per week.
TABLE 1
Composition # Fisetin (mg) DHA (mg)
The nutraceutical formulations for use in accordance with the present invention can be formulated in a conventional manner using one or more physiologically acceptable carriers or excipients. Agents used in the formulations and their physiologically acceptable salts and solvates can be prepared for administration by various methods. In an exemplary embodiment, administration of the formulations is oral (ideally solid or liquid). In an alternative embodiment, administration is parenteral, e.g., intravenous, subcutaneous, intramuscular, intracranial, intraorbital, ophthalmic, intraventricular, intracapsular, intraspinal, intracistemal, intraperitoneal, or transmucosal. The compositions can be formulated in various ways, according to the route of administration.
For oral administration, the formulations can take the form of, for example, tablets or capsules, prepared by conventional means with pharmaceutically acceptable excipients such as binding agents (for example, pregelatinised maize starch, polyvinylpyrrolidone or hydroxypropyl methylcellulose); fillers (for example, lactose, microcrystalline cellulose or calcium hydrogen phosphate); lubricants (for example, magnesium stearate, talc or silica); disintegrants (for example, potato starch or sodium starch glycolate); or wetting agents (for example, sodium lauryl sulphate). The tablets can be coated by methods well known in the art. Also included are bars and other chewable formulations.
Liquid preparations for oral administration can take the form of, for example, solutions, syrups or suspensions, or they can be presented as a dry product for constitution with water or other suitable vehicle before use. In one embodiment, the liquid preparations can be formulated for administration with fruit juice, e.g., apple juice. Such liquid preparations can be prepared by conventional means with pharmaceutically acceptable additives such as suspending agents (for example, sorbitol syrup, cellulose derivatives or hydrogenated edible fats); emulsifying agents (for example, lecithin or acacia); non-aqueous vehicles (for example, almond oil, oily esters, ethyl alcohol or fractionated vegetable oils); and preservatives (for example, methyl or propyl-p-hydroxybenzoates or sorbic acid). Other suitable non-aqueous vehicles may include neuroprotective foods, e.g., fish oil, flax seed oil, etc. The preparations can also contain buffer salts, flavoring, coloring and sweetening agents as appropriate.
Preparations for oral administration may be provided as a unit dosage form, for example, as tablets, capsules, etc. These can be presented in blister packs or in multi-dose containers. Preparations for oral administration can also be suitably formulated to give controlled release of the active compound.
For buccal or sublingual administration the formulations can take the form of tablets or lozenges formulated in conventional manner. The formulations can be prepared for parenteral administration by injection, for example, by bolus injection or continuous infusion. Formulations for injection can be presented in unit dosage form, for example, in ampoules or in multi-dose containers, with an added preservative. The formulations can take such forms as suspensions, solutions or emulsions in oily or aqueous vehicles, and can contain formulatory agents such as suspending, stabilizing and/or dispersing agents. Alternatively, the active ingredients can be in powder form for constitution with a suitable vehicle, for example, sterile pyrogen-free water, before use.
The formulations can also be prepared in rectal compositions such as suppositories or retention enemas, for example, containing conventional suppository bases such as cocoa butter or other glycerides.
The formulations can also be provided as a depot preparation. Such long acting formulations may be administered by implantation (for example subcutaneously or intramuscularly) or by intramuscular injection. Thus, for example, the formulations can be prepared with suitable polymeric or hydrophobic materials (for example as an emulsion in an acceptable oil) or ion exchange resins, or as sparingly soluble derivatives, for example, as a sparingly soluble salt.
The formulations can be presented in a pack or dispenser device, which may contain one or more unit dosage forms containing the active ingredient. The pack may, for example, comprise metal or plastic foil, such as a blister pack. The pack or dispenser device can be accompanied by instructions for administration.
The therapeutic formulations of the invention can also contain a carrier or excipient, many of which are known to skilled artisans. Excipients that can be used include buffers (for example, citrate buffer, phosphate buffer, acetate buffer, and bicarbonate buffer), amino acids, urea, alcohols, ascorbic acid, phospholipids, proteins (for example, serum albumin), EDTA, sodium chloride, liposomes, mannitol, sorbitol, and glycerol.
Methods useful for making formulations are known in the art and can be found in, for example, Remington's Pharmaceutical Sciences (Gennaro, ed., Williams & Wilkins, Baltimore, Md.).
The invention additionally features methods of treating a neurodegenerative disorder in a subject, involving administering a therapeutically effective amount of a composition of the invention to the subject, such that the disorder is treated. The invention further features methods of reducing symptoms associated with a neurodegenerative disorder in a subject, involving administering a therapeutically effective amount of a composition of the invention to the subject, such that the symptoms of the disorder are reduced.
In general, the instant composition may act, for example, by (i) eliminating a cognitive disorder, (ii) ameliorating one or more symptoms of a cognitive disorder, (iii) slowing the progression of a cognitive disorder, (iv) delaying the onset of a cognitive disorder's symptoms, and (v) reducing the likelihood of a cognitive disorder's onset.
Importantly, the combination of components of the formulations has been discovered to be more effective than the individual components in the uses of the invention. As the results described herein indicate, the degree of efficacy of the particular formulations of the invention was completely unanticipated, indicating that these unique combinations synergistically provide neuroprotection. Although convenient for administration, it is not necessary for the agents or components of the nutraceutical formulations to be compounded together for administration to a subject. Instead, they can be administered concurrently, or in close enough succession so that the desired dosage level for all components is achieved in the bloodstream at the same time.
In general, a nutraceutical formulation is formulated by combining appropriate concentrations of stock agent (e.g., in solution or solid) of the components in a medium. The components can be administered together, in rapid succession, or at intervals. A composition may be tested to determine whether it is an effective nutraceutical formulation in an in vitro cell culture system of primary, secondary, or immortalized neural cells, for example, cells that exhibit the molecular and biochemical characteristics of normal neural cells, or cells that exhibit at least some of the molecular and biochemical characteristics of a neurologic disorder. Such cells and methods of evaluating the effects of the formulations are known in the art, and exemplary cells and methods are described in the Examples. Biochemical and physical criteria can be used to measure the ability of a nutraceutical formulation to ameliorate adverse events associated with aging and inflammation. Biochemical and physical criteria can additionally be used to measure the ability of a nutraceutical formulation to ameliorate adverse effects associated with a disorder in these cell culture systems.
Animal models are likewise useful for evaluating the efficacy of a nutraceutical formulation. Nutraceutical formulations can be evaluated in vivo using an animal model, for example, an animal model for AD. Examples of animal models for AD are mice expressing the human E4 allele of ApoE, mice expressing the human form of the protein Tau, and other transgenic mouse lines in which expression of the amyloid precursor protein (APP) gene is affected (Reaume et al., J. Biol. Chem., 271:23380-23388, 1996; Hsiao et al., Science, 276:99-102, 1996; and Games et al., Nature, 373:523-527, 1995). Nutraceutical formulations can also be evaluated for their efficacy in preventing or ameliorating the effects of AD using an animal model. The effects of a nutraceutical formulation on ameliorating behavioral and biochemical symptoms in such mice are evaluated after administering a nutraceutical formulation to these mice both early in life and after symptoms begin to develop. Mice are evaluated for the development or progression of learning and memory disorders as well as for pathophysiologic and biochemical abnormalities such as the presence of plaques in cortical and limbic structures of the brain. Prevention of the onset or progression of symptoms, or the amelioration of existing symptoms, indicates that the nutraceutical formulation is effective for treating Alzheimer disease in humans.
While is not necessary for all of the components of a nutraceutical formulation to be administered in the same excipient, in the same form, or delivered at precisely the same time during a day, the components should be administered so they are present in the treated subject at the same time (e.g., present in a cell that is the target of treatment), and thus, one formulation, including both components, is generally provided in a convenient dosage form.
The nutraceutical formulations of the invention may be components in kits. These kits can also include instructions for administration of the formulations to a subject, and optionally may include one or more other nutraceuticals, e.g., ginko biloba, fish oil, apple juice, flax seed oil, and other nutraceutical foods or formulations known in the art. These kits may additionally include instructions for administration, and/or other nutraceutical foods or formulations.
This invention is further illustrated by the following examples, which should not be construed as limiting. The contents of all references, patents and published patent applications cited throughout this application are incorporated herein by reference.
The invention has been described herein with reference to certain examples and embodiments only. No effort has been made to exhaustively describe all possible examples and embodiments of the invention. Indeed, those of skill in the art will appreciate that various additions, deletions, modifications and other changes may be made to the above-described examples and embodiments, without departing from the intended spirit and scope of the invention as recited in the following claims. It is intended that all such additions, deletions, modifications and other changes be included within the scope of the following claims.
EXAMPLES
The fish oil enriched omega 3 fatty acid DHA has been reported to be ineffective in treating mild to moderate Alzheimer disease patients who have extensive beta amyloid plaques and tangles. It has been shown that DHA can reduce the accumulation of beta amyloid but DHA appears to lose efficacy over time as pathology develops in an animal model with both beta amyloid and tangles (tau pathology), the 3×AD transgenic mouse. We found that fish oil alone was insufficient to treat the 3×AD Tg mouse.
Fisetin, a natural flavonoid enriched in strawberries, is neuroprotective and can improve cognitive function in various animal models including aging mice and beta amyloid plaque accumulating transgenic mice. We have tested 0.6% DHA alone, 500 ppm fisetin alone and DHA+fisetin in combination delivered as a food supplement (in chow) to older 3×AD Tg mice with combined amyloid and tau pathology. The study ran from ̃9 months to 13-14 months of age. Cognitive deficits were improved by DHA+fisetin but not by DHA alone or fisetin alone (FIG. 1).
As shown in FIG. 1, Morris Water Maze results show no improvement in time (latency) to find a hidden platform with additional blocks of training when 12-13 month old 3×AD Tg mice are tested on control bad (high safflower oil) diet with or without DHA (green) or fisetin (light blue) but marked improvement over time with DHA+fisetin together.
FIG. 2. Shows that the dietary combination of fisetin and DHA improved retention in the probe test. In this test the platform is removed, and the mice are allowed to swim for 60 seconds, while the time in the target quadrant is quantified. Fisetin alone also improved retention, but the combination resulted in the best performance on this test.
FIG. 3. Western analysis of proteins from brain of the same 3×AD triple mice studied on the same diets as in FIG. 1 behavior studies. The excitatory synaptic marker PSD-95 is partially protected (increased) by DHA alone (trend) but not by fisetin but significantly protected by DHA+fisetin (p<0.001).
These data demonstrate that a treatment combining DHA and fisetin or fish oil and fisetin is a synergistic treatment for cognitive deficits. Since beta amyloid and tau/tangle pathology occur in normal aging and many years prior to Alzheimer diagnosis, DHA and fisetin can be effective in slowing cognitive decline with aging or Alzheimer's, or an effective treatment for these or other conditions with beta amyloid and or tauopathy related cognitive deficits including frontal temporal dementia, Pick's, progressive supranuclear palsy.
To demonstrate the synergistic activity of the two agents and to establish an appropriate fixed-dose ratio for clinical investigation, varying amounts of fisetin and DHA have been added to N9 microglia cells under simulated inflammatory conditions (i.e., 10 μg/ml LPS). After 24 hr NO release was measured in the culture medium as NO2 using the Griess assay. Results are presented as the % of NO2 in the culture medium of cells treated with LPS alone The combination of the two agents was found to have a strong synergistic effect on inflammation, particularly at the indicated enzyme ratios.
These data show that a combination of DHA and fisetin provide a synergistic improvement in anti-inflammatory activity. Effective ratios include without limitation those where fisetin is provided at a concentration of at least 5 μM, and where the ratio of DHA to fisetin may be at least about 1:2, 1:5, 1:10 or more.
Neuroprotective
polyphenol analogs
US2014186328
US2014186328
The present invention provides neuroprotective polyphenol compounds, which can be synthetic analogs of fisetin, baicalein or chlorogenic acid, that maintain neuroprotective, anti-inflammatory, glutathione promoting, and/or antioxidant properties. The neuroprotective polyphenol compounds are useful for promoting, enhancing and/or increasing neuron protection, growth and/or regeneration. The polyphenol compounds further find use for increasing and or maintaining intracellular glutathione (GSH) levels. The polyphenol compounds are also useful for treating, preventing, mitigating and/or delaying neurodegenerative conditions, including diabetes, Parkinson's disease, Huntington's disease, Alzheimer's disease, non-Alzherimer's dementias, multiple sclerosis, traumatic brain injury, spinal cord injury or ALS.
FIELD
[0003] The present invention provides compounds having neuroprotective, neurotrophic, anti-inflammatory and/or anti-oxidant properties. The compounds are useful for promoting, enhancing and/or increasing the protection, growth and/or regeneration of neurons. The compounds also find use to increase, enhance and/or maintain intracellular glutathione (GSH) levels. The invention further relates to methods for the treatment, prevention, and mitigation of neurodegenerative conditions, and methods for the treatment, prevention, and mitigation of diabetes and Huntington's disease, comprising administering to a subject in need thereof an effective amount of a compound as disclosed and claimed herein.
BACKGROUND
[0004] There are currently no drugs available that prevent the nerve cell death associated with the majority of age-related disorders of the CNS. There are a number of reasons for this but probably the most important is that multiple factors contribute to the nerve cell death such that targeting a single pathway is unlikely to be successful. One example of this problem is ischemic stroke which is the leading cause of adult disability and the third leading cause of death in the US (Véronique, et al., Circulation. (2011) 123 (4), e18-e209). Worldwide, approximately 5 million people die each year of stroke and the mortality rates are estimated to double by the year 2020 (Donnan, et al., The Lancet. (2008), 371 (9624), 1612-1623). The nerve cell death associated with cerebral ischemia is due to multiple factors resulting from the lack of oxygen to support respiration and ATP synthesis, acidosis due to the buildup of the glycolytic product lactic acid, the loss of neurotrophic support, multiple metabolic stresses and inflammation (Lipton, Physiol. Rev. (1999) 79, 1431-1568; and Pandya, et al., Cent. Nerv. Syst. Agents. Med. Chem. (2011) April 27, PMID:21521165). While the focus of current drug discovery paradigms is on the development of high affinity, single target ligands, it is unlikely that a drug directed against a single molecular target will be effective in treating the nerve cell death associated with conditions such as stroke because of the multitude of insults that contribute to the cell's demise. This conclusion is supported by the failure of the single, high affinity target approach to drug development to identify treatments for stroke. Indeed, the only FDA-approved treatment to date is recombinant tissue-type plasminogen activator (rt-PA) (Green, et al., Drug Discov. Today. (2006) 11, 681-693), which is a vascular agent. An alternative approach is to identify small molecules that have multiple biological activities relevant to the maintenance of neurological function.
[0005] The flavonal Fisetin has been found to be an orally active, novel neuroprotective and cognition-enhancing molecule (Maher, Genes. Nutr. (2009), September 10, PMID:19756810). Fisetin not only has direct antioxidant activity but it can also increase the intracellular levels of glutathione, the major intracellular antioxidant, via the activation of transcription factors such as Nrf25. Fisetin can also maintain mitochondrial function in the presence of oxidative stress. In addition, it has anti-inflammatory activity against immune cells and inhibits the activity of 5-lipoxygenase, thereby reducing the production of lipid peroxides and their pro-inflammatory by-products (Maher, Genes. Nutr. (2009), supra). This wide range of actions suggests that Fisetin has the ability to reduce the loss of neurological function associated with multiple disorders, including stroke.
[0006] Although Fisetin has been shown to be effective in the rabbit small clot embolism model of stroke (Maher, et al., Brain Research. (2007) 1173, 117-125), its relatively high EC50 in cell based assays (2-5 M) and also low lipophilicity (CLogP 1.24), high tPSA (107 Å), more hydrogen bond donors (HBD=5) and poor bioavailability (Shia, et al., J. Agric. Food Chem. (2009) 57 (1), 83-89) suggest that there is room for medicinal chemical improvement if Fisetin is to be used therapeutically for treating neurological disorders such as stroke. However, given its ability to activate multiple target pathways related to neuroprotection, screening for improvements is significantly more complicated than with the current classical approach to the development of a single target drug. The present invention is based in part, on the use of a multi-tiered approach to screening that has facilitated the identification of Fisetin derivatives with significantly enhanced neuroprotective activity in an in vitro ischemia model while at the same time maintaining other key actions including anti-inflammatory and neurotrophic activity as well as the ability to maintain glutathione under conditions of oxidative stress.
SUMMARY
[0007] In various embodiments, the invention is directed to polyphenol compounds and analogs that can be used in treatment of patients afflicted with medical conditions such as diabetes, Huntington's disease, Parkinson's disease, Alzheimer's dementia, non-Alzheimer's dementia, multiple sclerosis, traumatic brain injury, spinal cord injury, and ALS, as well as for treatment of conditions involving ischemia, such as ischemic or embolic stroke, and their symptoms and sequelae. The compounds of the invention can be used to maintain glutathione levels in patients, and can provide neuroprotective effects …
METHODS
OF USING FLAVONOIDS TO ENHANCE MEMORY
US7897637
US7897637
Described herein are flavonoids (e.g., 5-desoxy flavones and/or 5-desoxy flavonols, including without limitation fisetin and its derivatives) that activate ERK and induce CREB phosphorylation in neuronal cultures, facilitate long-term potentiation in hippocampal slices and enhance object recognition in vivo. Methods of using these flavonoids, for instance, for enhancing memory are described.
FIELD
This disclosure concerns the use of 5-desoxy-flavones and 5-desoxy-flavonols, such as fisetin and/or its derivatives, to enhance memory in vivo and/or to promote nerve cell differentiation and/or protect nerve cells from oxidative damage.
BACKGROUND
Memory is the recollection of past experiences. Almost everyone forgets something occasionally and, typically, forgetfulness increases as a person grows older. Normal aging also may result in trouble learning new material or requiring longer time to recall learned material. Age-associated memory impairments are currently estimated to affect at least 16% of everyone over the age of 50 (Tully et al., Nature Rev. Drug Discov., 2:267-277, 2003). Mild memory loss, while a nuisance, does not usually affect a person's normal day-to-day functioning. Other forms of memory loss can be more severe and have a functional impact. Common causes of memory loss of various severities include, among others, aging, Alzheimer's disease, neurodegenerative illness, head trauma or injury, seizures, general anesthetics (such as halothane, isoflurane, and fentanyl), alcoholism, stroke or transient ischemic attack (TIA), transient global amnesia, drugs (such as barbiturates or benzodiazepines), electroconvulsive therapy (especially if prolonged), temporal lobe brain surgery, brain masses (caused by tumors or infection), herpes encephalitis or other brain infections, and/or depression.
Long term potentiation (LTP) is considered to be the cellular basis of learning and memory and is dependent on synaptic plasticity (Bliss and Collingridge, Nature, 361:31-39, 1993), which can be defined as the long-lasting strengthening of the connections between two nerve cells. Synaptic plasticity in turn is believed to be dependent on a complex interplay of protein kinases, phosphatases, and transcription factors that ultimately give rise to long-term changes in the connections between nerve cells (Gaiarsa et al., Trends Neurosci. 25:564-570, 2002). Put more simply, a particular experience is registered in the brain as a circuit-specific pattern of neural activity and, due to synaptic plasticity, the structure of the circuit is modified so as to form a memory.
LTP was originally discovered in the hippocampus but has since been observed in other regions of the brain including the cerebral cortex, cerebellum and amygdala (Malenka and Bear, Neuron, 44:5-21, 2004). One model of learning postulates that the hippocampus is the gateway to long-term memory and, once the hippocampus has registered a memory, the memory is propagated to relevant portions of the cortex for storage (e.g., visual memory to visual cortex, auditory memory to auditory cortex, etc.) (for reviews, see, Tully et al., Nature Rev. Drug Discov., 2:267-277, 2003; Adams and Sweatt, Annu. Rev. Pharmacol. Toxicol., 42:135-163, 2002).
Neurotrophic factors, which modulate short- and long-term changes in neurons of the central nervous system (CNS), have been suggested to play roles in neuronal plasticity such as learning and memory (Lo, Neuron, 15:979-981, 1995; Thoenen, Science, 270:593-598, 1995). Neurotrophic factors support the survival, differentiation and functional maintenance of nerve cells. Because of these properties, neurotrophic factors have the potential to treat a variety of chronic and acute disorders of the CNS, including memory loss. However, many classical neurotrophic factors, such as nerve growth factor, are not well suited for therapeutic purposes due to their large size and proteinaceous nature (Levy et al., BioDrugs, 19:97-127, 2005). Thus, the identification of small molecules that can mimic some or all of the properties of neurotrophic factors could have great potential for treating CNS disorders, such as memory deficits.
Twenty eight different flavonoids, including representatives of all of the six different flavonoid classes (e.g., flavanones, flavan-3-ols, flavonols, flavones, anthocyanidins and isoflavones; U.S. Department of Agriculture, USDA database for the flavonoid content of selected foods, Beltsville, Md.:U.S. Department of Agriculture; 2003), were previously assayed for their ability to promote neurite outgrowth in PC12 cells (Sagara et al., J. Neurochem., 90:1144-1155, 2004). Among the flavonoids tested, only four were found to promote PC12 cell differentiation and of these, fisetin (3,7,3′,4′,-tetrahydroxyflanone; a flavonol) was by far the most effective. Fisetin has an EC50 for differentiation of 5 μM and at 10 μM routinely induces the differentiation of 75-80% of the cells The other three flavonoids that induced differentiation of PC12 cells, Iuteolin (5,7,3′,4′-tetrahydroxyflavone; a flavone), quercetin (3,5,7,3′,4′-pentahydroxyflavone; a flavonol) and isorhamnetin (3′-methoxy-3,5,7,4′-tetrahydroxyflavone; a flavonol), had EC50s of 10 μM and at best induced the differentiation of only 50% of the cells.
The induction of differentiation by fisetin was dependent on the activation of the Ras-ERK cascade because inhibitors of this cascade blocked differentiation (Sagara et al., J. Neurochem., 90:1144-1155, 2004). In addition to promoting nerve cell differentiation fisetin has also been shown to protect nerve cells from oxidative stress-induced death (Ishige et al., Free Radic. Biol. Med., 30:433-446, 2001). However, the structural features of fisetin that underlie its functions of promoting nerve cell differentiation and/or protecting such cells from oxidative damage are unknown. Also unknown is whether the ability of fisetin to promote nerve cell differentiation and/or protect neural cells from oxidative damage have in vivo correlates.
A need exists for the identification of small molecules that can mimic some or all of the properties of neurotrophic factors. Such molecules have potential for treating CNS disorders, such as memory loss.
SUMMARY
Disclosed herein are fisetin derivatives that are surprisingly effective neurotrophic factors. These derivatives (including, for example, 3,3′,4′-trihydroxyflavone and 3′,4′-dihydroxyflavone) promote nerve cell differentiation and/or protect such cells from oxidative damage at significantly lower concentrations than does, for example, fisetin.
This disclosure also concerns the discovery that 5-desoxy-flavones and/or 5-desoxy-flavonols (such as fisetin and certain of its derivatives) improves memory in vivo. This important discovery enables, for instance, methods of enhancing memory in wide variety of subjects, including normal subjects or subjects suffering from memory loss.=
Excerpt :
http://www.livescience.com/51000-cucumber-nutrition.html
May 28, 2015
Cucumbers:
Health Benefits & Nutrition Facts
by
Jessie Szalay
by
Jessie Szalay
Health
benefits of cucumbers
Hydration
According to Ware, “Cucumbers are 95 percent water." This makes cucumbers a great way to stay hydrated, especially during the summer. A cup of cucumber slices is “nearly as thirst-quenching as a glass of water,” according to Eating Well magazine.
The anti-inflammatory compounds in cucumbers help remove waste from the body and reduce skin irritation, according to the Cleveland Clinic. Preliminary research also suggests cucumbers promote anti-wrinkling and anti-aging activity, according to an article in the journal Filoterapia.
Cancer prevention
Cucumbers contain two phytonutrient compounds associated with anti-cancer benefits: lignans and cucurbitacins. In recent years, pharmaceutical companies have been paying special attention to cucurbitacins, hoping to use them in new cancer drugs. According to a 2010 research review published in Scientific World Journal, scientists have found that cucurbitacins can help block the signaling pathways that are important for cancer cell proliferation and survival.
Cucurbitacins can also inhibits the growth of pancreatic cancer cells, according to a 2009 study published in the Journal of Cancer Research looked at cucurbitacin B (which cucumber contains) on human pancreatic cancer cells and found that cucurbitacin supplements inhibited the growth of seven pancreatic cancer cell lines by 50 percent, and also increased apoptosis, or "death by suicide," of pancreatic cancer cells.
According to World’s Healthiest Foods, lignans may protect against cancer through working with the bacteria in the digestive tract. The bacteria take the lignans and convert them into compounds such as enterodiol and enterolactone, which can bind onto estrogen receptors and possibly reduce the risk of estrogen-related cancers, such as ovarian, breast, endometrial and prostate cancers. The research is not yet clear on whether lignans actually assert anti-cancer benefits.
A 2009 meta-analysis published in the British Journal of Cancer found little or no association between lignan intake and reduced breast cancer risk. Similarly, most studies have not found significant correlations between lignan intake and reduced prostate cancer risk, according to the Linus Pauling Institute at Oregon State University, though one study of older Scottish men published in the British Journal of Nutrition showed that consuming an enterolactone-containing serum reduced the risk of prostate cancer.
On the other hand, a Journal of Nutrition study of nearly 800 American women found that those with those with the highest lignan intake had the lowest risk of ovarian cancer. Furthermore, a study published in the Journal of the National Cancer Institute looked at nearly 1,000 women in the San Francisco area and found that postmenopausal women with the highest lignan intakes had the lowest risk of endometrial cancer.
Skin
You’ve probably seen pictures of people at a spa relaxing with cucumber slices over their eyes. It turns out there’s science behind this pampering ritual. Ware explained, “Cucumbers have a cooling and soothing effect that decreases swelling, irritation and inflammation when used topically. Cucumber slices can be placed on the eyes can decrease morning puffiness or alleviate and treat sunburn when placed on the affected areas.” She also noted that high vegetable intake is associated with a healthy complexion in general.
Bone health
According to the University of Maryland Medical Center, in the past few decades, it has become clear that vitamin K is important to bone health, and one cup of cucumber contains about 19 percent of the recommended daily intake of vitamin K. One review published in Nutrition noted that vitamin K intake might reduce fracture rates, work with vitamin D to increase bone density and positively affect calcium balance.
The human body uses vitamin K when building bones, and the effects seem to be especially important for women. A large 2003 American Journal of Clinical Nutrition study showed that low vitamin K levels were associated with low bone density in women, but not in men. Another study published in the American Journal of Clinical Nutrition in 1999 found that low intakes of vitamin K were associated with an increased risk of hip fractures in middle-age women. This is especially interesting because the women saw results from eating lettuce, showing that dietary consumption of vitamin K via eating vegetables (not supplements) is beneficial. When it comes to men, the affects of vitamin K and bone health may become more apparent as they age: A 2000 study saw reduced risk of hip fracture among both elderly women and elderly men who consumed more vitamin K.
Antioxidants
Cucumbers contain several antioxidants, including vitamin C, beta-carotene and manganese, as well as flavonoids, triterpenes and lignans that have anti-inflammatory properties. Vitamin C is well known for its immune system benefits, and beta-carotene has been shown to be beneficial for vision, according to the Mayo Clinic.
According to a 2010 animal study published in the Journal of Young Pharmacists, fresh extracts from cucumber showed increased scavenging of free radicals. Free radicals are associated with a variety of human diseases, but can sometimes be held in check by antioxidants, according to the Pharmacognosy Review.
Another study of cucumber extracts in animals, published in the Archives of Dermatological Research, found increased overall antioxidant benefits. Though this study focused on the cosmetic applications of this use of cucumbers, decreased free radicals can improve your inside organs as well as your skin.
An additional study published in Current Pharmaceutical Design found a positive association between the triterpene cucurbitacin and reduced inflammation, particularly in cancer cells. A review of triterpenes on the immune system, published in the Journal of Ethnopharmacology, suggested that they can help with inflammation and encouraged future research.
Heart health
“Eating a variety of fruits and vegetables of all kinds is associated with a reduced risk for many health conditions, such as heart disease, diabetes, stroke and obesity,” said Ware. Cucumbers’ potassium content may be especially helpful in this regard. One cup of sliced cukes contains only about 4 percent of the body’s daily potassium needs, but it comes with significantly fewer calories than most high-potassium foods like bananas. Potassium is an essential part of heart health, according to the American Heart Association. Many studies have linked it with lower blood pressure because it promotes vasodiliation (widening of the blood vessels), according to Today’s Dietitian. A study of 12,000 adults, published in Archives of Internal Medicine, showed that those who consumed 4,069 mg of potassium each day lowered their risk of cardiovascular disease and ischemic heart disease by 37 percent and 49 percent, respectively, compared to those who took 1,793 mg per day.
The vitamin K in cucumbers is also known to be essential in the blood-clotting process, according to the Harvard School of Public Health.
Risks of eating cucumbers
There can be a few risks from eating cukes. Pesticide consumption is one concern. Ware explained, “The Environmental Working Group produces a list each year of fruits and vegetables with the highest levels of pesticide residue, known as the Dirty Dozen. Cucumbers are one of the fruits and vegetables that the Environmental Working Group has placed on its Dirty Dozen list, meaning the exposure to pesticide residue is high.”
Additionally, cucumbers may be waxed to help protect them during shipping. According to World’s Healthiest Foods, both organic and conventionally grown cukes may be waxed, but organic ones can only use non-synthetic waxes with chemicals approved under organic regulations. For this reason and the pesticide concerns, World’s Healthiest Foods encourages buying organic cucumbers. But Ware stipulated, “This does not mean you should avoid cucumbers altogether if you can’t find or afford organic. The nutritional benefit of eating conventionally grown produce outweighs the risk of not eating produce at all.”...
Patents
: Extraction of Fisetin
http://worldwide.espacenet.com/advancedSearch?locale=en_EP
CN104262310
Preparation method of fisetin capable of inhibiting tyrosinase activity through rapid separation in medium-pressure column
Preparation method of fisetin capable of inhibiting tyrosinase activity through rapid separation in medium-pressure column
Inventor(s): WANG CHENGZHANG, et al.
The invention relates to a preparation method of fisetin capable of inhibiting tyrosinase activity through rapid separation in a medium-pressure column, and belongs to the fields of development and applications of plant extract, natural drugs, and cosmetics. Fisetin with purity more than 95% can be obtained through the following steps: taking lacquer tree wood powder as the raw material, extracting the wood powder, and subjecting the extract to a middle-low pressure chromatogram and HPLC (high performance liquid chromatogram). The component obtained from lacquer tree can inhibit the tyrosinase activity efficiently, and the inhibition rate on the tyrosinase activity is greater than 95% when the concentration of the component is 150 [mu]g/mL or more. The preparation is simple and fast, and thus the provided method can be used to produce a natural tyrosinase inhibitor.
The present invention relates to a medium pressure column Separates preparation method of inhibiting the activity of tyrosinase paint flavin, belonging to the field of development and application of plant extracts, natural medicines and cosmetics. In sumac wood flour as raw material, extraction, extraction, chromatography and high performance liquid preparation obtained more than 95% of the low-pressure paint flavin. The present invention has obtained from sumac efficient inhibit tyrosinase activity ingredient in 150μg / mL concentration or more, of the activity of tyrosinase inhibition rate of> 95%. The preparation process is fast and simple, is a good source of development potential natural tyrosinase inhibitors.
TECHNICAL FIELD
The present invention relates to a medium pressure column Separates preparation method of inhibiting the activity of tyrosinase paint flavin, belonging to plant extracts, natural medicine and technology in the field of cosmetics.
Background technique
Sumac (Rhus verniciflua stokes), for the Anacardiaceae (Anacardiaceae) genus Rhus ((Toxicodendron) is a deciduous tree. China is the origin of sumac, sumac is an important special use forests, mainly located in Shaanxi, Sichuan, Chongqing, Hubei, Guizhou, Gansu and Yunnan seven provinces. Currently, the use of chemical sumac sumac and paint mainly in two aspects of seed, few studies on sumac wood actives. Sumac itself is an excellent medicinal wood, sumac leaves, flowers, roots, bark, fruit, dry paint and wooden heart can be used as medicine, cough, eliminate addiction, pass through, insecticide, swelling and other effects. Korea sumac wooden parts more active substances, a large number of studies have shown that flavonoids lacquer wooden parts have excellent oxidation resistance, anti-tumor, antibacterial and immunomodulatory effects.
Wood lacquer paint flavin is part of polyphenol compounds in the most typical active. Fisetin can effectively improve the retinal pigment epithelial cells from the damage caused by oxidative stress, can also inhibit the UV-induced cell death and generation of oxygen free radicals, thus preventing the further development of cataracts. Fisetin can be suppressed vascular endothelial growth factor (TNF-, 1L-6,1L-8, MCP-1) production and the expression of rheumatoid arthritis treatment. It can effectively inhibit prostate cancer cell proliferation and induce apoptosis and proliferation of breast cancer, cervical cancer, leukemia and other cells and promote apoptosis. According to existing reports, separated from the lacquer paint flavin mainly commonly used solvent extraction, silica gel column or column repeated separation, complex and lengthy process. Medium pressure column chromatography on a sample having a large, efficient, fast, etc., is a new type of separation and purification technology, widely used in natural products, food, biological medicine.
Tyrosinase [EC 1.14.18.1, Tyrosinase] is a dual-core copper ion oxidoreductase, a polyphenol oxidase. Widely distributed in the living body, catalysis key rate-limiting enzyme in melanin synthesis, is closely related to the organism of important physiological processes. There are many biologically active substances, plant polyphenols nature natural plant is the main source of natural tyrosinase inhibitors. Although currently active in the sumac Flavonoids evaluation done a lot, but not to develop natural tyrosinase inhibitor is the main purpose.
SUMMARY
To achieve the above object, the inventors of the medium pressure column Separates preparation method of inhibiting the activity of tyrosinase paint flavin, characterized by the following steps:
The first step, skim:
Sumac wood powder drying, crushing, and wood flour using petroleum ether or hexane by mass volume ratio 1: 15-35 g / mL was degreased, temperature 30 ° C-80 ° C, time of 1-3 h, extraction times 1- 4, the filter residue was filtered, dried, defatted wood powder lacquer;
The second step, to extract:
The degreased wood flour and sumac 30% to 100% methanol aqueous solution or ethanol aqueous solution ratio by mass Volume 1: 15-30g / mL extracted 1-4 times, extraction temperature 60-80 ° C, the extraction time of 1-3 h, extracted , filtered and the combined filtrate concentrated in vacuo to give a crude extract of sumac;
The third step is extraction:
The crude extract with ethyl acetate sumac mass volume ratio 1: 15-30 (g / mL) was extracted 2-5 times the combined ethyl acetate phase was concentrated in vacuo to give ethyl acetate extract;
The fourth step, the pressure column purification:
The ethyl acetate extract with medium pressure column packing mass ratio of 1: 15-50 adsorption medium pressure column column length 20-300 cm, column diameter of 2-30 cm, with different proportions of eluent elution column pressure of 3-20 MPa, detection wavelength 280-360 nm, flow rate of 2-200 mL / min, fisetin enriched portion, concentrated in vacuo, solvent recovery, the concentrate is fisetin extract analyzed by HPLC, yellow paint Su content of more than 70%;
The fifth step, HPLC preparative separation:
More than 70% of the paint Flavin extract HPLC to give the paint Flavin, by HPLC analysis, paint flavin content of more than 95%; this method uses an alcohol solution extracted through extraction, separation medium pressure column fast get more than 70% of the paint flavin, then further 95% or more paint flavin by preparative HPLC separation;
The sixth step, paint flavin inhibit tyrosinase activity assessment:
Fisetin preparative HPLC was subjected to inhibit tyrosinase activity evaluation, testing dopa generation amount of pigment by UV chromatography, to determine when the paint Flavin methanol solution concentration range of 100 ~ 200ug / mL, the inhibition rate of 60 to 100% , an IC 50 value 69.46μg / mL.
The patent-pressure column packing optional 200-500 mesh silica and alumina in one or two in any ratio, eluent petroleum ether, ethyl ether, n-hexane, ethyl acetate and chloroform in one or more the mixed solution also choose a pore diameter of 60 a, 40-60 μm of ODS C18, C8 and materials Sephedex LH-20, eluent mixture of one or several of the solution of methanol, ethanol and water.
This patent HPLC analysis conditions: RP-18 endcapped column (4.6 × 250mm, 5μm), the mobile phase was methanol-0.5% by volume aqueous acetic acid 35:65, UV detector, detection wavelength of 280 and 360nm, flow rate 1mL / min. HPLC conditions for preparation: RP-18 endcapped column (4.6 × 250mm, 5μm), the mobile phase was methanol-0.5% by volume aqueous acetic acid 35:65, UV detector, detection wavelength of 280 and 360nm, flow rate of 1mL / min.
The beneficial effects of the invention:
The present invention provides a medium pressure column Separates preparation method of inhibiting the activity of tyrosinase paint flavin, but also has the following characteristics:
(1) of the present invention, medium pressure column chromatography can be prepared quickly and efficiently prepared paint flavin compound high levels;
(2) by preparative chromatography can be obtained high purity paint Flavin;
(3) paint flavin monomer having a low concentration inhibiting tyrosine activity, the human body without toxic effects;
(4) operation of the present invention, the process is simple, easy to implement, high purity, high activity;
(5) lacquer paint in yellow on tyrosinase has significant inhibitory effect, can be used for whitening cosmetics, due to the prevention and treatment of human melanoma caused by excessive pigmentation diseases.
BRIEF DESCRIPTION
Figure 1 is a HPLC chromatogram after medium pressure column rapid separation of compounds;
Figure 2 is a preparative liquid monomer after 3D-DAD-HPLC (190-800 nm) chromatogram;
Figure 3 is the IR spectrum of the monomer;
Figure 4 is a MS spectrum of the monomer;
Figure 5 is a 1H-NMR of a monomer spectrum;
CN104072465
Process for extracting fisetin from cotinus coggygria
Process for extracting fisetin from cotinus coggygria
Inventor(s): LIU DONGFENG, et al.
The invention relates to a process for extracting fisetin from cotinus coggygria. The process comprises the following steps: (1) crushing anacardiaceae plant, namely, cotinus coggygria raw material, and then adding ethanol to preprocess; (2) ultrasonically extracting, filtering, and performing pressure-reduction concentration for the filtrate; (3) absorbing concentration liquid through macroporous resin, washing with water to remove impurity, eluting with ethanol, and collecting the eluant; (4) concentrating and drying the eluant, and separating and purifying through a high-speed counter current chromatograph; and (5) crystallizing. The process has the advantages that the process is simple, the operation is safe and convenient, and the process is suitable for the industrial production.
The present invention relates to an extract from the sumac paint flavin technology, process steps are: (1) the Anacardiaceae sumac plant raw material crushing, adding ethanol pretreatment; (2) ultrasonic extraction, filtration, and the filtrate concentrated under reduced pressure; (3 ) concentrate on macroporous resin adsorption, purify water, ethanol eluate; (4) eluate was concentrated by high speed countercurrent chromatography separation and purification after drying; (5) crystals. Advantage of the present invention is a simple process, easy to operate safe, suitable for industrial production.
TECHNICAL FIELD
The present invention belongs to the field of natural medicine extraction, particularly to a paint flavin extracted from sumac in the process.
Background technique
Joseph, also known as non-prime paint Flavin, acid yellow paint, purple riveting element, fisetin, fisetin formula C 15 H 10 O 6, molecular weight 286.23, the formula:
Fisetin slightly soluble in acetone, methanol, ethanol, almost insoluble in ethyl acetate, chloroform, benzene and acidic aqueous solution, an aqueous alkaline solution, increased solubility as the pH rises.
Existing paint flavin extraction method is generally used conventional extraction methods of flavonoids, due to the different nature of the flavonoids, extraction and purification methods are different, the same flavonoids extraction methods are also different in different plants, and therefore, the method is too complicated process of flavonoids , the cost is too high. Therefore, to provide a simple process of paint flavin extraction process is imperative.
SUMMARY
The present invention provides a method for extracting from sumac in paint flavin, the process is simple, low cost, easy to operate safe, suitable for industrial production.
The present invention is by following these steps:
A process for extracting paint flavin from sumac, which is characterized by comprising the steps of:
1) The sumac and dried leaves crushed, over 40 mesh sieve, added 8-10 times the amount of ethanol pretreatment;
2) raw materials preprocessed ultrasonic extraction 1-2 times, filtered, and the filtrate was concentrated under reduced pressure;
3) concentrate by macroporous resin, washed with water, then 6-8BV40% -60% ethanol eluted resin column eluent is collected;
4) eluate was concentrated and dried using high speed countercurrent chromatography separation and purification to give the paint flavin crude;
5) The crude product was dissolved in ethanol was heated to reflux, hot filtered and the filtrate was slowly dropped deionized water, allowed to stand for crystallization, crystals are filtered and dried to obtain paint flavin products.
Step 1) pretreating the 60% -80% ethanol solution soak 1-2h.
Step 2) The ultrasonic extraction auxiliary heating temperature of 30 ° C, the extraction time is 20-40min, power 500-800w.
Step 3) of the macroporous resin can be selected SPD100, HPD100, D101, AB-8, XAD-4 or XAD-7 one.
Step 4) of the high-speed countercurrent chromatography with ethyl acetate - n-butanol - water (3 to 6: 1 to 5: 4-7) for the two-phase solvent, relative to the stationary phase and the lower phase as the mobile phase, lower phase sample is dissolved, the host speed of 800 ~ 1000rmp, the mobile phase was 1 ~ 3ml / min.
The present invention uses ultrasonic extraction cycle is short, effective material to avoid prolonged heating; high-speed countercurrent chromatography of the crude extract of Chinese medicine separation and purification, simple, rapid, while avoiding sample loss; solve the traditional process of separating long period, a small amount of separation, low product purity disadvantage, suitable for industrial production.
detailed description:
Example 1:
Take 5kg sumac and dried leaves, crushed, over 40 mesh sieve, investment in equipment chamber, adding 8 times the amount of 60% ethanol extraction, ultrasonic 20min, temperature 30 ° C, ultrasonic power 800w, filtered, and the filtrate was concentrated under reduced pressure until no alcohol taste concentrated solution through AB-8 macroporous resin column, first washing to no alcohol taste, then 6BV50% ethanol eluted alcohol eluate was concentrated and dried to obtain dried;
The ethyl acetate - n-butanol - water 3: 1: 4 volume ratio, uniformly mixed Still stratification, lower phase as the mobile phase, relative to the stationary phase, dried material dissolved in the lower phase, the stationary phase pump full column, and then adjust the speed of the host 1000rmp, pumped flow rate of 3ml / min, until after the column within the solvent system dynamic equilibrium established by the injection valve injection, and then the spectrum detector receives the target fractions and evaporated to dryness to obtain paint flavin crude 36g, the crude product was dissolved in ethanol was heated to reflux, hot filtered and the filtrate was slowly dropped deionized water, standing crystallized crystals were filtered off and dried to obtain the paint flavin products by HPLC content of 99.7%.
Example 2:
Take 10kg sumac and dried leaves, crushed, over 40 mesh sieve, investment in equipment chamber, 10 times the amount of 80% ethanol extraction, ultrasonic 40min, temperature 30 ° C, ultrasonic power 600w, filtered, and the filtrate was concentrated under reduced pressure until no alcohol taste , concentrate over XAD-4 type macroporous resin column, first washing to no alcohol taste, then 8BV60% ethanol eluted alcohol eluate was concentrated and dried to obtain dried;
The ethyl acetate - n-butanol - water 6: 5: 7 volume ratio, uniformly mixed Still stratification, lower phase as the mobile phase, relative to the stationary phase, dried material dissolved in the lower phase, the stationary phase pump full column, and then adjust the speed of the host 900rmp, pumped flow rate of 2ml / min, until after the column within the solvent system dynamic equilibrium established by the injection valve injection, and then the spectrum detector receives the target fractions evaporated to dryness to give the crude was fisetin 68g, the crude product was dissolved in ethanol was heated to reflux, hot filtered and the filtrate was slowly dropped deionized water, standing crystallized crystals were filtered off and dried to obtain paint flavin products by HPLC detecting content of 98.8%.
US8551537
Rhus verniciflua stokes extract having increased content of active flavonoid compound and method for preparing same
Rhus verniciflua stokes extract having increased content of active flavonoid compound and method for preparing same
Inventor(s): PARK SANG-JAE; KIM KYUNG-HEE, etal.
The present invention relates to Rhus verniciflua Stokes extract having an increased content of an active flavonoid compound through a gas bubbling treatment, a method for preparing the Rhus verniciflua Stokes extract, and a method for converting fustin into fisetin through the gas bubbling treatment of a solution containing fustin. The method for preparing the Rhus verniciflua Stokes extract of the present invention can convert the conventional Rhus verniciflua Stokes extract having high content of fustin and low content of fisetin into a Rhus verniciflua Stokes extract having high content of fisetin. Therefore, the Rhus verniciflua Stokes extract of the present invention can be commercialized into natural anticancer agents, anticancer and cancer prevention food products, and health functional foods due to the excellent bioactivity thereof, and promote the development of relevant industries through the high added-value of Rhus verniciflua Stokes. In addition, the method for converting fustin into fisetin can be applied to other natural substances and be used as a method for enhancing the functionality thereof by increasing the content of highly active flavonoid, thereby becoming an enabling technology that can lead high-functionalization of natural substances.
that they are complete, up-to-date or fit for specific purposes.
CROSS-REFERENCE TO RELATED APPLICATION
This application is a national phase entry of international patent application no. PCT/KR2010/009375 filed Dec. 27, 2010 and claims the benefit of Korean patent application No. 2010-0077410, filed Aug. 11, 2010, the disclosures of which are incorporated herein by reference in its entirety.
BACKGROUND
1. Field of the Invention
The present invention relates to a method for preparing a Rhus verniciflua stokes extract having an increased content of an active flavonoid compound by performing a gas bubbling treatment on the Rhus verniciflua stokes extract; the Rhus verniciflua stokes extract prepared by the above method; and a method of converting fustin into fisetin by performing a gas bubbling treatment on a solution containing fustin.
2. Discussion of Related Art
Rhus verniciflua stokes is fallen leaves broad leaf arborescent belonging to the Anacardiaceae family and it is known that Rhus verniciflua stokes is originally from the Himalayas and highlands of Central Asia. It is now distributed widely throughout the world, from the subtropics to the temperate regions, with the tropics as the center. Rhus verniciflua stokes is fallen leaves or evergreen trees and most of them are shrub trees or tall trees, but some of them are climbing trees (Barkley Fred Alexander., Ann. of the Missouri Bot. Garden., 24(3), pp 265-500, 1937).
A sap of Rhus verniciflua stokes is known as an oriental lacquer. In terms of oriental medicine, it is widely known that a dried oriental lacquer removes extravasated blood; improves blood circulation; and is effective in reducing or alleviating hookworm, stomachache, excessive acid in the stomach, thick sputum, tuberculosis, period pains, constipation, diabetes, malaria, anti-inflammatory, arthritis, and is useful for use as a preservative, strengthening the stomach, easing menstruation pain, and the like, and it is recently known that it is effective in preventing cancer (Namba, T., Colored Illustrations of Wakan Yaku. p 215, Hoikusha Publishing Co. Ltd., Osaka, 1980). In addition, in terms of common medicine, it is known that it is useful as a digestive medicine; it controls extravasated blood in the liver; it controls cardiac disease as a drug for cleaning blood in the heart; removes tuberculosis germs in the lung; and also it is an excellent drug for neuralgia, arthritis, skin diseases, and the like because Rhus verniciflua stokes and a wild ginseng are comparable to each other in effect.
It is already known from much research that Rhus verniciflua stokes include a lot of anti-oxidative substances. Specifically, it is known that the Rhus verniciflua stokes extract that is extracted by using ethanol has strong anti-oxidative activity and also the fraction that is isolated and purified by using a silica column has an ability for inhibiting the growth of tumor cells in the human blood. In addition, it is also known that a substance having an antimicrobial effect is isolated from the Rhus verniciflua stokes extract that is extracted by using ethanol and Rhus verniciflua stokes skin extract that is extracted by using methanol has various bioactivity effects related to the Rhus verniciflua stokes extract, such as an effect on suppressing obesity.
Compounds that are found in Rhus verniciflua stokes genus until now include fisetin, fustin, agathisflavone, eicosanedioic acid, europetein, butein, corilagin, 3′4′-dihydroxy-flavone, lantabetulic acid, myricetin, syringin, semialatic acid, palasitrin, sulfuretin, 3-pentadecyl-1,2-benzenediol, demethoxykanugin, ovalitenone, semimornic acid, 2-(3,4-dihydroxybenzyl)-2,6-dihydroxy-3(2H)-benzofuranone, mesuaferrone A, resokaempferol, rhoifolin, rhusflavanone, succedaneaflavanone, fisetin; 7-0-β-D-glucopyranoside, bhilawanol, tannin, hydrolaccol, stellacyanin, quercetin, cynarine, and the like, and the largest content of the components among them is fustin.
The above components are mostly flavonoid-based materials and flavonoids, such as fisetin and fustin, which play a role in protecting blood vessels or capillaries. And also, the fisetin and fustin are very excellent bioactive substances having antioxidative activity, anti-inflammatory properties, and anticancer properties. However, the fustin that is plentifully included in the Rhus verniciflua stokes extract has a disadvantage wherein its activity is greatly decreased as compared to the activity of fisetin in terms of bioactivity. On the other hand, the fisetin has activitystrong anticancer activity so that it is now being used as an anticancer drug, but there is a disadvantage that a major component of most conventional Rhus verniciflua stokes extracts is the fustin and the fisetin is included in small quantity as compared to the fustin.
Accordingly, when the Rhus verniciflua stokes extract containing a lot of fisetin with excellent bioactivity may be prepared, the Rhus verniciflua stokes extract may have relatively excellent anticancer activity and bioactivity so that it can be expected to be very useful in the food and medicine industries.
SUMMARY OF THE INVENTION
Accordingly, the present inventors preformed research into a method for preparing a Rhus verniciflua stokes extract having very excellent bioactivity by controlling the content of a flavonoid compound in the Rhus verniciflua stokes extract.
Therefore, the present inventors completed the present invention by developing a method for preparing new Rhus verniciflua stokes extract including a large quantity of fisetin having various bioactivities, as well as anti-oxidative activities and anticancer activities.
Accordingly, it is an object of the present invention to provide Rhus verniciflua stokes extract with an increased content of an active flavonoid compound by converting fustin, a flavonoid compound, into fisetin, an active flavonoid compound, in which the active flavonoid compound is plentifully included in the Rhus verniciflua stokes extract.
In order to achieve the above objects, there is provided a method for preparing a Rhus verniciflua stokes extract with an increased content of an active flavonoid compound, including: extracting Rhus verniciflua stokes extract using an extraction solvent; and performing a gas bubbling treatment into the Rhus verniciflua stokes extract.
In an embodiment of the present invention, the gas may include oxygen.
In an embodiment of the present invention, the gas bubbling treatment may be performed by using at least one way of implementation thereof along with the extraction of Rhus verniciflua stokes at the same time, or after completing the extraction of Rhus verniciflua stokes. Preferably, the gas bubbling treatment may be performed by at least one method of directly bubbling gas through the extract solution inside an extractor, or contacting gas with the extract solution outside the extractor.
In an embodiment of the present invention, the active flavonoid is fisetin.
In an embodiment of the present invention, the Rhus verniciflua stokes extract may be extracted by at least one solvent for extracting selected from the group consisting of water, alcoholic solvent, methanol, ethanol, propanol, isopropanol, butanol, acetone, ether, benzene, chloroform, ethylacetate, methylenechloride, n-hexane, hydrochloric acid, acetic acid, formic acid, citric acid, cyclohexane, and combination thereof.
In an embodiment of the present invention, the gas may be air and the time for the gas bubbling treatment may be 6 to 24 hours. Preferably, the time for the gas bubbling treatment may be 8 to 12 hours.
In an embodiment of the present invention, the gas may be oxygen with high purity, preferably at least 95% (v/v), and more preferably pure oxygen, and the time for the gas bubbling treatment may be 5 to 12 hours. Preferably, the time for the gas bubbling treatment may be 7 to 10 hours.
In addition, the present invention provides a Rhus verniciflua stokes extract including fisetin prepared by performing a gas bubbling treatment, wherein the content ratio of fisetin:fustin in the extract is 1:0 to 2:1. This means that the content ratio value of fisetin/fustin is 2 or more.
In addition, the present invention provides a method of converting fustin into fisetin, including performing a gas bubbling treatment into a solution containing fustin.
In an embodiment of the present invention, the gas may be air and the time for the gas bubbling treatment may be 6 to 24 hours. Preferably, the time for the gas bubbling treatment may be 8 to 12 hours.
In an embodiment of the present invention, the gas may be oxygen with high purity, preferably at least 95% (v/v), and more preferably pure oxygen, and the time for the gas bubbling treatment may be 5 to 12 hours. Preferably, the time for the gas bubbling treatment may be 7 to 10 hours.
In addition, the present invention provides a Rhus verniciflua stokes extract including fisetin, wherein the content ratio of fisetin:fustin is 1:0 to 2:1.
In addition, the present invention provides a pharmaceutical composition including the Rhus verniciflua stokes extract as an effective component for preventing or treating cancer.
In addition, the present invention provides a health functional food including the Rhus verniciflua stokes extract as an effective component for preventing cancer or improving cancer symptoms.
BRIEF DESCRIPTION OF THE DRAWINGS
The above and other objects, features and advantages of the present invention will become more apparent to those of ordinary skill in the art by describing in detail exemplary embodiments thereof with reference to the attached drawings, in which:
FIG. 1 is a graph showing an effect on converting fustin into fisetin according to time when air bubbling treatment is applied to Rhus verniciflua stokes extract; and
FIG. 2 is a graph showing an effect on converting fustin into fisetin according to time when high purity oxygen bubbling treatment is applied to Rhus verniciflua stokes extract.
DETAILED DESCRIPTION OF EXEMPLARY EMBODIMENTS
The present invention provides a method for preparing a Rhus verniciflua stokes extract having an increased content of an active flavonoid compound, including extracting Rhus verniciflua stokes extract using an extraction solvent; and performing a gas bubbling treatment into the Rhus verniciflua stokes extract.
Rhus verniciflua stokes is known as a fallen leaves broad leaf arborescent belonging to the Anacardiaceae family; it is spread throughout subtropical zones and tropical regions; and 60 genus and 400 species trees belong to Rhus verniciflua stokes all over the world. It is known that one of active flavonoids that are included in Rhus verniciflua stokes extract, fisetin (2-(3,4-dihydroxyphenyl)-3,7-dihydroxy-4H-chromen-4-one-hydrate, 3,3′,4′,7′-tetrahydroxyflavone) has an excellent effect in a wide variety of fields, as follows: anticancer activities (Haddad A Q et. al., Nutr. Cancer, 2010, 62(5), 668-81), anti-dementia activities (Zheng L T et. al., Int. Immunopharmacol., 2008, 8(3), 484-94), memory improvement (Maher P. et. al., PNAS, 2006, 103(44), 16568-73), an improvement of symptoms of arthritis (Lee J D et. al., Int. Immunopharmacol., 2009, 9(3), 268-76), anti-inflammatory activities (Geraets L. et. al., Biochem. Biophys Res Commun. 2009, 382(3), 598-603), an improvement of blood circulation (Park Y H et. al., J. Ocul Pharmacol. Ther., 2004, 20(3), 189-200), and the like.
In Rhus verniciflua stokes, the content of flavonoid component that is known as a medical component is increased with tree age. Generally, only when the tree age is over 10 years, the content of flavonoid may be included in about 15 to 20% (w/w) among the extract based on the extract. 13 to 17% (w/w) among them is fustin (2,3-Dihydrofisetin) represented by Chemical Formula 1, 1 to 3% (w/w) is fisetin represented by Chemical Formula 2, and then trace amounts of sulfuretin, butein, and the like are included.
It is known that fustin that is plentifully included in Rhus verniciflua stokes extract has antioxidative activity, anticancer activity, etc. like fisetin, but useful bioactivity of fustin is greatly decreased as compared to that of fisetin. The comparison experiment for antioxidative activities between fustin and fisetin that was confirmed by the present inventors supports the above fact (see Table 1). That is, according to an embodiment of the present invention, it has been seen that the antioxidative activity of fisetin was excellent by at least 5 times as compared with that of fustin as a result of the comparison experiment for anti-oxidative activities between fustin and fisetin.
TABLE 1
Anti-oxidative Activity
Substance (IC50, ug/ml)
Fustin 33
Fisetin 8
Accordingly, since fustin has relatively low bioactivity effect as compared with that of fisetin that is an active flavonoid having excellent anticancer and anti-oxidative activities, fustin refers to a “non-active flavonoid” for the present invention.
Therefore, the present invention provides a method for preparing a Rhus verniciflua stokes extract by converting fustin that is plentifully included in Rhus verniciflua stokes extract into fisetin that is an active flavonoid with excellent bioactivity, and the Rhus verniciflua stokes extract prepared according to the present invention includes a large quantity of an active flavonoid compound.
The method for preparing the Rhus verniciflua stokes extract according to the present invention includes performing a gas bubbling treatment on Rhus verniciflua stokes extract.
The Rhus verniciflua stokes refers to the trees belonging to the Anacardiaceae family, and a type of useable Rhus verniciflua stokes may include any one or more selected from the group consisting of Rhus trichocarpa MIQ, Rhus verniciflua, Rhus cotinus, Rhus ambigua H.Lev., Succedanea Sumac, and Rhus succedanea L., but the present invention is not limited thereto. Preferably, Rhus verniciflua may be used.
A proper solvent for extracting Rhus verniciflua stokes extract according to the present invention may include water and an organic solvent, and preferably may include alone or a combination of various solvents, such as water, alcoholic solvent, methanol, ethanol, propanol, isopropanol, butanol, acetone, ether, benzene, chloroform, ethylacetate, methylenechloride, n-hexane, hydrochloric acid, acetic acid, formic acid, citric acid, cyclohexane, petroleum ether, and the like. More preferably, water, an alcoholic solvent, and methanol may be used, and most preferably, water may be used.
The Rhus verniciflua stokes extract according to the present invention may be prepared by a general method for preparing an extract, and specifically, may be prepared by a hot-water extraction method, a macerating extraction method, a digesting extraction method, and the like, and may be prepared by using a general extracting machine, sonicator, or fractionators. In addition, the extract is extracted by using a solvent and then filtering, concentrating, or drying may be selectively performed to remove the solvent from the extract or all of filtering, concentrating, and drying may be performed to remove the solvent from the extract. Specifically, the filtering may include decompression filtering or pressurization filtering using a filter for filtering, and the concentrating may include decompression concentrating in a vacuum. In addition, water of the resulting extract may be completely removed through the concentrating and drying, and the Rhus verniciflua stokes extract without water may be used in a type of powder or the powder may be solved in distilled water or general solvent to use. Accordingly, the Rhus verniciflua stokes extract including a high content of an active flavonoid compound that is obtained by extracting and converting the Rhus verniciflua stokes extract according to the present invention may be commercialized as an extract powder or extract solution through filtering, concentrating, or drying process.
In addition, useable gas in the bubbling treatment may include gas containing oxygen, if it does not disrupt the reaction, preferably, high purity oxygen, and more preferably, pure oxygen, but the present invention is not limited thereto.
A method for preparing the Rhus verniciflua stokes extract with an increased content of an active flavonoid compound according to the present invention are described in more detail, as follows:
Water, alcoholic solvent, an organic solvent, or diluents solution thereof is added as a solvent for extracting Rhus verniciflua stokes, and then is heated to extract. In the extracting process, gas, preferably the gas containing oxygen is bubbled through the extraction solution or concentration solution. The above process is performed to convert fustin, a non-active flavonoid compound into fisetin, active flavonoid, in which the fustin is plentifully included in the extract extracted from Rhus verniciflua stokes. The gas bubbling process may be performed with extracting at the same time, after extracting for a certain time, or after making the concentration solution through the concentrating of the extraction solution that is completely extracted.
The bubbling gas may include a gas containing oxygen, preferably, air or high purity air, and preferably, pure oxygen. When the purity degree of oxygen is high, it has an advantage that even though the processing time is short; the same effect can be obtained. The purity of oxygen included in the bubbling gas is 20% (v/v) in the case of air. According to a method of supplying high purity oxygen with at least 95% (v/v) purity, the higher the purity of oxygen, the shorter the time required for converting fustin into fisetin; and the lower the purity of oxygen, the longer the time required thereby requiring a considerable time for completely converting.
When the bubbling treatment is performed for a short time, less than 1 hour, fustin is only slightly or very slightly converted into fisetin so that there is a Lag-time, in which the components are not changed for a period of time, such that a proper gas bubbling treatment time is required. On the other hand, excessive oxygen bubbling causes decomposition after converting fustin into fisetin so that it is important that a proper time should be selected.
A case of applying the similar method as the method of the present invention to remove an allergy inducing material is disclosed in Korean Patent No. 10-0918326, but there is no technology for converting fustin into fisetin with high activity. The removal of an allergy inducing material is performed along with a supply of oxygen at the same time and completed within a short time, while the conversion of fustin into fisetin is gradually preformed at the end of completing the removal of the allergy inducing material. Accordingly, the conversion of fustin into fisetin may be a new technological idea that is not disclosed or has not been invented in the conventional art for a processing condition thereof.
When the gas for bubbling is air, it is preferable that the air bubbling treatment is continuously maintained for 6 to 24 hours, and more preferably for 8 to 12 hours in order to prepare Rhus verniciflua stokes extract with an increased content of an active flavonoid compound according to the present invention.
In addition, when the gas for bubbling is high purity oxygen, it is preferable that the bubbling treatment is continuously maintained for 5 to 12 hours, and more preferably for 7 to 10 hours in order to prepare Rhus verniciflua stokes extract with an increased content of an active flavonoid compound according to the present invention.
The amount and time for bubbling oxygen may be generally based on volumetric flow rate (l/min) of gas, and preferably, unit volumetric flow rate (VVM, l/min/volume) which is the volumetric flow rate divided by a volume of solution. The time for supplying of air may be 1 to 40 hours depending on a gas supplying flow. However, when the time passes 12 hours, most of fustin is converted into fisetin, such that further supplying of air is not substantially need. Of course, when the supply rate of gas is very slow, the time for completely converting into fisetin may require at least 20 hours. Accordingly, the conversion rate of fustin into fisetin may be possibly controlled by changing a flow rate of gas or the time for supplying gas.
Therefore, the present invention may provide Rhus verniciflua stokes extract with an increased content of an active flavonoid compound due to the gas bubbling treatment, and also provide Rhus verniciflua stokes extract, in which 45 to 100% of the conventional content of fustin presented in Rhus verniciflua stokes extract may be converted into fisetin. That is, all of fustin that is an essential component of the conventional Rhus verniciflua stokes extract can be substantially converted into fisetin through a method of extracting Rhus verniciflua stokes according to the present invention so that there may be substantially no fustin in the Rhus verniciflua stokes extract according to the present invention.
Therefore, the present invention may provide Rhus verniciflua stokes extract having the content ratio of 1:0 to 2:1 of fisetin:fustin.
The bubbling condition of gas through Rhus verniciflua stokes extract is varied according to the pressure or purity of the used gas. That is, in relation to the pressure, the bubbling may be continuously performed on the bottom of an extractor through a blower, etc. under atmospheric pressure, or may be performed by supplying high-pressure gas. It may not be affected by the internal pressure of the extractor. That is, even when the internal pressure of the extractor or concentrator is maintained within a range of 0 to 10 atmospheric pressures, the effect may not be changed. The greater the pressure, the greater the solubility of oxygen is increased so that the processing time may be reduced, but economic feasibility and stability may be significantly reduced due to a sharp increase of installation cost in order to maintain high pressure.
For a method for preparing a Rhus verniciflua stokes extract according to the present invention, the gas bubbling may be generally disclosed in the case of supplying at the bottom of the extractor, but may be performed by using a way of supplying gas through an appropriate pipe after installing the appropriate pipe inside the extractor. Also, the gas bubbling may be performed by a way of separately supplying a gas to allow for contact between the extract solution and the bubbling gas outside the extractor. Here, it may be a general system of contacting liquid and gas, and the system may include a wider area that can allow for contact between the gas and extract solution as a separate space or in the upper of the extractor. Accordingly, the gas bubbling treatment according to the present invention may include a method of bubbling gas through the extract solution of Rhus verniciflua stokes inside the extractor that is used for extracting, a method of contacting the bubbling gas and the extract solution outside the extractor, a method of flowing the gas and extract solution in the same directions or reverse directions at the same time, or a method of using an apparatus, such as an inline mixer, as a machine for mixing the gas and extract solution.
The Rhus verniciflua stokes extract prepared by the method of the present invention may be reconstituted as a fractional material with a high content of fisetin through a further fractionation process. That is, when the solution fractionation may be performed by using acetone, ethyl acetate, n-butanol, chloroform, and the like, as organic solvent, the fractional material that is improved by 30 to 45% (w/w) of fisetin content may be prepared from the extract with 5 to 25% (w/w) of fisetin content. In addition, the purified material with at least 90% (w/w) purity of fisetin may be obtained by performing re-crystallization of the same. However, when the method for preparing a Rhus verniciflua stokes extract according to the present invention is not applied, the fisetin with high purity may not be easily obtained by fractionation and/or re-crystallization due to an interference of fustin even though the organic solvent is used. Accordingly, in case of performing the fractionation and/or re-crystallization according to the inventive conversion method, there is provided an effective way of obtaining plentiful amounts of high purity fisetin.
Generally, a method for preparing a Rhus verniciflua stokes extract may include adding water, alcoholic solvent, or diluted alcoholic solvent to Rhus verniciflua stokes, extracting the same, and then concentrating and/or drying the same. In this case, the composition of flavonoid in the Rhus verniciflua stokes extract may be composed of 10 to 30% (w/w) of fustin, 1 to 4% (w/w) of fisetin, and 0.1 to 2%(w/w) of other flavonoid content. In the case of flavonoid, the content is increased with tree age. The rate of fustin and fisetin in Rhus verniciflua stokes may be usually maintained within a constant range, and the whole content is increased with tree age. In this case, the content ratio of fisetin:fustin may be from the minimum 1:2.5 to the maximum 1:30, and generally 1:5 to 1:15.
Like this, the content ratio of fisetin:fustin may be generally in the range of 1:2.5 to 30. However, in the case of Rhus verniciflua stokes extract according to the inventive method for preparing a Rhus verniciflua stokes extract, the content ratio of fisetin:fustin may be adjusted low by less than 1:0.2. Generally, the difference of activities may be great since the content ratio of fisetin:fustin of 1:1, and it may preferably be 5:1, which means that the content of fisetin is 5 times the content of fustin and also the content of fisetin is significantly increased as compared with the conventional Rhus verniciflua stokes extract. In this case, the content of polyphenol that is known to have bioactivity, such as anti-oxidative activity, immunity-boosting properties, and the like is also very highly maintained at 40 to 70% (w/w).
When fustin is substantially and completely converted into fisetin by supplying high purity oxygen, the content ratio of fisetin:fustin may be maintained at a level of 1:0.001. It means that almost all of fustin is converted into fisetin and the Rhus verniciflua stokes extract with high content of fisetin obtained from the above process may become a Rhus verniciflua stokes extract having high bioactivity.
Accordingly, as compared with the conventional Rhus verniciflua stokes extract, the Rhus verniciflua stokes extract having an increased content of active flavonoid compounds according to the present invention may have excellent bioactivities, such as anticancer activity, anti-dementia activity, an improvement of memory, an improvement of arthritis, anti-inflammatory activity, anti-oxidative activity, an improvement of blood circulation, and the like.
According to an embodiment of the present invention, as compared with the conventional Rhus verniciflua stokes extract, it has been seen that the Rhus verniciflua stokes extract prepared through the inventive preparation method has very excellent anticancer and anti-oxidative activities (see Examples 3 and 4). Especially, with reference to the anticancer activity of the Rhus verniciflua stokes extract according to the present invention, according to an embodiment of the present invention, it has been seen as a result of comparing the effects on inhibiting growth of cancer cells in mice injected with of the conventional Rhus verniciflua stokes extract and the Rhus verniciflua stokes extract of the present invention that the weight and volume of cancer in the mice injected with the Rhus verniciflua stokes extract having a large quantity of fisetin according to the present invention are significantly decreased as compared with the mice injected with the conventional Rhus verniciflua stokes extract (see Table 3). The above result supports the reported excellent anticancer activity of the Rhus verniciflua stokes extract according to the present invention having a large quantity of fisetin that is known to have excellent anticancer activity.
Therefore, the present invention may provide an anticancer composition including the Rhus verniciflua stokes extract with the content ratio of fisetin:fustin of 1:0 to 2:1 as an effective component, and the composition according to the present invention may be used for treating and preventing cancer as a pharmaceutical composition. In addition, the anticancer composition according to the present invention may include 0.1 wt % to 100 wt % of the Rhus verniciflua stokes extract based on total weight of the composition.
A type of cancer that can be treated with the composition according to the present invention may be liver cancer, stomach cancer, colorectal cancer, lung cancer, breast cancer, rectal cancer, leukemia, pancreatic cancer, and the like, but the present invention is not limited thereto.
The anticancer composition according to the present invention may include alone a pharmaceutically effective amount of the Rhus verniciflua stokes extract, or at least one of a pharmaceutically acceptable carrier, excipient, or diluents. The term “a pharmaceutically effective amount” refers to a sufficient amount for preventing, improving and/or treating cancer symptoms.
A pharmaceutically effective amount of the Rhus verniciflua stokes extract according to the present invention may be 0.5 to 100 mg/day/weight kg, and preferably 0.5 to 5 mg/day/weight kg. However, the pharmaceutically effective amount may be appropriately changed according to a degree of cancer symptoms, an age of patient, a weight of patient, a health condition of patient, a sex of patient, an administering route, a period of treatment, and the like.
The expression “pharmaceutically acceptable” refers that it may be physiologically acceptable, and a side effect or the similar effect thereof, such as a gastroenteric trouble, and dizziness, may not generally be caused when administering to humans. Examples of the carrier, excipient, and diluents may include lactose, dextrose, sucrose, sorbitol, mannitol, xylitol, erythritol, maltitol, starch, acacia rubber, alginate, gelatin, calcium phosphate, calcium silicate, cellulose, methylcellulose, polyvinylpyrrolidone, water, methyl hydroxybenzoate, propyl hydroxybenzoate, talc, magnesium stearate, and minerals. In addition, they may further include a filler, anticoagulants, lubricant, wetting agent, flavoring, emulsifying agent, preservatives, and the like.
In addition, the composition of the present invention may be formulated by using a method that is known in the art in order to provide a rapid, continuous, or delayed release type of an active component after administering to a mammal. The dosage form may be a type of powder, granule, tablet, emulsion, syrup, aerosol, soft or hard gelatin capsule, a sterile injection solution, or a sterile powder.
The anticancer composition according to the present invention may be administered through various routes, such as oral, a percutaneous, a subcutaneous, an intravenous, or an intramuscular injection. The dosage of active component may be appropriately selected according to various factors, such as an administering route, an age, a sex, and a weight of patient, severity of patient, and the like. In addition, the anticancer composition of the present invention may be administered along with the compounds that are known to have an effect on preventing, improving, or treating cancer symptoms. Accordingly, the present invention may provide a medicine for preventing and/or treating cancer symptoms, in which the medicine includes the Rhus verniciflua stokes extract according to the present invention.
Moreover, the anticancer composition according to the present invention may provide an effect on relieving cancer symptoms through functions of excellent anticancer and antioxidative activities, and also since it may be added to food in order to prevent or improve cancer symptoms, the composition of the present invention may be used for food in order to prevent and improve cancer symptoms. Accordingly, the composition of the present invention may be useful in foods having effects on preventing and improving cancer symptoms, such as, main raw materials or added raw materials of foods, food additives, functional foods or beverages.
According to the present invention, the term “food” refers to a natural material or processed material that may include one or more nutrients, and preferably to be eaten after some processing. As a general meaning, it may refer that all of foods, food additives, functional foods, and beverages are included.
The foods that can include the anticancer composition according to the present invention may be, for example, all sorts of foods, beverages, gums, teas, vitamin complexes, functional foods, and the like. Additionally, the foods that may be used in the present invention may be special nutritious foods (for example, milk formulas, young children and baby food, etc.), processed meat food, fish meat food, bean curd food, muk (gel type food), noodles (for example, ramen, noodles, etc.), breads, dietary supplements, seasoning food (for example, soy sauce, Doenjang (Korean traditional sauce), red pepper paste, etc.), sauces, confectionery (for example, snacks), candy, chocolates, gums, ice creams, milk products (for example, fermented milk, cheese, etc.), other processing foods, Kimchi, salting foods (all sorts of Kimchis, Jangajjis, etc.), beverages (for example, fruit beverages, vegetable beverages, soybean milk, fermented drinks, etc.), natural seasoning (for example, ramen soups, etc.), and the like, but the present invention is not limited thereto. The above foods, beverages, or food additives may be prepared by using a general preparation method.
In addition, the term “functional food” refers to a food group of giving a high added value to food function and expresses a function of a relevant food for a particular purpose using physical, biochemistry, and/or biotechnology methods, and the like and also a processed food by designing for sufficiently expressing a body controlling function related to a body defense mechanism, prevention and recovery of disease, and the like, which are functions of food. Specifically, it may be a health functional food. The functional food may include a cytologically acceptable food supplement and may further include an appropriate carrier, excipient, and diluents that are generally used for preparing a functional food.
In addition, the term “beverage” refers to the generic term for drinking for quenching thirst or enjoying taste, and may include a functional beverage. The beverage may include a composition with the disclosed rate for preventing and improving anticancer symptoms as an essential component, and other components, without any special limitation, and also may include various flavouring agents, natural carbohydrates, and the like, as a further component, like the general beverage.
In addition to the above disclosed components, the food including the composition according to the present invention may include various nutritional supplements, vitamins, minerals (electrolyte), flavouring agents, such as synthetic flavouring agents, natural flavouring agents, and the like, colorings, fillers (cheese, chocolates, etc.), pectic acid and salts thereof, alginic acid and salt thereof, organic acid, protective colloid thickener, pH control agent, stabilizer, preservatives, glycerin, alcohol, carbonation agent that is used for a carbonated drink, and the like, and the above components may be used alone or in combination.
For the food including the anticancer composition of the present invention, the amount of the composition of the present invention may be included in 0.001 wt % to 100 wt %, and preferably, 0.1 wt % to 40 wt %. For the beverage, it may be included in the range of 0.001 g to 5 g, and preferably 0.01 g to 2 g based on 100 ml, but it may be below the above ranges when it is taken for a long time for improving health and hygiene, and for managing health. Since, an effective component has no problem in terms of stability, it may be used in the amount of the above ranges, and the present invention is not limited thereto.
Therefore, the present invention may provide a health functional food including the Rhus verniciflua stokes extract as an effective component according to the present invention for preventing cancer or improving cancer symptoms, and a type of the food may be powder, granule, tablet, capsule, or drink, but the present invention is not limited thereto.
On the other hand, a method for preparing a Rhus verniciflua stokes extract having an increased content of an active flavonoid compound according to the present invention may convert a non-active flavonoid compound having a relatively low bioactivity into an active flavonoid compound having excellent bioactivity as mentioned above. At this point, the non-active flavonoid compound may be fustin and the active flavonoid compound may be fisetin. Accordingly, the present invention may provide the method of converting fustin into fisetin.
The method of converting fustin into fisetin may include performing a gas bubbling treatment on a solution containing fustin and the gas bubbling treatment may be performed by bubbling gas through an extract solution inside the extractor or contacting bubbling gas with the extract solution outside the extractor. The solution containing fustin may be an extract extracted from a natural material; and it may be a solution or extract containing fustin and fisetin at the same time; or it may be a solution or extract containing fustin only.
In addition, when the gas for bubbling is air, preferably, the bubbling treatment may be continuously maintained for 6 to 24 hours and more preferably, for 8 to 12 hours in order to prepare the solution containing high content of fisetin through the method of converting fustin into fisetin according to the present invention.
On the other hand, when the gas for bubbling is high purity oxygen, preferably, the bubbling treatment may be continuously maintained for 5 to 12 hours and more preferably, for 7 to 10 hours in order to prepare the solution containing high content of fisetin through the method of converting fustin into fisetin according to the present invention.
Accordingly, the present invention may convert the extract or solution having a large quantity of fustin into the extract or solution having a large quantity of fisetin that is an active flavonoid compound with excellent bioactivity.
Hereinafter, the present invention will be described in more detail with reference to the following Examples.
However, the following Examples are only for illustrating the present invention, and the content of the present invention is not limited to the following Examples.
Example 1
Preparation of Rhus Verniciflua Stokes Extract Through Gas Bubbling
The contents of fustin and fisetin that are included in Rhus verniciflua stokes extract were analyzed using HPLC. The standard products were purchased from Sigma Aldrich. As conditions of analyzing by HPLC, UV of 254 nm was used as a detector and 5% mixing solution of acetic acid:methanol:acetonitrile=70:20:10 was used for a moving phase. Total of 10 ul was injected at the flow rate of 1 ml/min. C18 (250 mm*4.6 mm, YMC Pack) was used as a column. An analysis of polyphenol was performed by using tannic acid as a standard material with Folin-ciocalteu's phenol reagent. An absorbance was measured at 725 nm after developing color, and calculated using an external calibration method with a standard material.
<1-1> Preparation of Rhus Verniciflua Stokes Extract Through Air Bubbling
100 l of water was added to 10 kg of Rhus verniciflua stokes and then extracted at 95° C. for 6 hours. When a large quantity of flavonoid component was included in the extract after 4 hours during extraction, air (rate of oxygen: 20% (v/v)) was bubbled at the bottom of the extractor at the rate of 1.5 l/min. The time for bubbling was totally 12 hours and the components were analyzed by collecting a sample every hour.
As a result, the content of fustin was significantly decreased as time passes and the content of fisetin was increased (FIG. 1). After 11.5 hours, substantially 100% of fustin was converted into fisetin. The Rhus verniciflua stokes extract was filtered, vacuum-concentrated, and then powderized to obtain 0.56 kg of brown dry powder; and the content of fustin in the extract powder that is a final product was 0.01% (w/w); the content of fisetin was 16.7% (w/w); and the content of polyphenol was 56.4% (w/w). That is, the present inventors may obtain the extract, in which all of fustin having low activity was converted into fisetin, an active flavonoid, through a method for preparing a Rhus verniciflua stokes extract according to the present invention.
<1-2> Preparation of Rhus Verniciflua Stokes Extract Through High Purity Oxygen Bubbling
As disclosed in the above Example, <1-1>, 100 l of water was added to 10 kg of Rhus verniciflua stokes and then Rhus verniciflua stokes was extracted at 95° C. When a large quantity of flavonoid component was included in the extract through 4 hours of the extraction time, high purity oxygen (purity: 95% (v/v)) was bubbled at the bottom of the extractor at the rate of 1.2 l/min using an oxygen cylinder. The time for bubbling was totally 12 hours and the components were analyzed by collecting a sample every hour.
As a result, the content of fustin was significantly decreased as time passes and the content of fisetin was increased according to the decrease of fustin content (FIG. 2). At 8.8 hours of treatment time, substantially 100% of fustin was converted into fisetin. The rate of converting was rapid and the conversion was completely processed as compared with the use of air. In addition, when the bubbling time is more than necessary, the content of fisetin prepared by converting fustin was slightly reduced so that it can be known that the proper treating time may be important. The extract was filtered, vacuum-concentrated, and then powderized to obtain 0.55 kg of brown dry powder. The content of fustin in the extract powder that is a final product was 0.00% (w/w); the content of fisetin was 12.9% (w/w); and the content of polyphenol was 58.2%.
Comparative Example 1
Preparation of General Rhus Verniciflua Stokes Extract
100 l of water was added to 10 kg of Rhus verniciflua stokes and then extracted while it was heated at 95° C. for 6 hours. The resulting extract was filtered, vacuum-concentrated, and then powderized to obtain 0.57 kg of brown dry powder. The content of fustin in the extract powder that is a final product was 17.3% (w/w); and the content of fisetin was 1.63% (w/w). The content ratio of fisetin:fustin that were included in Rhus verniciflua stokes extract through a method for preparing the conventional Rhus verniciflua stokes extract was 1:10.6 and the content of polyphenol was 53.1%. That is, in the case of Rhus verniciflua stokes extract through a common method of extracting without a bubbling treatment, the content of fustin was at least 10 times higher than that of the content of fisetin.
Example 2
Preparation of Rhus Verniciflua Stokes Extract Using Various Solvents
The Rhus verniciflua stokes extracts according to the present invention were prepared by using various extraction solvents. To achieve this, purified water, 50% alcoholic solvent, 80% alcoholic solvent, 80% methanol, and 100% alcoholic solvent were used as an extraction solvent. The conversion rate of fustin into fisetin according to hours during extraction process using each of extraction solvents was investigated and then the times for converting 90% were compared each other. The Rhus verniciflua stokes extracts were extracted at 95° C. by adding the extraction solvents that were 10 times to 10 kg of Rhus verniciflua stokes as disclosed in the above Example. And then the conversion rate into fisetin was investigated by sampling the extract every hour while bubbling high purity oxygen at the bottom of an extractor in 0.3 VVM (volume/volume/min). The results are shown in the following Table 2.
TABLE 2
Time Required for 90% Conversion of Fustin into Fisetin According to a Type of Solvent.
Type of Solvent
50% 80% 100%
Alcoholic Alcoholic Alcoholic 80%
Water Solvent Solvent Solvent Methanol
Time required 7.9 12.9 22.2 38.5 24.9
(hrs)
Content ratio of 0.18 0.21 0.23 0.35 0.25
Fustin:Fisetin
As shown in the above Table 2, it has been seen that for the extract prepared by using the method for preparing the Rhus verniciflua stokes extract according to the present invention, the fustin in the extract can be converted into fisetin by performing a gas bubbling treatment regardless of a type of used solvents. However, when the content of water in the solvent was decreased, the processing time was increased. Accordingly, from the above result, the present inventors can confirm that the processing time should be controlled in order to increase the conversion rate into fisetin according to a type of solvent in the extraction conditions.
Example 3
Analysis of Anticancer Activity of Rhus Verniciflua Stokes Extract According to the Present Invention
In order to analyze anticancer activity of the Rhus verniciflua stokes extract according to the present invention, firstly the anticancer activities of Rhus verniciflua stokes extract prepared by the general method of extracting Rhus verniciflua stokes in the above Comparative Example 1, the Rhus verniciflua stokes extract according to the present invention prepared from Example <1-1>, and Sample, “A” of Example 6 were mutually compared.
The analysis of anticancer activity were performed by using a comparison of effect on inhibiting a growth of cancer cell after orally administrating the Rhus verniciflua stokes extract according to the present invention using a mouse. 5×10<5 >cells of human non-small cell lung cancer cell line, A549, were subcutaneously injected to a nude mouse (Immunodeficient mice (male), CanN. Cg-Foxn1nu/CrljBgi). 7 nude mice were assigned for one group after dividing a control group, the group of Comparative Example 1 and the group of Example <1-1>. The extract was not administrated to the control group, the conventional Rhus verniciflua stokes extract extracted from Comparative Example 1 was administrated to the group of Comparative Example 1, and the Rhus verniciflua stokes extract according to the present invention extracted in Example <1-1> was administrated to the group of Example <1-1>. 300 mg/kg volume was orally administrated for 24 days. A tumor volume was calculated by using the following formula (A=Long length, B=Short length) after measuring the long length and short length of the tumor using Caliper two times per one week during an administration period; then at 27 days, the tumor was removed and then its weight was measured.
[mathematical formula]
As a result, as shown in Table 3, it has been seen that the tumor weight and volume in the mouse of Example <1-1> group were significantly small as compared with the animal model group administrated with the conventional extract. From this, it has been seen that the Rhus verniciflua stokes extract according to the present invention has an excellent effect on inhibiting the growth of tumor.
TABLE 3
Comparison of Anticancer Activity of Rhus verniciflua Stokes Extract
<Example <Comparative
Control 1-1> Example 1> Example
Group Group Group 6 “A”
Tumor 2 ± 0.4 0.75 ± 0.12 1.7 ± 0.52 0.45 ± 0.12
Weight (g)
Tumor 610 ± 180 370 ± 92 550 ± 115 180 ± 101
Volume (mm<3>)
Example 4
Analysis of Antioxidative Activity of Rhus Verniciflua Stokes Extract According to the Present Invention
In order to analyze anti-oxidative activity of the Rhus verniciflua stokes extract according to the present invention, firstly the anticancer activities of Rhus verniciflua stokes extract prepared by the general method of extracting Rhus verniciflua stokes in the above Comparative Example 1 and the Rhus verniciflua stokes extract according to the present invention prepared from Example <1-1> were mutually compared.
A measurement of anti-oxidative activity was performed by using a method of measuring an electron-donating ability with DPPH method. DPPH is an abbreviation for 1,1-diphenyl-2-picrylhydrazyl, and is widely used a marker for measuring anti-oxidative activity due to its radical. The testing processes were as follows: (1) 2 ml of 0.1 mM DPPH (Sigma, D-9132) dissolved in ethanol was treated to 2 ml of sample dissolved in ethanol; (2) it was mixed for 10 minutes, and then maintained at a dark place for 30 minutes; (3) an absorbance was measured at 520 nm; and (4) the degree of anti-oxidative ability (electron-donating ability, EDA) was calculated. Vitamin E (Fluka, 95420) was used as a positive control group and Formula was as follow.
[mathematical formula]
In the above formula, Cabs denotes an absorbance of a negative control group (in the case of treating only ethanol instead of sample) and Sabs denotes an absorbance of sample.
TABLE 4
Comparison of Anti-oxidative Activityof Rhus Verniciflua Stokes Extract
Anti-oxidative Activity
Sample Name (IC50, ug/ml)
General Rhus verniciflua 58.9
stokes extract (Comparative Example 1)
Rhus verniciflua stokes 13.3
extract according to the present invention (Example 1-1)
“A” of Example 6 10.5
Pure Fustin 32
Pure Fisetin 7.8
As a result, as shown in Table 4, it has been seen that the anti-oxidative activity of the Rhus verniciflua stokes extract with an enhanced fisetin according to the present invention was at least 4 times excellent as compared with the conventional Rhus verniciflua stokes extract. It means that the anti-oxidative activity was significantly improved by converting fustin that is plentifully included in the conventional Rhus verniciflua stokes extract into fisetin that is an active flavonoid.
Example 5
Preparation of Rhus Verniciflua Stokes Extract Powder with High Content of Fisetin Using Concentration Solution
As shown in Example <1-1>, 10 kg of Rhus verniciflua stokes was added with 10 times water to extract two times and then concentrated to make 20 l of the concentration solution. Oxygen gas was bubbled at 0.4 VVM for 12 hours, and then the prepared Rhus verniciflua stokes extract was concentrated to obtain 470 g of Rhus verniciflua stokes extract powder, in which the content of fustin was 1.2% (w/w) and the content of fisetin was 15.8% (w/w). At this time, the content ratio of fisetin:fustin was 1:0.076.
Example 6
Preparation of Solvent Fraction with High Content of Fisetin
Each 100 g of the extracts obtained from the above Example <1-1> and Comparative Example 1 was dissolved in 5,000 ml of water; 7,500 ml of ethyl acetate was added; then it was intensely mixed for 10 minutes using a mixer; and then it was maintained at room temperature for separating a layer. An upper layer of ethyl acetate was collected; then the solvent was removed; and then components were analyzed. As a result, the content of fisetin was 42.5% (w/w) in the case of Example <1-1> sample (referred to “A”) and the content of fisetin was 7.5% (w/w) in the case of Comparative Example 1 sample (referred to “B”).
Example 7
Preparation of Fisetin Crystalline
10 g of the solid obtained from the above Example 6 was dissolved in 50 ml of ethanol; and then poured into cold water (cold storage) at once. At this time, yellow crystalline of fisetin was weakly produced at the same time. It was centrifuged at 3,000 rpm for 20 minutes to recover. The crystalline obtained by this example was dried and then its purity was analyzed with HPLC. As a result, the content of water was 2.8% (w/w) and the purity of fisetin was 96.5% (w/w).
A method for preparing a Rhus verniciflua stokes extract according to the present invention can provide Rhus verniciflua stokes extract having high content of fisetin by converting fustin into fisetin through a gas bubbling treatment to the conventional Rhus verniciflua stokes extract including low content of fisetin and high content of fustin. Accordingly, the Rhus verniciflua stokes extract having a large quantity of fisetin according to the present invention can be commercialized as anticancer drugs, anticancer foods, foods for preventing cancer, health functional foods, and the like due to its excellent bioactivity. In addition, the developments of the related industries can be induced due to a high added-value of Rhus verniciflua stokes. Also, a method of converting fustin into fisetin according to the present invention can be used as a method of increasing the content of a high active flavonoid by applying to other natural substances thereby becoming the base technology for leading high functionalization of natural substances.
While the invention has been shown and described with reference to certain exemplary embodiments thereof, it will be understood by those skilled in the art that various changes in form and details may be made therein without departing from the spirit and scope of the invention as defined by the appended claims.
CN102924419
Method for extracting fisetin from boxwood
Method for extracting fisetin from boxwood
Inventor(s): FAN XIAOPING; LIU CHUNLIANG; JING FENG +
The invention provides a method for extracting fisetin from boxwood. The method comprises that boxwood as a raw material is subjected to crushing, extraction and crystallization to produce fisetin crystals. The method has the advantages that processes are simple and easy; equipment requirements are low and only simple extraction condensation equipment is adopted; extracted fisetin has the purity of 98%; a fisetin yield is greatly improved and 15 to 20kg of fisetin can be extracted from 1T of the raw material having the content of 1 to 2%; an active ingredient loss is less in the processes; and compared with the prior art, the method can improve a yield about 4-5 times.
DESCRIPTION
The present invention provides a method for extracting from boxwood in paint flavin. It boxwood as raw material, crushing, extraction, crystallization, and then have to paint flavin crystals. Advantage of the present invention is the process simple and easy to operate, less demanding on the equipment, with a simple extraction and concentration equipment, the extracted paint flavin can do 98% purity, and extracted paint flavin yield greatly improved content of 1 % -2% of one ton of raw material can be extracted 15-20Kg product, a small loss of process active ingredients, increased about 4-5 times higher than the yield of the prior art.
The present invention relates to a method for extracting from boxwood in paint flavin.
Background technique
Extraction of the existing paint flavin extraction method is generally used existing flavonoids, mostly organic reagents Extraction or alkaline extraction using hot water extraction and purification methods use more crystallization or column separation from In different types of flavonoids, extraction and purification methods are different, there are different plants of the same flavonoids extract The method is also different. And this extraction method for existing equipment requirements, often using column separation or Extraction, when using column separation, it needs at least one adsorption adsorption plant and ancillary equipment, and the operation The multi-step, the loss of active ingredient must increase, while the use of extraction solvent phase will have each have Ingredients residual effect, so that the desired composition will be dispersed, so in the end can not be all out, it will also Losses during extraction of the active ingredient. Such extraction yield will be low, typically at levels from 1% -2% of one ton Only less than 5Kg raw material extracted products.
SUMMARY
Propose a method for extracting from boxwood in paint flavin present invention, it is possible to effectively overcome the prior art Intraoperative deficiencies and improve the paint flavin extraction yield, but the extraction process is simple.
Aspect of the present invention is achieved by: a method of extracting from boxwood in paint flavin, it Boxwood as raw material, crushing, extraction, crystallization, and then have to paint flavin crystals.
Further, the pulverizing process: the boxwood rods, sticks to smash the teeth 2-15mm grain.
Further, the extraction process is: 1-4% by weight of boxwood slaked lime was added to the crushed Boxwood particles process after the stir, then with 80% ethanol extraction.
Further, the extraction was extracted with 80% ethanol three times, three times in ethanol were added 4-6 times the weight of boxwood, boxwood weight 2-4 times, 2-4 times the weight of boxwood, extraction time was 2h, 1.5h, 1.5h, for the first time to extract the cold soak 25-35min, then heat extraction.
Further, the crystallization process is divided into: coarse crystals fried mention crystallization, high alcohol crystals.
Further, the crude crystals is extract was adjusted to PH 6-8, and concentrated under reduced pressure to gravity 1.05-1.10, precipitate was allowed to stand for cooling, the crude crystals were separated by filtration.
Further, the crystalline mention fried in crude crystals of purified water was added to dissolve boiling under reflux for 25-35min, Then put the liquid, filtered and the filtrate quiet cool placement, and so forth fried mention, crystallization, fried mention crystallization.
Further, the high alcohol is added to the crystalline crystals fried frying mention mention high-quality crystalline 4-6 times Alcohol dissolved once fully dissolved sedimentation after standing, separating the supernatant was added to dissolve crystals of higher alcohols, the amount of body fluid 13-20% of the volume of water, which crystallized on standing to give a high alcohol crystals obtained after vacuum drying paint flavin crystals.
The beneficial effect of the present invention is that: the process is simple and easy to operate, less demanding on the equipment, with a simple mention Take enrichment equipment, the extracted paint flavin purity can do 98%, and the extraction yield improved paint flavin, Content of 1% to 2% of one ton of raw materials can be extracted 15-20Kg products, improved yields than in the prior art 4-5 times.
detailed description
The following example will be the technical implementation of the program of the present invention clearly and completely describe Obviously, the description The embodiment described is only one embodiment of the present invention, but not all embodiments. Based on the present invention The embodiments, those skilled in the art without creative efforts shall be made available to all Other embodiments are within the scope of the present invention is protected.
Example 1:
A method of extracting from boxwood in paint flavin, it boxwood as raw material, crushing, extraction, Crystallization, then give fisetin crystals.
(1) crushing: the 1000Kg content of 1% -2% of boxwood rods, sticks to 3mm pulverized particles.
(2) Extraction: Boxwood particles 15Kg slaked lime was added to the grinding process after the stir, then After extraction with 80% ethanol three times, three times in ethanol were added 4000Kg, 4000Kg, 2000Kg, Extraction time was 2h, 1.5h, 1.5h, performed for the first time to extract the first cold soak 28min, then heated extract.
(3) crystallized: the crystallization process is divided into: coarse crystals fried mention crystallization, high alcohol crystals.
a, crude crystals: the crude crystals is extract was adjusted to PH 6, and concentrated under reduced pressure to a specific gravity of 1.05, The precipitate was allowed to stand to cool, 110Kg give crude crystals were isolated by filtration (wherein the weight of dry matter);
b, fried mention crystallization: the fried mention crystallization in crude crystals of purified water was added to dissolve boiling under reflux for 25min, Then put the liquid, filtered and the filtrate quiet cool placement, and so forth fried mention, crystallization, 25Kg crystallization content of 70% Fried mention about crystallization (weight of dry matter basis);
c, high crystalline alcohol: the alcohol high degree of crystallization is to add alcohol to mention fried crystals ≥95% methanol 112.5Kg Dissolved once fully dissolved sedimentation after standing, separating supernatant and add a high amount of liquid crystals were dissolved the alcohol by volume 13% water, which crystallized on standing to give a high alcohol content ≥98% crystalline 16.7Kg (weight of dry matter basis), true After air drying to obtain paint flavin crystals.
Example 2:
A method of extracting from boxwood in paint flavin, it boxwood as raw material, crushing, extraction, Crystallization, then give fisetin crystals.
(1) crushing: the 1000Kg content of 1% -2% of boxwood rods, sticks to smash particles of 7mm.
(2) Extraction: Boxwood particles 20Kg slaked lime was added to the grinding process after the stir, then After extraction with 80% ethanol three times, three times in ethanol were added 5000Kg, 3000Kg, 3000Kg, Extraction time was 2h, 1.5h, 1.5h, performed for the first time to extract the first cold soak 30min, then heated extract.
(3) crystallized: the crystallization process is divided into: coarse crystals fried mention crystallization, high alcohol crystals.
a, crude crystals: the crude crystals is extract was adjusted to PH 7, and concentrated under reduced pressure to specific gravity 1.07, The precipitate was allowed to stand to cool, 120Kg give crude crystals were isolated by filtration (wherein the weight of dry matter);
b, fried mention crystallization: the fried mention crystallization in crude crystals of purified water was added to dissolve boiling under reflux for 30min, Then put the liquid, filtered and the filtrate quiet cool placement, and so forth fried mention, crystallization, 30Kg crystallization content of 70% Fried mention about crystallization (weight of dry matter basis);
c, high crystalline alcohol: the alcohol high degree of crystallization is to add alcohol to mention fried crystals ≥95% ethanol 150Kg Dissolved once fully dissolved sedimentation after standing, separating supernatant and add a high amount of liquid crystals were dissolved the alcohol by volume 15% water, which crystallized on standing to give a high alcohol content ≥98% crystalline 20.5Kg (weight of dry matter basis), true After air drying to obtain paint flavin crystals.
Example 3:
A method of extracting from boxwood in paint flavin, it boxwood as raw material, crushing, extraction, Crystallization, then give fisetin crystals.
(1) crushing: the 1000Kg content of 1% -2% of boxwood rods, sticks crushed to 13mm particles.
(2) Extraction: Boxwood particles 40Kg slaked lime was added to the grinding process after the stir, then After extraction with 80% ethanol three times, three times in ethanol were added to 5500Kg, 3000Kg, 2500Kg, Extraction time was 2h, 1.5h, 1.5h, performed for the first time to extract the first cold soak 35min, then heated extract.
(3) crystallized: the crystallization process is divided into: coarse crystals fried mention crystallization, high alcohol crystals.
a, crude crystals: the crude crystals is extract was adjusted to PH 8, and concentrated under reduced pressure to a specific gravity of 1.10, The precipitate was allowed to stand to cool, 110Kg give crude crystals were isolated by filtration (wherein the weight of dry matter);
b, fried mention crystallization: the fried mention crystallization in crude crystals of purified water was added to dissolve boiling under reflux for 30min, Then put the liquid, filtered and the filtrate quiet cool placement, and so forth fried mention, crystallization, 25Kg crystallization content of 70% Fried mention about crystallization (weight of dry matter basis);
c, high crystalline alcohol: the alcohol high degree of crystallization is to add alcohol to mention fried crystals ≥95% ethanol 137.5Kg Dissolved once fully dissolved sedimentation after standing, separating supernatant and add a high amount of liquid crystals were dissolved the alcohol by volume 18% water, which crystallized on standing to give a high alcohol content ≥98% crystalline 15.5Kg (weight of dry matter basis), true After air drying to obtain paint flavin crystals.
1000Kg content derived from Example 1-3 in 1% -2% of the raw material extracted boxwood and paint flavin Comparative yield prior extraction techniques in the following table:
From the above table it can be seen according to the present invention, a method of extracting the paint flavin yield than conventional extraction Yield technologies about five times higher, because at least in the present invention, the extraction step of the process, the more steps Less of the active ingredient loss is small, the higher the yield of the product, and the resulting crystalline content can also More than 98%.
The foregoing is only preferred embodiments of the present invention, it is not intended to limit the present invention, where the present Within the spirit and principles of, and any changes made, equivalent substitution and improvement should be included in the present Within the scope of the invention.
CN102526153
Vernonia anthelmintica flavone components, preparation method and application thereof
Vernonia anthelmintica flavone components, preparation method and application thereof
Inventor(s): MING YAN; SHIXIA HUO; LI GAO; XIAOMING PENG +
The invention relates to vernonia anthelmintica flavone components, a preparation method and application thereof. The flavone components are fisetin, butein, 7,8,3',4'-tetrahydroxy flavone, 5,7,8,3',4'-pentahydroxy chalcone, 6,8,3',5'-tetrahydroxy-dihydroflavone, liquiritigenin and isoliquiritigenin, which are prepared from plant vernonia anthelmintica through extraction, separation and purification.
Application of each flavone component in preparation of a medicament for treating leucoderma provides a new medicament choice for treating leucoderma.
The present invention relates to a method of Vernonia flavonoid fractions and preparation and use of the flavonoid component riveting prime purple, purple flower riveting element, 7,8,3 ', 4'-tetrahydroxy flavone, 5,7 , 8,3 ', 4'-hydroxy chalcone five, 6,8,3', 5'-hydroxy flavanone four, glycyrrhizin and ISL seven flavonoids component is from plants of Vernonia seeds used in extraction, separation and purification of flavonoids obtained components, then each component as a flavonoid for the preparation of the treatment of vitiligo drugs, drug treatment of vitiligo provides a new choice.
FIELD:
The present invention relates to a drug-dimensional Vernonia flavonoid fractions and preparation and use, Especially in the manufacture of a medicament for the anti-vitiligo.
Background technique:
Vernonia Asteraceae Compositae Vernonia is an annual herb insecticide spot Dove Ju Vernonia anthelmintica (Linn. ) Willd, medicinal parts of its ripe fruit Solid, three dry heat and dampness swelling, alias India Mountain fennel ( "Flora of China"), Ait Day Rally ( "Uighur"). Only contained in the "Flora of China", "Standard Uygur medicine Quasi "and" Uygur medicine Chi "(the book) are also described. Hotan, Aksu, Yunnan Western cultivation, India, Pakistan and other countries are also planted. Xinjiang Uygur of medication, Uygur medicine called 卡拉孜 its course, is one of the pigment Uygur medicine doctors most commonly used to increase. Included in the People's Republic of China Ministry of Health Drug Standards - Volume Uygur medicine, doctors recognize Uighur Cleared to Vernonia abnormal mucus quality, insect, swelling, pain and cold. For wet cold Stomach pain and liver disease, such as vitiligo. Chinese herbal medicine Uighur drug volume records of Vernonia promote color Pigmentation, restoring skin color, attending vitiligo. Vernonia mainly containing flavonoids And volatile oil; the main active ingredient in the treatment of vitiligo as flavonoids, having Activation of tyrosinase activity, increase skin sensitive to light, improve skin micro-vitiligo lesion site Circulation, immune regulation, cell proliferation and melanocyte functions such as micronutrient supplementation; deworming currently
Vernonia extract has been made of a variety of formulations used in clinical treatment of vitiligo, and achieved remarkable Significant effect.
For the current study reported that the chemical composition of Vernonia less, the present invention is natural medicine Chemically as a means of Vernonia were extraction, separation, purification, for the first time from insecticide Vernonia isolated the active ingredient in the treatment of vitiligo, by in vivo experiments to evaluate the in vitro Efficacy, and the preparation of a pharmaceutical treatment of vitiligo.
SUMMARY:
Object of the present invention is to provide a Vernonia flavonoid components and preparing Law and uses the flavonoid component purple riveting prime purple flower riveting element, 7,8,3 ', 4'-tetrahydroxy yellow Ketone, 5,7,8,3 ', 4'-hydroxy chalcone five, 6,8,3', 5'-dihydro-tetrahydroxy Flavonoids, glycyrrhizin and ISL seven kinds of flavonoids ingredients from plant seeds of Vernonia Using extraction, separation and purification of flavonoids obtained component, then as a component in the manufacture of various flavonoids rule The use of drug treatment of vitiligo, vitiligo treatment for drug provides a new choice.
A flavonoid component of Vernonia present invention, the flavonoid component purple riveting Su, Su purple flower riveting, 7,8,3 ', 4'-tetrahydroxy flavone, 5,7,8,3', 4'-serotonin Chalcone group, 6,8,3 ', 5'-tetrahydroxy-dihydro-flavonoids, glycyrrhizin and ISL, Wherein the total flavonoid content of the component 50 to 99.9%, content of each fraction flavonoid 20-99.9%.
Said Vernonia preparation flavonoid component, the flavonoid component is a drug for peacekeeping The separation and purification of Vernonia, the specific operation the following steps:
a, the Vernonia seeds crushed to 5-80 mesh, 6-12 times the amount of the solvent is water Or concentration of 20 to 95% aqueous ethanol extract 1-4 times, temperature 40-100 ° C, each mention Take time 1-3 hours, the combined extracts were recovered solvent, concentration, vacuum drying, dry Extract to give a total crude extract;
b, step a crude extract of the total concentration of 30-90% water or aqueous ethanol was fully dissolved Solution, with petroleum ether, ethyl acetate or chloroform 2-7 times, each time with an amount extraction solvent 1:1-4 solution volume ratio of the extract was concentrated to obtain dry extract;
c, chloroform - methanol solution or an aqueous solution at a concentration of 40-90% ethanol eluent, Polyamide, dextran or silica gel chromatography separation eluting 6-30 column Volume, respectively eluate eluate concentrated under reduced pressure, drying, Vernonia Flavone components.
Or step to give a total crude extracts of Vernonia Huang high-speed countercurrent chromatography separation One component.
The use of the flavonoid component of Vernonia, and the Vernonia flavonoid group Points in the preparation of drugs in the treatment of vitiligo.
Vernonia flavonoid ingredients and preparation methods and uses of the present invention, wherein Vernonia flavonoid component riveting prime purple, purple flower riveting element, 7,8,3 ', 4'-tetrahydroxy yellow Ketone, 5,7,8,3 ', 4'-hydroxy chalcone five, 6,8,3', 5'-dihydro-tetrahydroxy Flavonoids, glycyrrhizin and ISL, the chemical structural formula are of formula (1) - (7).
(1) purple riveting element; (2) riveting purple flower elements;
Of formula (3) is 6,8,3 ', 5'-tetrahydroxy-flavone dihydro; of formula (4) is 5,7,8,3', 4'-serotonin Chalcone group;
Formula (5) of 7,8,3 ', 4'-tetrahydroxy flavone; formula (6) is isoliquiritigenin;
Formula (7) is glycyrrhizin.
Vernonia flavonoid ingredients and preparation methods and uses of the present invention, in order to To investigate the efficacy of the material basis of Vernonia treatment of vitiligo, looking for the active ingredient of the present invention Related experiments using pharmacological methods in vivo pharmacodynamics evaluation from Vernoniaanthelmintica Flavonoids efficacy of the components chrysanthemum isolated, indicating Vernonia flavonoid components are used Aspects of the treatment of vitiligo drug use.
Vernonia flavonoid components and in the manufacture of pharmaceutical treatment of vitiligo according to the present invention Application, experiments show Vernonia each flavonoid components alone or mixed use, Can be melanocyte cell proliferation, increased tyrosinase activity, increased melanin content, promote black Prostaglandin synthesis; vitiligo animal model of skin melanoma cells in the basal layer, the base layer containing black pigment particles Number of neutrophils increased significantly, tyrosinase-related protein expression increased.
detailed description
The following specific embodiments of the present invention will be described in detail:
Example 1
a, the Vernonia seeds 5kg, crushed to 20 mesh, to mention a 6-fold amount of water Take 3 times, the temperature of 100 ° C, 3 hours extraction time, the combined extracts were recovered solvent, Concentrated and dried under vacuum to obtain dry extract to give a total crude extract;
b, total crude extract with a concentration of 30% aqueous ethanol to dissolve sufficiently, and extracted with petroleum ether Take 2 times the amount of solvent extraction and solution volume ratio of approximately 1:4, the extract was concentrated to give a dry dip paste;
c, chloroform - methanol as eluent, separated by polyamide eluting six times Column volumes were collected eluent eluent was concentrated under reduced pressure and dried to obtain Vernoniaanthelmintica
Chrysanthemum flavonoid components on a dry weight basis containing each product flavonoid content of 50.8%, riveting purple pigment content 20%, purple flower riveting pigment content of 40.5% 7,8,3 ', 4'-tetrahydroxy flavonoid content 68.5%, 5,7,8,3 ', 4'-hydroxy chalcone five content of 55.4%, 6,8,3', 5'- Tetrahydroxy flavanone 55.6% glycyrrhizin content of 80.4%, ISL content of 99.9%.
Example 2
a, the Vernonia seeds 5kg, crushed to 60 mesh, with 10 times the amount of 20% ethanol Aqueous extracted once, the temperature of 80 ° C, the extraction time is 3 hours, the combined extracts were recovered The solvent was concentrated and dried under vacuum to obtain dry extract to give a total crude extract;
b, total crude extract with an aqueous solution of sufficiently dissolved, extracted 7 times with chloroform, each extraction Take the amount of solvent and the solution volume ratio of approximately 1:1, the extract was concentrated to obtain dry extract;
c, 40% ethanol as the eluent, were isolated and purified using chromatography on silica gel, Eluted with 10 column volumes were collected eluent eluent was concentrated under reduced pressure, drying, Vernonia flavonoids have the ingredients on a dry weight basis product containing 60.2% each of the total content of flavonoids, Purple pigment content of 67.3% riveting, riveting purple flower pigment content of 62.5%, 7,8,3 ', 4'-tetrahydroxy Flavonoid content of 70.4%, 5,7,8,3 ', 4'-five hydroxy chalcone content of 60.8%, 6,8, 3 ', 5'-tetrahydroxy flavanone 70.0%, glycyrrhizin content of 99.4%, including ISL An amount of 80.2%.
Example 3
a, the Vernonia seeds 5kg, crushed to 5 mesh, with 6 times the amount of 40% ethanol water Solution was extracted twice, a temperature of 60 ° C, the extraction time of 1 hour each time, the combined extracts were back Received solvent, concentration, vacuum drying, dry extract to give a total crude extract;
b, total crude extract with 70% aqueous ethanol sufficiently dissolved, extracted with ethyl acetate 5 Times the amount of solvent extraction and solution volume ratio of approximately 1:3, the extract was concentrated to obtain dry extract;
c, 90% aqueous ethanol as the eluent, were isolated and purified using sephadex, 25 column volumes of the elution, the eluate were collected, the eluate was concentrated under reduced pressure, and dried, i.e., Vernonia flavonoids have the ingredients on a dry weight basis product containing 84.4% each of the total content of flavonoids, Purple pigment content of 88.8% riveting, riveting purple flower pigment content of 30.5%, 7,8,3 ', 4'-tetrahydroxy Flavonoid content of 40.4%, 5,7,8,3 ', 4'-five hydroxy chalcone content of 70.7%, 6,8, 3 ', 5'-tetrahydroxy flavanone 82.9.0% glycyrrhizin content of 90.2%, ISL Content of 90.4%.
Example 4
a, the Vernonia seeds 5kg, crushed to 80 mesh, with 12 times the amount of 60% ethanol Aqueous solution was extracted three times, the temperature of 60 ° C, 2 hours extraction time, the combined extracts were Recovered solvent, concentration, vacuum drying, dry extract to give a total crude extract;
b, total crude extract with 70% aqueous ethanol to dissolve sufficiently, and extracted with petroleum ether 5 Times the amount of solvent extraction and solution volume ratio of approximately 1:3, the extract
was concentrated to obtain dry extract;
c, 60% ethanol as the eluent, were isolated and purified using chromatography on silica gel, Eluted with 30 column volumes eluate The eluate was concentrated under reduced pressure, drying, or flooding Insect Vernonia flavonoid components on a dry weight basis product each containing flavonoid content of 90.0%, purple riveting Pigment content of 99.9%, riveting purple flower pigment content of 80.4%, 7,8,3 ', 4'-tetrahydroxy flavone Content of 90.4%, 5,7,8,3 ', 4'-hydroxy chalcone five content of 70.7%, 6,8, 3 ', 5'-tetrahydroxy flavanone 90.9% glycyrrhizin content of 89.4%, including ISL An amount of 30.4%.
Example 5
a, the Vernonia seeds 5kg, crushed to 40 mesh, with 8 times the amount of water extracted 4 Time, temperature 80 ° C, 3 hours extraction time, the combined extracts were recovered solvent, concentrated Shrink, vacuum drying, dry extract to give a total crude extract;
b, the total crude extract with high-speed countercurrent chromatography separation by conventional methods, you can get To Vernonia flavonoid components on a dry weight basis containing each product flavonoid content of 99.9%, and purple Riveting pigment content of 75.4%, riveting purple flower pigment content of 99.9%, 7,8,3 ', 4'-tetrahydroxy yellow Ketone content 95.2%, 5,7,8,3 ', 4'-hydroxy chalcone five content of 88.7%, 6,8, 3 ', 5'-tetrahydroxy flavanone 99.9%, glycyrrhizin content of 70.4%, including ISL An amount of 50.2%.
Example 6
a, will 5kg, Vernonia seeds crushed to 80 mesh, with a concentration of 8 times the amount of 95% Aqueous ethanol extract 3 times, temperature 80 ° C, 3 hours extraction time, and the combined extracts Liquid, solvent recovery, concentration, vacuum drying, dry extract to give a total crude extract;
b, then the total crude extract with a concentration of 60% aqueous ethanol sufficiently dissolved with chloroform Extraction 6 times the amount of solvent extraction and solution volume ratio of approximately 1:2 extracts were dried and concentrated to give extract;
c, 90% aqueous ethanol as the eluent, were isolated and purified using sephadex, Eluted with 15 column volumes were collected eluent eluent was concentrated under reduced pressure, drying, Vernonia flavonoids have the ingredients on a dry weight basis product containing 80.0% each of the total content of flavonoids, Riveting purple pigment content 50.2%, riveting purple flower pigment content of 80.4%, 7,8,3 ', 4'-tetrahydroxy Flavonoid content of 99.9%, 5,7,8,3 ', 4'-hydroxy chalcone five content of 99.9%, 6, 8,3 ', 5'-tetrahydroxy flavanone 70.2%, 30.4% glycyrrhizin content, ISL Content of 66.3%.
Example 7
a, the Vernonia seeds 5kg, crushed to 60 mesh, with 10 times the amount of concentration 60% aqueous ethanol extract 3 times, temperature 70 ° C, extraction time one hour, to mention merged Take liquid solvent is recovered, concentrated and dried under vacuum to obtain dry extract to give a total crude extract;
b, total crude extract with an aqueous solution of sufficiently dissolved, extracted with ethyl acetate three times, each Times the amount of solvent extraction and solution volume ratio of approximately 1:1, the extract was concentrated to obtain dry extract;
c, chloroform - methanol solution as the eluent, were isolated and purified using sephadex, Eluted with 20 column volumes were collected eluent eluent was concentrated under reduced pressure, drying, Vernonia flavonoids have the ingredients on a dry weight basis product containing 90.4% each of the total content of flavonoids, Riveting purple pigment content 92.2%, riveting purple flower pigment content of 92.3%, 7,8,3 ', 4'-tetrahydroxy Flavonoid content of 78.7%, 5,7,8,3 ', 4'-five hydroxy chalcone content of 56.8%, 6, 8,3 ', 5'-tetrahydroxy flavanone 54.2%, 99.9% glycyrrhizin content, ISL Content of 90.5%…
CN102442987
Fisetin extraction method
Fisetin extraction method
Inventor(s): FAQING LI; CHENGDONG YANG; DONGFENG LIU +
The invention relates to a fisetin extraction method. The process method comprises the following steps: crushing dried branches and leaves of smoke trees; adding 10-20 times of saturated limewater solution, and soaking and extracting at normal temperature; filtering the extracting solution, regulating the pH value to 7, and adding a macroporous resin column for adsorption; eluting with a 60-70% ethanol solution; recovering ethanol in the eluent while depressurizing, standing for crystallization, and filtering out coarse crystals; dissolving in a 5% sodium carbonate solution; sequentially leaching with n-butyl alcohol and ethyl acetate; removing the organic phase, regulating the water phase with hydrochloric acid until the pH value is 4-5, and standing for precipitation; and recrystallizing the precipitate, and drying to obtain the product. The fisetin extraction method has the advantages of easy acquisition of raw materials, simple process operation and low production cost, and is applicable to industrial production.
FIELD:
The present invention belongs to the field of natural product separation technology, and more particularly to a method for extracting paint flavin.
Background technique:
Joseph also known as non-prime paint Flavin, acid yellow paint, purple riveting prime fisetin.
Molecular formula: C15H10O6.
Properties: yellow needle crystallization (crystallized from dilute ethanol), decomposition point 330 ° C, soluble in ethanol, acetone, ethyl Acid, alkali hydroxide solution, insoluble in water, ether, benzene, chloroform and petroleum ether. In a dilute solution of sodium hydroxide in ethanol Showing dark green fluorescence.
Fisetin inhibit prostaglandin and protease, but also can spasm, against its acetylcholine-induced spasm The effectiveness of the role of papaverine was 166%. Treatment of rheumatoid joint pain, diarrhea, stomach pain, colic, bloating, pain, bruises Injury, carbuncle sore throat. In addition, Japan, US researchers found that experimental, paint flavin help improve memory. therefore Fisetin can do drugs and health products, broad market prospects.
Sumac is Anacardiaceae sumac species, herbs sumac contains arbutus pigment, paint Flavin, iso-quercetin, kaempferol, There are a lot of tannin and triterpenoids.
Existing methods to extract paint flavin generally used conventional extraction methods of flavonoids, mostly organic extraction reagents, alkaline Water extraction, hot water extraction, crystallization purification process is more or column separation. Due to the different nature of the flavonoids, extraction and purification The method is also different, the same plants flavonoids have different extraction methods are different. Accordingly there is provided a fisetin The extraction method is also necessary.
SUMMARY:
The present invention to solve the technical problem is to provide a simple process and easy-to-paint flavin extraction method.
The object of the present invention is achieved by the following technical scheme:
Extraction method paint flavin, characterized by comprising the steps of: raw material crushing, adding 10-20 times the amount of Saturated lime solution soak 2-3 times extracted extract was filtered after adjusting pH7 added macroporous resin column absorption, adsorption knot Toe, washing sugar color, then 4-6BV60-70% ethanol eluate vacuum recovery of ethanol placed Junction Grain, crude crystals were filtered off, 5% sodium carbonate solution was dissolved and washed successively with n-butanol, ethyl acetate 2-3 times, go organic Phase, the aqueous phase was adjusted to HCl pH4-5 placed precipitate, the precipitate was filtered off and dissolved in 50-100% alcohol solution under reflux, adjusting pH3-5 crystalline 2-3 times, drying derived products.
Said step of raw materials for the Anacardiaceae sumac plant dry leaves.
The step-type non-polar macroporous resin macroporous resin. Preferably HZ816 type macroporous resin.
To sum up the advantages of the present invention are:
1) readily available raw materials. In the city, there is every year a large number of sumac and dried leaves are processed as municipal waste, consume large The amount of manpower and resources. The present invention utilizes sumac leaves as raw material, can change waste into treasure, value creation, addition, you can put The residue was extracted after the supply of the paper mill.
2) The present invention is immersed in water with stirring limestone extraction, macroporous resin, and then extract, reducing energy consumption to be reduced Less extraction reagent consumption, thereby reducing costs.
It will be further described in connection with specific embodiments of the present invention, but the scope of the claimed invention is not limited to the following Instance.
detailed description:
Example 1:
Dried sumac branches to the complex, crushing, taking 5kg, input from cans, 100kg was added a saturated aqueous solution of lime stirred Soak for 3 hours, filtration of liquid, together with 100kg extracted once with saturated lime solution, adjusted with hydrochloric acid extract was filtered pH7, added 5LHZ816 macroporous resin adsorption column, flow rate 20kg / h, after the end of adsorption, washing colored sugar, and then 20L70% ethanol eluate vacuum recovery of ethanol and left to crystallize, and filtered to give crude crystals were 200g, with 5% After dissolving sodium carbonate solution, washed with n-butanol, ethyl acetate, each time adding two volumes of organic reagent was extracted 2 Times, the organic phase, the aqueous phase was adjusted to pH4 hydrochloride placed precipitate, the
precipitate was filtered off and washed with 50% ethanol was refluxed for dissolution After adjusting pH3, crystallized, crystallization twice and dried to give crystals Fisetin product 38g, content of 94.1%.
Example 2:
Dried sumac branches to the complex, crushing, taking 5kg, input from cans, lime was added a saturated aqueous solution was stirred 50kg 2 hours of soaking, liquid filtration, together with 50kg extracted twice with saturated aqueous solution of lime, sulfuric acid extract was filtered adjusted pH7, 7L join the D101 macroporous resin adsorption column, flow rate 15kg / h, after the end of adsorption, washing colored sugar, and then 42L60% ethanol eluate vacuum recovery of ethanol and left to crystallize, and filtered to give crude crystals were 192g, with 5% After dissolving sodium carbonate solution, with 3 volumes of n-butanol extracted twice, the organic phase, and then 2 times 3 times the amount of ethyl acetate, Hydrochloric acid aqueous phase adjusted to pH5 placed precipitate, the precipitate was filtered off and the solution was refluxed for dissolution, pH5 using hydrochloric acid in ethanol, Place crystals were twice crystallized product was dried to give Fisetin 32g, the content was 92.3%.
Example 3:
Dried sumac branches to the complex, crushing, taking 5kg, input from cans, lime was added a saturated aqueous solution was stirred 70kg Soak for 3 hours, filtration of liquid, plus 100kg saturated lime solution was extracted twice, sulfuric acid extract was filtered adjusted pH7, added 5LHZ816 macroporous resin adsorption column, flow rate 16kg / h, after the end of adsorption, washing colored sugar, and then 25L60% ethanol eluate vacuum recovery of ethanol and left to crystallize, and filtered to give crude crystals were 185g, with 5% After dissolving sodium carbonate solution, washed with n-butanol, ethyl acetate, each time adding two volumes of organic reagent, and extracted three Times, the organic phase, the aqueous phase was adjusted to pH5 hydrochloric placed precipitate, the precipitate was filtered off and washed with 70% ethanol was refluxed for dissolution After adjusting pH5, crystallized, crystallization twice and dried to give crystals Fisetin product 35g, content of 98.2%.
Example 4:
Dried sumac branches to the complex, crushing, take 10kg, investment from cans, 100kg added a saturated aqueous solution of lime mix Hydrochloric stirred for 2 hours of soaking liquid filtration, together with 100kg of lime saturated aqueous solution was extracted twice, the filtered extract Section pH7, added 10LHZ816 macroporous resin adsorption column, flow rate 35kg / h, after the end of adsorption, washing colored sugar, and then With 50L65% ethanol eluate crystallized decompression recovery of ethanol, and filtered to give crude crystals were 392g, with After dissolving 5% sodium carbonate solution, washed with n-butanol, ethyl acetate, each time adding two volumes of organic reagents, extraction Take 3 times, the organic phase, the aqueous phase was adjusted to pH4 hydrochloride placed precipitate, the precipitate was filtered off and the solution was refluxed with 90% ethanol After dissolution, adjust pH4, crystallized, crystallization three times, and dried to give crystals Fisetin product 68g, content of 98.1%.
Example 5:
Dried sumac branches to the complex, crushing, take 20kg, investment from cans, 300kg added a saturated aqueous solution of lime mix Hydrochloric stirred for 3 hours soaking, liquid filtration, together with 300kg of lime saturated aqueous extracted once, filtered extract Section pH7, added 20LHZ816 macroporous resin adsorption column, flow rate 65kg / h, after the end of adsorption, washing colored sugar, and then With 100L70% ethanol eluate vacuum recovery of ethanol crystallized, and filtered to give crude crystals 765g, Dissolved with 5% sodium carbonate solution, washed with n-butanol, ethyl acetate was added per 1 volume of organic reagent, Was extracted 3 times, the organic phase, the aqueous phase was adjusted to pH5 hydrochloride precipitate is placed, the precipitate was filtered off and washed with 70% ethanol solution was allowed After the dissolution of the flow, adjust pH3, placed crystals were twice crystals are dried to give paint flavin products 142g, 98% content.
KR20080041169
PREPARATION METHOD OF EXTRACT, DO NOT CAUSING ALLERGY FROM BARK, DRIED LACQUER AND HEARTWOOD OF RHUS VERNICIFLUA AND COMPOSITION CONTAINING THE SAME
PREPARATION METHOD OF EXTRACT, DO NOT CAUSING ALLERGY FROM BARK, DRIED LACQUER AND HEARTWOOD OF RHUS VERNICIFLUA AND COMPOSITION CONTAINING THE SAME
Inventor(s): PARK SANG JAE, et al.
A method for preparing a Rhus verniciflua derived extract is provided to obtain the extract, which does not causing allergy and shows anti-cancer, pain alleviating and cancer preventing effects, by treating the extract with an alkaline water. A method for preparing a Rhus verniciflua derived extract comprises the steps of: (a) mixing Rhus verniciflua, Rhus verniciflua barks and Lacca Sinica Exsiccata in a weight ratio of 1:0.05-0.3:0.05-0.2 and pulverizing the mixture; (b) after adding water 5-20 times of the weight of the powder obtained from the step(a) to the powder, extracting and filtering it to obtain a water extract; (c) after adding ethanol 5-10 times of the weight of the remnant after the step(b) to the remnant, extracting it to obtain an ethanol extract; (d) concentrating the extracts obtained from the steps(b) and (c) to have a solid content concentration of 20-50 weight/volume%; and (e) after adding an alkaline water having a pH of 8.0-9.5 to the concentrate obtained from the step(d) to have a solid content concentration of 5-40 weight/volume%, adding gas containing oxygen thereto with mixing. A Rhus verniciflua derived extract obtained by the method comprises 5-26 wt.% of fustin, 1-10 wt.% of fisetin, 0.1-10 wt.% of 3,4-dihydroxybenzoic acid and 15-55 wt.% of polyphenol and does not include urushiol, butein, and butin. A composition for alleviating pain alleviating, anti-cancer or preventing cancer comprises the extract as an effective ingredient.
Chilpi, geonchil, does not cause an allergic chilmok derived extracts and pharmaceutical compositions that contain them {Preparation method of extract, do not causing allergy from bark, dried lacquer and heartwood of Rhus verniciflua and composition containing the same}
The present invention relates to a composition and method having the anti-cancer, pain, cancer preventive effect of the extract derived from sumac.
Sumac holds a large amount of antioxidants. Giminwon etc., Korean J. Food Sci. Technol. 31 (3), 855-63 from the lacquer tree bark extract from the (1999) were reported the isolation of antioxidative active substances, and the like imgyetaek Korean J. Food Sci. Technol. 29, 1248-54 (1997) was applied to the results reported for rat brain cells strong antioxidant activity of ethanol extract from poison ivy, etc. jeonghyeongjin Korean J. Plant. Res. 14 (3), were reported and antioxidant fraction from the current method of lacquer 220-8 (2001), J.-C. Lee et al and is a silica column using a sumac extract in Food and Chemical Technology 42, 1383-88 (2004) reported that the purified fractions have the ability to inhibit the growth of human cancer cells, blood, etc. Food Sci yijeongchae . Biotechnol. 9 (3), was reported to have a fraction always angry, antimicrobial effects of ethanol extract from Rhus 139-45 (2000). Choewonsik et al. J. Korean Soc. Agric. Chem. Biotechnol. 45 (3), at 168-72 (2002) were reported the antioxidant effect of a purified component fraction of methanol extract was reported not high cytotoxic activity against tumor cells results. Imgyetaek is Agric. Chem. Biotechnol. 45 (4), 173-9 (2002) was compared to the water in the lacquer effective in preventing cell death (apoptosis) in the liver cells of the extract and ethanol extract, report, if such power is herbal Society 3494), 339-43 (2003 ) extract from lacquer tree bark with methanol, which was reported to obesity inhibitory effect on fat fed rat. And the Republic of Korea Patent Application No. 10-1997-0013163, reported the anticancer composition containing urushiol components in 10-1997-0004193, and Fu Destin, avoiding the poison ivy extract 10-0257448-0000 patents in the Republic of Korea as a refined composition with a silica column paroxetine, the retinoic seolpu, and presents the anticancer composition containing an octane unit, the urushiol in the Republic of Korea Patent 10-0251526-0000 and presents the anticancer agent composition containing as a main component, in the Republic of Korea Patent Application No. 10-2002-0018186 sumac the ethanol extract and presents an extract having an antioxidant effect and anti-apoptotic effect by the method of low-temperature value, in the Republic of Korea Patent Application No. 10-2002-0071464 suggests a liver disease treatment using a sumac extract.
In the Republic of Korea Patent Application No. 10-2000-0004700 it suggests an anti-cancer effect of the medicine due to light aging method and suggests the use of anticancer drugs using the extract containing lacquer. For these applications as a water extract of the reference suggests only the content of the sumac and poison ivy extract is simple itjineun mentions the difference between the raw material composition. Sumac anacardiaceae deciduous hardwood neck sap belonging to the (Anacardiaceae) is being used for industrial and medicinal and as poison ivy or geonchil, especially being used in herbal and private as medicine, etc. judok, fever, anthelmintic, malaria, stomach pain, tonggyeong, constipation, as otdal It has been used as a food (gimtaejeong (1996) plant resources of South Korea II, 294, Seoul National University Press). Research on the physiological activity on the sumac extract for antioxidant activity was reported in the antimicrobial component urushiol, a flavonoid components and fluids (Journal of the Korea Food. 31: 855-63; Journal of Pharmacognosy 31: 345-50, and reported a), the cytotoxic effects on cancer cells of the urushiol component Arch. Pharm. Res. 22: 638-41 reported in (2000). Sumac, so results from the allergy example, even though a variety of bioactive been restrictions on their use. Many of the components that cause the allergic to urushiol lacquer has been identified by studying a variety of methods for removing them. Among them there is a method for the heat treatment, extraction with a solvent, and method for low-temperature value, method of treatment using oxygen and the like. It has an analgesic effect in natural products derived extracts, but a number have been reported, most of which were filled not characterized is limited to those only be perceived as a mere extract composition, but much research Angelica derived dikeo when playing with materials reported were reported during that , composition of the extract derived from sumac and Examples, but the analgesic effect of the proposed sumac extract extracted using an organic solvent, in Korea Patent Publication 10-2005-0047339, using the xylem of the lacquer as a raw material in its manufacturing method, as the organic solvent acetone , ethanol, and the present invention in that it uses, such as methanol, and presents a process for producing by using the column chromatography sees that there is a sharp difference in the production method and composition.
In Korea Patent No. 0.51953 million call, but to remove the allergens in the state added with oxygen, and presents a method for extracting and processing the alumina, which is contained in the If is the urushiol is polymerized allergens to polymers are insoluble in water extract it is assumed that it is not. The research and patents related to how far the poison ivy to remove the urushiol components of the anti-cancer activity and allergies that cause allergies, it was mostly about the physiological activity of the extract. The present invention relates to a composition having an analgesic effect and anti-cancer effects using the extract of the medicinal herbs derived from sumac in its effectiveness and the composition has a remarkable difference with the previous invention or report. Most with lacquer of patents extract the lacquer use of the extract with a solvent including water, and purified by the method of the columns and the like, as its main component urushiol, Fu Destin, blood paroxetine, seolpu Bulletin, part octane, suggests that containing the cafe, such as acid, gallic acid. This extract has a composition that is non-toxic lacquer derived from medicinal plants screen three kinds of allergens in an extract extracted by the raw material, and containing 3,4-didroxybenzoic acid and analgesic effect and anti-cancer effect of the present invention is characterized in that in that to have a manufacturing method is different from the composition and utility completely.
Sumac yeoteuna is a major purpose of mainly grown or come within a scratch on the skin in a state of wildlife in purpose or funeral culture to get chilaek of wood to get the chilaek, while licensed recently otdal research on the development of its purpose It has been in progress. Sumac is true that the extract and chilaek, is sensitive to a twig, so even just to reach the leaves cause side effects poison flowing decisive year for use as food or pharmaceutical drugs in general extract. In addition, maximizing the anti-cancer efficacy is based on the antioxidant effect by the unique composition of the already extract of the known functionality and the development of a production process development and the composition of the new lacquer derived extract was necessary in order to provide a composition having an analgesic effect, for which in the raw material were to maximize efficacy by mixing chilpi, chilmok, geonchil a suitable ratio, in the allergen was to use a process for treatment by the method of the alkaline water to neutralize the allergens. The extract does not induce a major aspect of allergies of the invention produced was fully solved by the alkaline treatment step, in the delivery of functional compositions adjusting the composition of raw materials as appropriate, and unique having a composition by developing the extraction process cancer, pain, developed a composition having cancer preventive effects. The compositions of the present invention may help to solve the cancer, health problems caused by the pain, the most even badly flowing chilaek poison come extract as compared to using only the xylem of the lacquer in order to avoid the flowing problems come a poison in conventional invention to uses in the production of it was able to provide superior function composition. Analgesic for functionality proposed in the present invention show a very superior functionality compared to the known so far, which extract may be further developed as new drugs or the like.
Sumac chilaek for producing lacquer, and its bark is used as a medicinal herb called by chilpi. Chilaek is also used as a herbal medicine by the name of geonchil dried. According to the xylem of Rhus remove the chilpi Donguibogam, light trapping, etc. hanuiseo paper feeding is known to be used for symptoms of edema, ongjeo or excuse, such as by saying chilmok. Chilmok is used less extremely in the extract content that corresponds to the xylem of Rhus, Fu Destin, avoid paroxetine and which contains the same flavonoids components allergen otdal content of the ingredients, urushiol is to remove what poison rising ingredient extremely down, etc. It is. If you remove the bark of the lacquer can be removed in most cases the poison rising temper. However, that is chilpi chilaek (geonchil) corresponding to the shell portion of the lacquer has to produce chilaek mothajiman michichi, it contains a number of urushiol poison can easily climb. Chilpi are contained resin component and a small amount of a flavonoid ingredients. Geonchil has been obtained from the lacquer to dry chilaek, chilaek has contained a large amount of ingredients, allergens and urushiol should exercise careful even in those taking. Geonchil also has a large number of polyphenols contained a large amount, and can contain a large amount also as a polymer resin of urushiol. The present invention is configured as follows. First, the raw material preparation step in the present invention to use the raw material is a composition to have most existing patents in the described lacquer or a lacquer of xylem Unlike chilmok, chilpi, geonchil three kinds of medicinal plants a dry weight ratio of 1: 0.05 to 0.3: 0.05 to 0.2 consists in mixing ratio of. Geonchil will be used instead of the geonchil chilaek the chilaek be dried and so that the water content of chilaek 30 ~ 70% (w / w) is approximately the case with the addition of excess amount in view of this. The second and extracted with added 5 ~ 20 liters of water with respect to the raw material the entire 1Kg as an extraction solvent for making these extracts mixing the raw material and granulation to dry powder, and, the temperature range to 50 ~ 110 ° C for 2 to 24 sigan and filtered to obtain a water extract, it was added 5 ~ 10 liters of ethanol 95% (v / v) with respect to the first raw material 1Kg the remaining extract foil, followed by extraction for 2 to 10 hours at 60 ~ 80 ° C obtaining the ethanol extract, , it is the step that allows mixing with the water extract, and removing the insoluble components by microfiltration with a filter of one micron, and was concentrated and the resulting liquid is the concentration of the solid content of 20 ~ 50% (w / v), fourth, here was added to an alkaline water of pH 8.0 ~ 9.5 the concentration of the solid content of 5 ~ 40% (w / v) is such that, air, pure oxygen or while adding 1 to 100 times per hour that the gas mixture in a volume ratio at 25 ~ 110 ° C 0.5 to 10 by treatment with mixing for a time by polymerizing the urushiol to cause allergic remove allergens characteristics, and is concentrated and dried to obtain a lacquer composed of the extract obtained in powder form.
The extract produced by the method of the present invention is based on the solid content of the composition of the component Fu Destin 5 ~ 20% (w / w), blood paroxetine 1 ~ 10% (w / w), 3,4-dihydroxybenzoic acid 0.1 ~ 10% (w / w), indicated as an effective component in the other sumac extract the far portion as a composition characterized in that the polyphenol content of 15 ~ 55% (w / v) in heptane (butein), butyne ( butin) is does not contain the composition, and strong analgesic effect, anti-cancer effects, to have antioxidant properties and cancer preventive effect by their characteristics. Hereinafter, a configuration of the invention in detail, the first stage stage of the raw material composition is different from what's described in the existing most of the patents in the raw material is used the composition. The present invention is a chilmok, chilpi, medicinal plants of the species geonchil 3 weight ratio of dry materials in the 1: and in that characterized in that the mixing ratio of 0.05 to 0.2: 0.05 to 0.3. Chilmok is high in flavonoid components as saying xylem of Rhus content, chilpi is high, such as small amounts of flavonoids ingredients and polyphenols, resins to say the outer bark of the sumac content, geonchil will pay a wound on the surface of the lacquer here dried sap that flows out of chilaek and contains a large amount of the resin component of the allergen urushiol poison oak and poison ivy to climb. Therefore geonchil may be very likely to cause allergies when taking the extract, if not completely removes allergens factors. Chilpi geonchil and is used as a medicinal herb eohyeol, abdominal pain, Jinhae, anthelmintic, etc. it can be used as a treatment to prevent poison ivy climbing a relationship that contains urushiol. In the configuration of the raw material drying chilpi based on the dry weight 1 of chilmok 0.05 to 0.3, is used as geonchil 0.05 to 0.2, and decreases the content of the flavonoid components of the extract The chilpi and high content of geonchil ratio of the composition to maximize the pharmacological ingredient , the content of urushiol in the extraction process that results in allergy can take a long time to completely remove high. Thus completely remove urushiol, and will determine the ratio of raw material to extract the pharmacological component having a configuration of the extract of the present invention as appropriate.
However, when the manufacturing method of the present invention may be used prior geonchil chilaek of drying as the case it is. In this case, because the water content of the normal chilaek 30 ~ 70% (w / w) in view of this need to increase the amount. Raw material is dried and then pulverized in the xylem and chilmok chilpi, in each geonchil below 60 ° C to prepare the dinner table with a saw and the size of 2mm or less, mixed in proportion. There is a less than 60 ° C in the drying is because the activity of the physiologically active substances of various enzymes contained in the raw material disappeared when dry of at least 60 ° C higher temperature. When used instead of the geonchil chilaek it can be prepared by the method of drying by grinding a chilpi, chilmok and mixed them. The second stage of the water extraction step by addition of water as extracting the aqueous extract 5-20 of the powdered raw material in the raw material composition by milling the total weight multiple step, and a step of obtaining the extract. The particle size of the raw material has been able to maximize the extraction yield in the case of less varies depending on the system that does not support the composition of the crushed 2mm extract depending on the size of the particles. Water is added to 5-20 gives a multiple of the weight of raw materials, five submerged enough to prevent a large volume of material related to the case when multiples are less than a multiple of 20 is to be put to the economical degradation of excess solvent. Heating the extract to the extraction for 2 to 24 hours at a temperature 60 ~ 110 ° C to obtain a water extract. Reason why the temperature to 60 ~ 110 ° C can be increased, so as to effect the extraction for improving the extraction effect of various pharmacological components including flavonoids solubility in water to extract the lower component at a high temperature. The extraction time was 24 hours or more to extract from 2 to 24 hours is no more than one extraction yield, they are extracted with less than 2 hours which results in significantly reduced yield of the extraction. The third step is to get the water extract as an ethanol extraction step, 95% of the remaining extract foil (v / v) was added and the first 5 to 10 multiple of the weight of the raw material of ethanol, followed by extraction for 2 to 10 hours at 60 ~ 80 ° C the ethanol extract is obtained.
Ethanol extraction process can allow the primary water because the water extraction step is thereby swell the wood car 2 By applying the ethanol extraction process increases the extraction yield to ethanol is far easily penetrate the inside of the raw material. On the other hand, in the case of ethanol as the extraction solvent of a hydrophilic material such as wood when used alone on the dry raw material it is hydrophobic it results in the extraction of components in the deep part of the xylem which is delayed or significantly reduced. Therefore, in the present invention preferentially extract to obtain a water extract as water, in which the swollen material, and then adding ethanol to here found a way to achieve even finer component extracted in the interior material. As the water after extraction remaining extraction foil already has a water content because it is more than 50% To the mixture was added more than 95% (v / v) ethanol give dilute the concentration of ethanol as appropriate can improve the extraction yield, also the extraction of hydrophobic substance It would increase. Extraction conditions was defined conditions to maximize the extraction yield from 2 to 10 hours at 60 ~ 80 ° C, the extraction yield is to be lowered when a lower temperature, or to the extraction time within 2 hours. When the extraction temperature is excessively high temperatures over 80 ° C because the boiling point of 78 ° C using ethanol leads to increased risk of sudden excessive pressure rise. Mixing the ethanol extract of water extraction liquid and is removed the insolubles by microfiltration with a filter of one micron, and consists of by concentration of the resultant extract under vacuum to have a 20-50% concentration of a solid content (w / v). The obtained concentrate has chilpi, chilmok, geonchil the allergen urushiol that is contained in (or chilaek) but some are naturally removed from the extraction procedure, most of which is possible is because the residue contained in a large amount. This perfectly solved by the following process.
Generally yieoteuna main way to eliminate the introduction of most of the allergenic removal step in the extraction process, the existing patents so far, in the present invention, only After concentrating the extract by polymerizing allergen removing its characteristics, the polymer is removed in terms of causes as contained in the extract it does not have a significant difference between the invention and the other.
This enables in that it allows access to the pharmacological effects of the polymer at the same time able to explain the superior pharmacological effects of the extract of the present invention. Fourth step is added to an alkaline water of pH 8.0 ~ 9.5 in extract solution concentrate as alkaline treatment step the concentration of the solid content and so that the 5 ~ 40% (w / v), was added 1 to 100 times per hour for air or pure oxygen in a volume ratio of extraction liquid while, by polymerizing polymerization the substance urushiol that to stir while treatment for 0.5 ~ 10 hours at 25 ~ 110 ° C causing the allergy and the content of urushiol in the concentrate to zero, the urushiol the polymerization the polymer is to remain in the concentrate It is present in polyphenols. Alkaline water used in the dilution of the concentrated solution is to use sodium hydroxide, calcium hydroxide, magnesium hydroxide, calcium hydroxide, etc. As an electrolytic water or alkaline agent manufactured by using the manufacturing method is electrolyzed as between pH 8.0 ~ 9.5 and the well is the alkaline using caustic soda. The reason for using the alkaline water here is how to increase the physiological activity of the resulting extract is presented the antioxidant, anti-cancer force, labor force and increased in this process. The reason is that many ingredients that stand out the acid, including the urushiol Among the components of flavonoids contained as a main component in the extract are the various components extracted from most of the other hand, that have a characteristic that is soluble in alkali, the extraction process of the present invention This is because the neutralization and can enhance the availability of these components. Urushiol is also polymerized but lost the characteristics already allergens as such, when the residual is to be consumed, so the final composition containing the concentrate can be obtained an increased effect of the increase in antioxidant, anti-cancer force. The reason is that it is urushiol and a phenolic structure of the urushiol in the base polymer is that it can be regarded as the polyphenols. In Korea Patent No. 0.51953 million call, but to remove the allergens in the state added with oxygen, and presents a method for extracting by treatment with alumina, which is contained in the If is the urushiol is polymerized allergens to polymers are insoluble in water extract it is assumed that it is not.
This is the portion apart from the present invention. Korea Patent No. 0.51953 million when the actual results showed a difference in favor and 7% difference between surveyed the content of phenolic components Polje. The extract of the present invention showed better results in the polyphenol content. Concentrate processing is complete allows concentrated and dried to remove the solvent consisting of a bar By obtaining a sumac extract obtained in powder form, to obtain a homogenized extract powder after dried. Extracts obtained by the present invention in the sustain Fu 5 ~ 26% (w / w) the composition as a flavonoid, quercetin blood is 1 ~ 10% (w / w), 3,4-dihydroxybenzoic acid 0.1 ~ 10% (w / w), 15 to 55% of the polyphenol content (anti-cancer efficacy and potency to inhibit pain, characterized in that w / v) which, provides a composition having an antioxidant effect, preventing cancer efficacy. Fu Destin, Phi Phi as one kinds of flavonoids quercetin is quercetin has been reported from a number of plants, but, Fu Destin is a lacquer containing large amounts specifically. Polyphenols can not be defined for the single-component, as shown in its ingredient with a substance that has been reported recently such as immunity enhancement effect, that is characterized according to the type of plant, having a wide variety of molecular structures depending on the site. Therefore, a tannin as the polyphenolic Assay was quantified according to the Republic of Korea Food process as that in the method the present invention as a standard substance. 3,4-dihydroxybenzoic acid is known to have anti-cancer activity as a kind of phenolic acids, anti-thrombotic action, and found that this has been reported in a number of plants. As causing a variation upon overdose gene it is also known. Therefore, the extract has been limited to the range of the content of the present invention 0.1 ~ 10% (w / w). Urushiol is known as a substance that causes the allergy has become toxic anger must be removed or derived from extracts of sumac should not cause any allergies. Extract of the present invention was confirmed safe from causing allergy appears to not detected in an amount of zero was analyzed by analysis of urushiol Korea Food and Drug Administration urushiol.
In addition, the sumac extract thereof in many invention butyne in a functional active substances such as anti-cancer, but to point out the portions retain, extract of the present invention is shown as a result of analyzing the content of butyne, parts retain a HPLC method not detected, the functional material it can be seen the component is not contained. Therefore, the invention also (i) the extract of the pharmacologically effective dose as an active ingredient and (ii) a pharmaceutically acceptable carrier, diluent, or excipient, or for a combination of analgesic, for that is configured to include the anti-cancer, cancer It provides a pharmaceutical composition for the prevention. Extracts according to the invention can be administered as a pharmaceutical composition mixed with other active ingredients and a suitable carrier or excipient or with, as in itself, or a combination therapy to a human patient. Or less, using an embodiment of the present invention will be described in detail. However, by the following Examples it is not limited the scope of the invention. Example 1. Preparation of extract 10 Years Old lacquer xylem (chilmok) 2Kg, sumac crusted (chilpi) 300g, dried geonchil 100g for each 50 ° C oven, it chilmok: and mixed in a ratio of 0.1: chilpi: geonchil to a weight ratio of 1: 0.15 to obtain a raw material it was pulverized using a grinder 1.2Kg. Was added to 10L of water to 1Kg of warmed to extract at 95 ° C for 5 hours to obtain 8.8L water extract, was added 8L of 95% ethanol concentration again on the remaining extract foil (v / v), at 75 ° C extraction for 5 hours to give 7.9L of ethanol extract. This is mixed with the water extract was filtered with a 1 micron filter, and concentrate the resulting extract. The volume of the concentrated liquid was a result of measuring the solid content to step 31Bx refractive index when a 200ml. It was added thereto by using a caustic soda of 1 normal concentration 100ml an alkaline tailored to pH 8.5, while stirring well for 5 hours to air at 88 ° C while bubbling at a rate of 3L / hr ring (bubbling) treatment, concentration and , dried to give 58g as a light brown dry powder. Analysis Example 1. To 10% (w / v) in order to analyze the functional component of the analysis sumac extract main components dissolved in water and fine-filtered by a 1 micrometer filter paper, and a volume ratio here 1: vigorously was added chloroform to 1, and mixed layer separation to obtain a chloroform layer of the bottom, mixed vigorously and then separated to remove the layer of ethyl acetate was added to the top of the water layer with ethyl acetate again to obtain an ethyl acetate fraction.
It was to concentrate the ethyl acetate fraction was dissolved in methanol so that the 10% (w / v) solid content, and then added to a column of silica in silica-filled it 70-230mesh, chloroform: methanol: water = 70: 21: 9 ratio of was to remove the lower layer of the mobile phase made of deployment as the mobile phase to get the five fractions, in these two fractions of the reverse phase silica (C18) is charged column again, and methanol: water = 65: to deploy a mobile phase of 35 5 a fraction. Analysis on the Dual Fu Destin, separating the blood paroxetine, from Sigma was purified by HPLC to purchase the standard materials was confirmed. Based on this, the sumac extract as Fu targeted Destin, paroxetine is blood using HPLC, 3,4-dihydroxybenzoic acid was analyzed by gas chromatography, the polyphenols were determined using the phenol method. The composition of the extract obtained in Example 1, the result is Fu Destin is 16.4% (w / w), paroxetine blood is 2.3% (w / w), 3,4-dihydroxybenzoic acid is 1.5% (w / w), polyphenol It found to be the 35.1% (w / w). Apparently move the extract of the present invention in a manner that is dissolved in methanol, loaded on a HPLC equipped with a C18 column for HPLC analysis 5% acetic acid: acetonitrile = 25: 75 to 1ml / min with the UV detector of a result other analyzes at 260nm part Thane (Butein), butyne (Butin) components known in the study were not detected. Analysis Example 2. Allergen removal confirmation investigate the urushiol in the content that are known to induce chilaek and allergenic extracts by using HPLC / MS of the present invention obtained from the raw lacquer in order to confirm the removal of urushiol by allergens of the extract of the invention in It was. Analysis of the urushiol is Korean J. Medical Crop Sci, 10 (4):. Followed to 288-93 (2002). The results showed in Table 1 of the. Analysis of the results urushiol content of urushiol chilaek molecular weight alkali-treated concentrate the final extract of the present invention 분말3141.5%(w/w)0.3%(w/w)불검출3160.1%(w/w)0.03%(w/w)불검출3180.13%(w/w)0.1%(w /w)불검출3201.1%(w/w)0.12%(w/w)불검출위 As can be seen in Table extract of the present invention obtained through the alkali treatment step it has been found that this completely remove urushiol known to cause allergies.
Patents
: Fisetin Applications
Nutritional composition containing a neurologic component of kaempferol and/or fisetin and uses thereof
TW201532528
A PHARMACEUTICAL COMPRISING FISETIN FOR TREATING OR PREVENTING ACUTE PANCREATITIS
KR20150096898
A COMPOSITION AND A METHOD FOR IMPROVING SOLUBILITY AND BIOAVAILABILITY OF FISETIN
KR20150084535
RADIATION PROTECTIVE COMPOSITION COMPRISING EFFECTIVE FISETIN COMPOUND
KR20140120123
MEDICAL FOOD FOR COGNITIVE DECLINE.
MX2013013512
COMPOSITION FOR AMELIORATING INFLAMMATION DISEASE COMPRISING FISETIN AND SULFURETIN AS EFFECTIVE COMPONENTS
KR101431957
Dietary Supplement System for Multifunctional Anti-Aging Management and Method of Use
US8974839
MELANIN PRODUCTION PROMOTER
JP5769387
A food formula to slow the aging process
TW201004637
PROMOTER FOR SUGAR UPTAKE BY MUSCLE
JP5594719
Application of fisetin for resisting against Eimeria tenella
CN102028680
COMPOSITION FOR REDUCING SKIN PORE SIZE CONTAINING POLYPHENOL COMPOUNDS
KR20110017599
PHARMACEUTICAL COMPOSITION FOR PREVENTION OR TREATMENT OF NEURODEGENERATIVE DISEASES COMPRISING FISETIN
KR20090107607
METHODS OF TREATING ANDROGEN DEPENDENT PROSTATE CANCER BY ADMINISTERING AN ACTIVE PHARMACEUTICAL INGREDIENT BEING FISETIN...
US2010010078
Anti-aging composition containing resveratrol and method of administration
US2009163580
Compositions for Regulation of Hair Growth
US2008254055
METHODS OF USING FLAVONOIDS TO ENHANCE MEMORY
WO2008011538
3-hydroxyflavones: their preparation and therapeutic application
US4591600
Compounds for treating symptoms of common cold, allergic rhinitis and infections relating to the respiratory tract
NZ524121
PHARMACEUTICAL COMPOSITION FOR PREVENTION AND TREATMENT OF FIBROSIS OR CIRRHOSIS OF THE LIVER
KR100511720
COMPOSITION FOR PREVENTION AND TREATMENT OF LIVER CANCER, CONTAINING PHENOLIC COMPOUND
KR100388787
COMPOSITION FOR PREVENTION AND TREATMENT OF LIVER CIRRHOSIS, CONTAINING PHENOLIC COMPOUND
KR100388786
LIVER PROTECTING COMPOSITION CONTAINING RHUS VERNICIFLUA EXTRACT
KR100499877
Application of fisetin in preparation of drug with anticoagulation, anti-thrombosis and cardioprotective effects
CN103536587
COMPOSITION FOR INHIBITING HIGH GLUCOSE-INDUCED INFLAMMATORY COMPRISING FISETION OR PHARMACEUTICALLY ACCEPTABLE SALT THEREOF AS AN ACTIVE INGREDIENT
KR20130106192
Application of fisetin in preparation of drug for resisting listeria infection
CN103191099
INHIBITOR OF EPITHELIAL SODIUM CHANNEL EXPRESSION
JP2013091613
Fisetin-isonicotinamide co-crystal and a method for preparing fisetin-isonicotinamide co-crystal
PL218833
Hemietanolat fisetin-nicotinamide co-crystal and a method for preparing fisetin-nicotinamide co-crystal
PL218893
NEW COMPOSITION FOR PREVENTING AND TREATING NEURODEGENERATIVE AND BLOOD COAGULATION DISORDERS
JP2013082723
Use of fisetin for preparing medicaments for treatment of depression.
CN102973551
Neuroprotective polyphenol analogs
CN103929959
USE OF A COMBINATION OF TWO COMPOUNDS FOR THE TREATMENT AND/OR PREVENTION OF BONE DISORDERS
WO2014184484
Fisetin-caffeine cocrystal and a method for obtaining fisetin-caffeine cocrystal
PL405443
NUTRITIONAL COMPOSITION CONTAINING A NEUROLOGIC COMPONENT OF KAEMPFEROL AND/OR FISETIN AND USES THEREOF
US2014107193