rexresearch
Maynard MURRAY
Seawater Agriculture
http://www.seaagri.com
Acres USA
July 2005
Remineralizating
Soils for Optimum Elemental Balance
by David Yarrow
Remineralization of our agricultural land and garden soils with
90-plus minerals is the goal of two lifelong advocates of
sustainable organic food production, Robert Cain and David
Yarrow, who were brought together by their common interest in
the research of Maynard Murray, M.D.
As most Acres U.S.A. readers are aware, Dr. Murray was a medical
doctor and research scientist who was troubled by the obvious
continual decline of American health and the subsequently
flourishing pharmaceutical industry. He searched for reasons, on
biological and chemical levels, as to why our bodies lose their
resistance to chronic illness and develop degenerative disease.
His studies led him to the sea, where, miraculously, cancer,
arthritis, arteriosclerosis and aging on a cellular level
seemingly did not exist. He discovered that sea life is
sustained in a balanced solution consisting of all 90-plus
atomic table elements. Murray observed that a cubic foot of
seawater contains considerably more living organisms than an
equivalent amount of soil.
Murray theorized that the apparent difference in disease
resistance and vitality between life on land and in the sea is
due to mineral deficiencies in our soil and food. He visualized
an endless cycle wherein continents rise from the sea rich with
minerals. The constant effects of climate — freezing, thawing,
rainfall, and erosion — combined with mankind’s historically
poor stewardship of the land and increasing acid rain cause
topsoil minerals to go into solution. These mineral solutions
then enter streams and rivers and subsequently flow into the
sea. Murray concluded that these minerals hold the key to human
health. Therefore, it made perfect sense to recapture them and
restore them to our soils.
Initially, he successfully experimented using diluted seawater
on soils and crops. Then he discovered that if water is totally
removed from pure, mineral-enriched seawater, 3.5 percent
remains as solids. He called these minerals “sea solids” and
used them exclusively, during many years of extensive research,
on all ranges of crops and soil types. Murray even developed a
specialized use of sea solids for hydroponics, and operated a
successful 13-acre hydroponic fresh-produce farm in southern
Florida. The results were consistently the same: the plants
flourished, matured more rapidly, were healthier, were more
disease and drought resistant, and produced outstanding taste
along with greater yields. In assays testing for nutrients,
foods grown with Murray’s sea solids had significantly more
minerals (ash content), vitamins (25 percent more vitamin C in
tomatoes; 40 percent more vitamin A in carrots) and sugars. In
addition, he witnessed the same amazing results in all types of
livestock and poultry that were offered feed grown in soil
enriched by his sea solids. Physiologically, these animals were
healthier, gained weight more rapidly, and reached maturity
sooner.
During his 30 years of research, Murray conclusively proved that
the proportions of trace minerals and elements present in pure
seawater are optimum for the growth and health of both land and
sea life. Additionally, he found that once these minerals and
trace elements are restored to the soil, reapplication is not
necessary for five or more years, given normal rainfall and
climatic conditions. Cain, under Dr. Murray’s direction, applied
these minerals to both soil and hydroponic food production, and
personally tasted and witnessed their outstanding effects.
Since creating sea solids by desalinization of seawater is very
costly, Murray searched the earth for the best source of sea
solids in their natural form. He required expansive tidal flats
on the banks of a mineral-rich, unpolluted sea in an arid region
with little or no rainfall. Prior to his death in 1983, he
disclosed to Cain the location he had found to be the purest.
Only recently, Cain and Yarrow have begun mining sea solids from
Murray’s source and distributing the product throughout North
America. Cain says he believes Dr. Murray discovered the true
“Fountain of Youth.” However, the fountain with its
life-enhancing properties is located not at a springhead where a
stream or river begins, but rather at the opposite end of the
ecosystem, where it empties into the sea.
Cain and Yarrow’s vision is to improve the quality of human
health through the food we eat by remineralizing the soil in
which it is grown. In their opinion, application of these
extraordinary sea solids with their 90-plus elements — the sea’s
full spectrum of minerals — to tired and depleted soils is the
perfect solution. They believe that as stewards of the land, it
is our responsibility to restore the mineral balance to soils
and subsequently the foods we ingest. Cain and Yarrow hope to
convince all stewards of the land to help sustain life on this
planet by remineralizing their soils and spreading the wisdom of
sea energy agriculture.
For more information on sea solids and the work of Robert Cain
and David Yarrow, contact
SeaAgri Inc., 4822 Kings Down Road, Atlanta, Georgia 30338
Phone (770) 361-7003
email <seaagri@bellsouth.net>
Maynard Murray

US Patent
# 3,071,457
Process of Applying Sea Solids
as Fertilizer
Maynard R. Murray
This invention relates to the process of applying sea water
solids as a fertilizer. by sea solids we mean the inorganic
salts that are dissolved in the water; the term as it will be
used in the specification hereinafter does not include living
organizms, plant or animal, but means merely the salts that are
dissolved, which will include the salts of the various elements
mentioned in this application.
This invention relates more particularly to the use of sea
solids in certain proportions for different crop requirements.
This invnetion further contmeplates the use as a fertilizer of
complete sea solids mixed with nitrogenous compounds, the
proportions of each and the total amount of fertilizer depending
upon the type of crop which it is desired to raise and the
condition of the soil...
When one is confronted with so many variables, to obtain a plant
raised under optimum conditions seems to be almost impossible.
It was therefore decided by the author to obtain the elements
from sea water in the proportion that they occur there. The most
soluble salts found in the land should be found in the most
abundant supply in the sea. Sodium chloride is found in a much
greater concentration in the sea than are the various barium
salts. It is known that sodium chloride, per se, in great
concentrations, is toxic to plants. Therefore it was deemed
advisable to start with very low concentrations of sea water to
test their effect on plants. This was done in pot and plot
experiments, and it was found, after considerable
experimentation, that sea solids comprising 3-1/2% of sea water
could be applied to the soil in fairly great concentrations,
without detriment to the plants. The solids were obtained by
evaporating the water completely and leaving the elements in
solid salt form. The optimum
amount found for most grain and vegetable plants grown in the
temperate zone of the USA was from 550 to 2200 pounds per
acre... The salt is first ground in a burr mill before
spreading. The hydroscopic nature of the salt required that it
be stirred from time to time as it was being applied to ensure
accurate spreading.
Oats, soy beans, grain and many varieties of garden vegetables
were grown on soil thus treated. In 1954. 1/4 acre of garden
vegetables were grown; 10 acres of treated and control corn have
been grown; 10 acres of treated oats along with 10 acres of
control oats were grown; also 3 acres of treated soy beans and 3
acres of control soy beans have been grown.
In my experiments to date, I have studied, or am in the process
of studying, various phases;
First, animals were fed a diet of 4 parts corn, 2 parts oats,
and 1 part soy beans, all grown on land treated with sea solids.
This was to determine the effect of these grains on normal
physiology and pathology. The
rats fed on the control, or untreated corn, oats, and soy
beans, developed xerapthaemia in 12 to 14 days. The rats fed
on the experimental feed did not show eye changes.
300 chickens were obtained from a local hatchery when one day
old. They were divided into 2 groups of 150 each. All were fed
the commercial concentrate, plus 4 parts of corn and 2 parts of
oats. The animals fed on the
experimental corn and oats matured approximately one month in
advance of the control. The experimental started to lay eggs 3
to 4 weeks earlier, and the eggs weighed 2 to 3 ounces more
per dozen in the experimental flock. Dressed experimental
roosters at 6 months of age weighed 1-1/2 lb more than the
control, and there was less food consumed per pound by weight
gain in the experimental chickens. There was a decided
difference in the skeletal structures of the
experimental and control chickens, as shown by x-ray.
Second, productivity. In oats, there was no manifest difference
in productivity; however, during the growing stage, just before
the oats headed, there was a marked difference in color. The
experimental lot was darker green, which was noticeable to the
eye and is also readily distinguishable in colored photographs.
The farmer who harvested this crop observed that the
experimental plot had many more rabbits, suggesting a taste
difference. There was also an
observable difference in the amount of "rust" -- being much
more prevalent in the control plot. The ash weight showed a
1.1% increase in the experimental. The second generation oats
showed excellent germination and production, although no
further applications of sea solids were put on the soil.
Second generation oats were essentially "rust" free.
In corn grown in 1952, the treated
plot yielded 19.6 bushels more per acre than the control... In
1954, the experimental plot of corn yielded about 13 bushels
more per acre than the control, the experimental plot of corn
yielded about 13 bushels more per acre than the control, and
the experimental showed an increase of 1.7% in ash weight.
The control soy beans yielded 8.87 bushels more per acre than
the experimental soy beans; however, the
experimental showed an increase of 14.6% in ash weight.
Second generation soy beans
grew larger and the production was slightly higher than the
control. There was also a 5.6% increase in ash weight in the
experimental, although no further applications of sea solids
were made.
The increase in ash weight of the
experimental garden vegetables over the control was a s
follows: Sweet potatoes 8.3%; onions 4.4%; tomatoes 18.7%.
Third, diseases in plants. There was a marked difference between
the treated and control plants in "curly leaf" of peach trees, the treated tree being
mcuh freer of the disease. In blight of tomatoes, the treated
plants showed a marked difference in resistance to the
disease. The most phenomenal difference in plant diseases
noted was in corn smut, which showed 384% more smut in the
control than in the experimental. These figures are
based upon the number of observable galls counted on 4.9 acres
in each plot. Not only were
the galls much less numerous in the experimental, but they
appeared smaller, and fewer were on the ears. These same
results were repeated on the second generation corn without
further application of sea solids to the soil.
It is known that there are many acres of soil unfit for the
growth of garden peas. This is said to be due to an infection of
the root of the plant caused by Aphanomyces and Fusaria, the former being very
specific for the pea plant, and the latter having the ability to
attack other hosts. In greenhouse experiments, I was able to grow the pea plant to
maturity in soil infested with these two organisms with the
addition of sea solids, using two different varieties of peas.
The control plants died at or near the blooming stage.
"Center rot" in turnips is said to
be due to a staphylococcus infection. In 100 plants on treated
and control soil, there was an incidence of center rot in 30
of the control, and none in the experimental.
Fourth, it was also decided to test the effect of sea solids on
the pH of the soil. The ordinary garden beet was used as an
indicator plant. In acid soil, this plant is supposed to
germinate and put forth two leaves which seemingly are healthy.
The second pair of leaves, however, usually die and the plant
will not grow to maturity if the soil is too acid. I obtained
soil with a pH of 4. After the
addition of my sea solids, I found that the pH decreased
slightly, but later returned to its original value. I
planted beets and radishes in this soil treated with sea solids
and was able to grow them to maturity. I feel that so-called
sour soil is deficient, and most probably not deficient in
calcium alone; that the pH itself is not the determining factor
as to whether or not the ordinary varieties of plants found in
this climate will grow. Radishes
were grown in treated soil with a pH of 4. Beets, a sour soil
indicator, grew beyond the third and fourth leaf.
Observations -- A number of
observations made during these experiments have been recorded
for their possible significance. Sheep ignored a field of
untreated hay to get to a ten foot square patch of treated hay,
indicating a taste difference.
Also, experimental stalks of corn were marked with tape, and
mixed with control corn. Cattle and sheep would nuzzle through
the corn to pick out the experimental stalks, again indicating a
taste difference. The farmer who harvested the oats noticed
noticed that the experimental oats attracted more rabbits and
grasshoppers. A taste difference was also noted in garden
vegetables. Onions and radishes were sweeter than the
control vegetables. There was also a difference in the taste of
lettuce, green beans and carrots. In apples and grapes, vitamin A and Vitamin C were
found in greater quantity in the experimental crop. The
experimental grapes were higher in sugar content...
Summary -- The list of
elements found to be important in the normal development and
health of plants and animals has increased steadily over the
years. The problem has icnreased steadily over the years. The
problem has been made even more complex by the discovery that
the availability of an element to the plant may be dependent
upon the presence or absence of other elements in the soil. The
experiments of this report are based on three hypotheses:
1 -- That all of the elements may be important in polant and
animal physiology.
2 -- That the elements should be added to the soil in the exact
proportion and balance as they are found in sea water, including
the sodium chloride, This is based on the assumption that the
solubility of an element determines its rate of leaching from
the soil, and the amount of it found in sea water,
3 -- That most animals need to have the inorganic elements
hooked up by plants for proper utilization.
Tolerance experimetns indicated that the amount of complete sea
solids ( including sodium chloride ) that could be added to
mid-western and eastern soils ranged from 550 to 2200 pounds per
acre.
As a specific illustration of the use of sea solids as a
fertilizer, an experiment was conducted in recent months on
tomatoes, with the following results :
It will thus be noted that 1100 lb per acre of the fertilizer is
about the proper amount for the best growth of tomatoes.
Complete sea solids are obtained by drying sea water obtained
from any ocean to complete dryness. The end product contains all
elements soluble in water or saline solution, as found in sea
water...
To one ton of these sea solids, ground in a burr mill, I have
added:
80 to 800 lb of ammonium nitrate
pellets or crystals; or
100 to 1100 lb of ammonium sulfate;
or
50 to 400 lb of urea.
The range of these nitrogenous compounds is accounted for by the
fact that different crops require different amounts of N in
proportion to sea solids. Tolerance experiments with this mixed
fertilizer have indicated that from 550 to 2200 lb per acre can
be used on field crops, fruits and vegetables.
I have varied the above process in the following ways: I have
used sea water with the same proportion of elements described
above, mixed with proportional amounts of nitrogenous compounds.
Also, I have applied the sea solids to the land first, and then
applied the nitrogenous compound afterward.
As hereinbefore described, crops grown on soil fertilized with
the above-described fertilizer have been analyzed for ash
weight, vitamins and elements; and production has been noted.
The results indicate an increase in ash weight, vitamins, number
and proportion of elements, yield and resistance to plant
disease. Animals have been fed products grown on fertilized soil
with a stimulus in growth and improvement in bone and tissue
structure. Thus it can be seen that the beneficial results of
controlled use of sea solids or sea solids mixed with
nitrogenous compounds are readily apparent. From experiments
described herein, it is apparent that equally beneficial results
will be obtained by the controlled use of sea solids in the
growth of other grains, vegetables and fruits.
In this discussion I have used the range of 550 to 2200 lb per
acre as applied to midwestern or eastern soils. Also, the crop
to be raised on the land determines the amount to be used. It will be noted that 2200 lb per
acre increased the production of corn, made no difference in
production of oats, and decreased the production of soy beans.
Therefore, soy beans should have no more than 1100 lb per
acre. Garden vegetables that were outlined in the
discussion above had 2200 lb, all with the same production as
untreated vegetables.
When 550 lb are applied, I apply
that amount each year for 4 years. The amount of 2200 lb, when
applied at once, and 550 lb, when applied each year for a
period of 4 years, will last, on soil with ordinary drainage,
for a period of 5 years. I analyze the soil to see when
sea solids should be applied again...
USP # 3,250,606
Nutrient Sea-Solids Solution for Hydroponic Farming

...In one set of experiments, crops of beans, tomatoes and
cucumbers were grown in 32 hydroponic beds in accordance with
the method illustrated in Figure 2. The seeds were sprouted in a
dilute solution of aqueous sea-solids and transferred to the
large beds as seedlings approximately 4 inches high.
Alternatively, the plants can be grown directly from seeds
without transplanting. The
growing plants were fed twice a day using a nutrient solution
made up by dissolving 116 lb of complete sea-solids in 100,000
lb water, i.e., 9.3 lb / 1000 gal water. full production was
achieved in about 100 days. Preferably, the concentration of
sea-solids in the nutrient solution employed should not exceed
about 8000 ppm by weight. While some growth can be
achieved using more dilute solutions, in general it will be
necessary to use solutions of sea-solids containing at least
about 1000 ppm. Solutions of greater than about 8000 ppm tend to
retard the plants in a manner similar to that observed with the
over application of conventional fertilizers, and generally
should not be employed.
In the manner described above, various crops including wheat,
oats, radishes, carrots, turnips, beets, tomatoes, corn,
strawberries, onions, and the like, have been grown successfully
with nutrient solutions comprising fresh water containing
containing in the range of about 1000 to 8000 ppm of dissolved
complete sea solids. These results are all the more surprising
in view of the comparative experiments which have been made
using solutions containing equivalent amounts of sodium chloride
only. Here, it was observed, that dissolved sodium chloride
solutions are definitely toxic to plants but that solutions of
complete sea-solids, even though they contained the same
quantity of dissolved sodium chloride, which when used alone was
toxic, can be used beneficially as a nutrient solution for the
growing plants.
If desired, conventional nitrogenous fertilzer materials and
inorganic salts can be employed together with diluted sea water,
or with a dilute solution of complete sea-solids in conventional
concentrations. For example, in similar experiments described
above, about 200 ppm of
K-nitrate sucessfully was employed for each 1000 ppm of
complete sea solids. Surprisingly, however, direct
supplementary nitrogenous fertilizer is not required with
sea-solid nutrient solutions as the hydroponic beds can be
inoculated with azabactor which is nourished well in nutrient
solutions comprising sea-solids and has the ability to fix
sufficient nitrogen out of the air. One method of supplying
azobacter bacteria to the hydroponic beds is to flow the
nutrient solution through a bed containing a legume crop such
as beans where the nutrient solution can come in contact with
the nodules on the roots of the legumes. Alternatively,
bean crops, or other legumes can be grown in hydroponic beds
interspersed with other beds connected in series so that an
adequate supply of azabacter is assured...


Related Patents
CN101152964
Solar energy sea water desalinating
warmhouse
Inventor: RENQUAN YANG [CN] ; ZENGCHAN ZHOU
2008-04-02
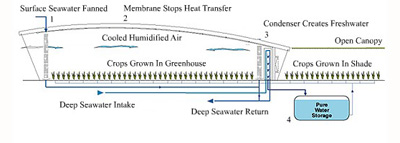
Abstract -- The present invention discloses a solar
seawater desalinating greenhouse, comprising a main greenhouse,
which is used for cultivating crops. A seawater distillation top
level is added and arranged on the front part of the top of the
greenhouse. An inner layer of the seawater distillation top
level adopts a wavelength selective color plastic plate. The
cover face of an outer lay of a roof adopts a translucent panel.
A freshwater water collecting groove is arranged at the gutter
in an inner layer of the roof. The freshwater collecting groove
is communicated with a freshwater storage pool through a
pipeline. A seawater storage pool, which is used for pumping and
storing the seawater is connected and fixed on a jetting pipe in
the inner layer of the roof through a water feeding column.
Jetting holes are opened on the two sides of the jetting pipe.
The present invention integrates the solar seawater desalinating
device and the environmental controlled greenhouse in a whole,
which is suitable for producing freshwater used in agriculture;
in addition, the present invention also has the functions of sun
shading and cooling, which can be used for desalinating the
seawater and cultivating crops. The present invention can
satisfy the need for vegetables supply and the drinking water
for men and animals in coastal areas, islands, desert alkali
region and so on where have seawater or brackish water but lack
of freshwater resource.
DE102005003754
Solar powered seawater desalination
method for greenhouse irrigation
Inventor: CHAFIK EFAT
2006-08-10
Abstract -- In a greenhouse system with solar-powered
seawater demineralisation, where greenhouse, evaporator and
cooler for condensation are combined due to the biological
microclimate of the crops to a functionally and economically
complementing unit, evaporation and condensation take place
within the greenhouse. The water is evaporated in open
containers, and condensation takes place around the plants. The
waste heat of the cooling system used for the condensation is
fed to an evaporator.
DE3332499
Greenhouse system with
solar-powered seawater demineralisation
Inventor: VERZICHT DES ERFINDERS AUF NENNUNG
1985-04-11
Abstract -- In a greenhouse system with solar-powered
seawater demineralisation, where greenhouse, evaporator and
cooler for condensation are combined due to the biological
microclimate of the crops to a functionally and economically
complementing unit, evaporation and condensation take place
within the greenhouse. The water is evaporated in open
containers, and condensation takes place around the plants. The
waste heat of the cooling system used for the condensation is
fed to an evaporator.
EA001982
GREENHOUSE
Inventor: VEN LIVIEN DOMEIN [BE] ; SUREDA MICHAEL
2001-10-22
CN1198885
Marine sunlight green house
Inventor: LIANG YONGLIN
1998-11-18
JP5057272
SOLAR FRESH WATER GENERATOR
Inventor: YAMANE AKIHIRO ; ISHIKAWA KIYOE
1993-03-09
FR2561637
Process and device for producing
distilled water from seawater
Inventor: GUIFFRAY MICHEL
1985-09-27
US4383891
Device for desalting brackish water
Inventor: CLAVIER PHILIPPE
1983-05-17
JP2009056343
NEW FLASH SEAWATER DESALINATION
DEVICES AND THEIR RELATED ENVIRONMENTAL SYSTEM
Inventor: KAMIUCHI KINGO
2009-03-19
CN101358443
Technical scheme of seawater
development for treating desert
Inventor: BAIZHONG ZHANG
2009-02-04
EP2017223
Method and device for energy
generation and desalination of sea water
Inventor: KRASS FLORIAN DR [CH] ; PRETTEREBNER JULIUS DR
2009-01-21
WO2008142459
COMPOSITE SOLAR TOWER CHIMNEY
Inventor: DERMITZAKIS EMMANUIL [GR] ; DERMITZAKIS
ARISTEIDIS
2008-11-27
US2006272933
Distillation methods and devices in
particular for producing potable water
Inventor: DOMEN JEAN-PAUL [FR] ; VIANNAY STEPHANE
2006-12-07
JP2008019152
METHOD OF PRODUCING SALT FOR
BUSINESS
Inventor: NAKAYAMA YOSHIHISA
2008-01-31
CN101084727
Method for controlling desert and
globe warming by seawater
Inventor: SHIJUN ZHANG
2007-12-12
CN200964358
Solar energy sea water desalination
glasshouse
Inventor: YANG RENQUAN ZHOU
2007-10-24
JP2007064630
ESTIMATION METHOD OF AIR EXCHANGE
AMOUNT BETWEEN ATMOSPHERE AND OCEAN
Inventor: ABE OSAMU ; SARMA VEDULA
2007-03-15
CN2866474
Water purifier
Inventor: WAN QISHENG
2007-02-07
KR20060012335
MAKING FRESHWATER AND SALT FROM
SEAWATER USING THE SUN AND A GREENHOUSE
Inventor: HEO JIN HYUK
2006-02-07
Charlie PATON, et al. -- Light Works Seawater Greenhouse