1. Gasoline Vaporization
Gasoline will burn only as a vapor. For that reason, all engines attempt to vaporize gasoline prior to combustion, sometimes achieving as much as 60% vaporization with the rest of the gasoline in the form of droplets ranging in diameter from 20 to 100 microns or more. These liquid droplets, along with the vapor and air, are introduced into the combustion chamber and burn incompletely, often producing detonation (engine knock), carbon deposits, high emissions, unequal distribution to and within the cylinders, and lower than optimum fuel economy. Engines that have achieved 60% vaporization prior to combustion have done so by using exhaust heat to obtain high temperatures in the intake manifold (over 240° F). These high temperatures can increase the vaporization of gasoline by boiling it to the 60% level, but they have the associated disadvantage of decreasing volumetric efficiency. Fuel injection is a technique used by the auto industry to increase volumetric efficiency by using cooler air but, because injected fuel has very little time to vaporize and mix with the air in the cylinder, the timing must be retarded with a consequent loss in fuel economy. The optimum solution to this problem is to vaporize the gasoline at low temperature (around 130° F, the ambient temperature of the air entering the manifold) so that the negative effects of high temperature are not a factor. This, however, had not proven practical prior to development of the WHV, although it had been demonstrated experimentally by GM in single-cylinder engine tests in the mid-1970s (Ref. 37).
In order to convert a liquid (such as gasoline) to a gas, a considerable amount of energy is necessary to break the forces that hold the liquid together (Ref. 38). The amount of energy required is determined by the magnitude of the intermolecular forces to be overcome. Generally, the components having higher boiling ranges have higher chemical energy contents. In the case of gasoline, this is complicated by the fact that it has at least 16 different components, each with its own boiling point under standard conditions (Figure 5). It is not necessary, however, to heat gasoline to its final boiling point in order to achieve full vaporization. As long as the dew point of the gasoline (determined by the air/fuel ratio of the mixture, the fuel composition, and the inlet manifold pressure) is below the manifold temperature, then the fuel can be vaporized and evenly distributed (Ref. 39).
Figure 5 Distribution and Boiling Ranges of Gasoline Components (Ref. 40)
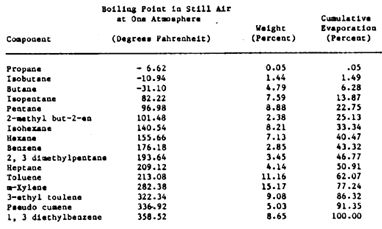
Figure 6 Typical Gasoline Distillation Curve (Ref. 41)

Because the heavier components of gasoline do not vaporize as readily as the lighter ones (because of their higher boiling points), there is a substantial amount of "heavy ends" (approximately 50% of the gasoline by weight) which is normally admitted to the combustion chamber as droplets (figure 6). There they coat the piston heads and cylinder walls and burn unevenly and incompletely, leading to crankcase dilution and contamination, engine deposits, gum and varnish formation, and spark plug fouling. The liquid gasoline gets past the rings and into the crankcase where it dilutes the oil and reduces its viscosity. The crankcase dilution is aggravated by the low engine operating temperatures associated with cold weather, especially in stop-and-go driving where engine temperatures do not reach levels high enough to evaporate the gasoline that is diluting the oil (Ref. 42). This promotes engine wear, poor performance, and high emissions. It has been widely suggested that if the volatility of these heavy ends could somehow be increased, then these negative effects would be greatly diminished (Ref. 43).
The importance of vaporization of the fuel to the engine performance is shown in Figure 7. In a standard engine, cold start problems are common and even as it warms up, the engine is subjected to stalling, carburetor icing, dilution, and engine deposits. If the vaporization level were about 95% of the intake mixture and the temperature in the intake manifold maintained at a level above 125° F (both are conditions which apply in Webster-Heise modifications), then the engine should operate in a zone of maximum power, smoothness, and economy under all speed and load demands.
Figure 7 Effects of Temperature and Vaporization on Engine Performance (Ref. 44)

The heat energy required to convert gasoline from the liquid to the gaseous state with no temperature change is called the latent heat of vaporization. The heat is transferred by both conduction and convection of energy between adjacent molecules (Ref. 45). Vaporization of gasoline may be accomplished, therefore, by increasing its manifold temperature (as in standard engines) or by lowering the pressure, by increasing the surface area of the droplets by forming more of them and reducing their diameters, and by increasing the heat transfer (as in the WHV). To obtain lower emissions, greater power, lower octane requirements, and greater fuel economy, it is far more effective to reduce manifold pressure and to increase heat transfer to the gasoline rather than to increase temperature.
At standard atmospheric pressure and temperature, some vapor will evolve from gasoline and given enough time the gasoline will evaporate completely. Because the pressure in the intake manifold is far less than one atmosphere (due to the vacuum created in the cylinders by the pumping action of the pistons and by the one-inch pressure drop caused the double screens), the boiling point of gasoline is lowered substantially (Ref. 46). At reduced pressure, latent heat is rapidly transferred to the hydrocarbon molecules by the large number of air molecules surrounding each fuel droplet, resulting in partial evaporation. If finely atomized gasoline is heated by the intake air in a zone of lower pressure (the partial vacuum of the intake manifold), it will undergo differential vaporization (rapid vaporization due to the sudden drop in pressure)(Ref. 47). This is the process at work in the WHV (Figure 8).
Figure 8 Evaporation of Gasoline (Differential evaporation begins at 125° F because the screens in the valve act as a nozzle matrix that creates low pressure streams of turbulent, high energy air and atomized fuel. The air temperature in the intake manifold does not exceed 130° F. Also note that vaporization with the WHV takes place in a fraction of a second whereas the theoretical evaporation curve is not time-limited. Source: Webster-Heise Corporation.)

The WHV allows the throttle plate to be closed to a greater degree than in a standard engine at the same speed and load. The valve adds one inch of Hg to the intake manifold vacuum and the throttle can add another 4 to 5 inches for a total vacuum that is 5 or 6 inches higher than standard at the same low speeds or loads. The vacuum decreases as the engine speed increases and, at wide-open throttle, the vacuum difference is reduced to one inch (See test 6, Summary of tests) because the vacuum due to throttling is no longer present. A differential of that size reduces the fuel/air ration mixture density ratio. In a standard engine, this would normally create a small power loss, but in the WH modification this facilitates vaporization and is apparently more than offset by a variety of combustion benefits and by greater volumetric efficiency. Although the density effect diminishes with higher speeds and loads as the vacuum decreases, there are marked benefits at low and moderate speeds and loads. These include an effective reduction in compression ratio, contributing to the tolerance for low-octane fuels.
The higher manifold vacuum created by the partial closing of the throttle enhances the vaporization of the fuel. More complete vaporization increases the evenness of fuel distribution to the cylinders and provides a more complete burn. Fuel consumption at a given speed is reduced in near proportion to the density ratio. Lower fuel consumption also reduces exhaust emissions. HC and CO are further reduced because the fuel is more completely vaporized before combustion. The lower effective compression reduces combustion peak temperatures and correspondingly reduces the formation of NOx. Early tests suggest that torque is enhanced by the better vaporization and distribution of the fuel. Further quantification of this effect on work output, engine speed, and fuel requirements would be valuable in assessing the performance of the valve.
According to the Webster-Heise Corporation, the valve has several features that are unique, without any of which it would not be effective. The valve, in self-regulated response to engine demand, moves up and down in the intake manifold. This is a critical characteristic because without the ability to sufficiently vary the screen area exposed to the air/fuel flow as needed there would be a restriction that would impede the flow of air and fuel and would result in a power drop. For decades single fixed screens have been tried, without success, to increase fuel vaporization, but because they were fixed they were optimized for only one power requirement. For all other states they actually constituted restriction that were worse than no screen at all. Because there was only one screen involved, there was also no control over the scale or intensity of the turbulence produced.
The WHV differs from these early attempts in that it consists of two stainless steel screens pressed together, a coarse stainless steel mesh (#50) followed immediately by a fine mesh (#120). The openings in the two adjacent screens constitute a series of thousands of micronozzles that force the liquid into small droplets (well under 10 microns in diameter) which quickly and totally vaporize within a few inches of the outer screens because of three improvements in the quality of the air that occur when it passes through the double screens:
1. more internal energy (from impingement and compression);
2. lower pressure (because of the Bernoulli Effect resulting from the air passing through the nozzles); and
3. greater intensity of turbulence (due to the small orifice sizes).
The screens must be tightly pressed together or the scale of turbulence will not be great enough and the rapid vaporization effect will be lost. The vaporization effect diminishes rapidly, and may be lost altogether, if the mesh sizes are varied by more than a few numbers. Very little variation in design can be tolerated in either case if maximum vaporization is to occur. Recent experiments suggest that even greater results may be obtained if one of the screens is rotated 45 degrees (in the same plane) with respect to the other so than the openings of the two screens are completely random and not in registration.
Another important factor in the operation of the valve is the "radial jump space" near the bottom of the valve. The air and fuel droplets are accelerated as they pass through the jump space and impinge on the vertical screens and are compressed as they enter he matrix, at which time much of their kinetic energy is converted into internal energy.
Flow through double screens of different mesh sizes is the equivalent of flow through a number of orifices or nozzles (a nozzle matrix)(Ref. 48). The pressure drop across the screens is controlled by a vacuum valve at a level of about one inch of mercury, although the pressure drop on the outer surface of the screen may be much greater (for a very short distance downstream from the valve) because of the relatively high vacuum that is created between each of the nozzle jets. The change in pressure is apparently sufficient to permit differential vaporization. Although the one-inch pressure drop is a slight restriction, it has the effect of reducing fuel consumption. The small negative effect that the one-inch pressure drop has on torque is more than offset by the greater density and combustibility of the charge in the cylinders (cool air mixed thoroughly with gasoline vapor) and the improved distribution to the weak cylinders. The greater mass of air available for expansion and the more favorable timing result in a net increase in available torque, despite the slightly higher vacuum.
Droplet size increases with the square of the orifice diameter (Ref. 49). The orifices in the WHV are exceptionally small, 0.0046 inches square or smaller. An orifice this size induces three times as much heat transfer as an orifice 0.008 inches in diameter (Ref. 50). Because the air is moving faster than the fuel droplets inside the valve, its high velocity can readily penetrate the liquid as it forms a film on the inner screen, producing the necessary turbulence and energy transfer to finely atomize the fuel (Ref. 51) Upon exiting the outer screen, transverse shear forces are set up by the intersecting sprays, which work in conjunction with the rapid drop in pressure to cause the fuel to vaporize. Before passing through the screen, the fuel is deposited as a film on the coarse mesh openings by turbulence within the valve. It is then accelerated through the smaller micronozzles (fine mesh), and then sheared by the other existing streams on the outside of the valve. This is important because only a slight reduction in droplet size causes a marked increase in fractional evaporation (Ref. 52). Small droplets evaporate virtually instantaneously, and a large proportion of the evaporation is accomplished during the deceleration of the droplets (Ref. 53). The diameters of the droplets exiting the valve are uniformly very small, probably less then 20% the size of the smallest droplets produced by the carburetor. Because of the droplet small diameters and the conditions created both inside and outside of the valve, most of the gasoline apparently vaporizes almost instantly (within 0.2-2.0 inches from the valve) at temperatures above 125° F.
The combined effect of internal circulation, oscillation, surface distortion, acceleration, deceleration, shearing, and depressurization, along with high intensity of air turbulence results in vaporization rate that is probably close to but not quite 95% (Ref. 54).
During experimentation with different screen sizes, it was found that only the combination of the #50 interior coarse mesh and the #120 exterior fine mesh would achieve the differential vaporization effect, probably because the intensity of turbulence is increased proportionately more than the scale of turbulence is decreased. These two meshes produce a reverse taper (wider spray) orifice with an exit diameter of at most 0.0046 inches. This is much smaller than conventional state-of-the-art micronozzles which have diameters as small as 0.008 inches. These valve-actuator combinations produce a very fine droplet size (Ref. 55). Because the heat and mass transfer value that results from dispersion by the WHV is exceedingly high (1/0.0046)2 or 47,260 (a gain of about 200% over a standard engine), ultra-fine droplets are favored (Ref. 56). The smallest commercial aerosol orifice is only (1/0.008)2 or 15,625 (a transfer rate one-third that of the WH rate). Because the orifice diameter is so small, the air stream is rotated and internally mixed within the nozzle and mixed again externally as it emerges from the nozzle, resulting in very fine sprays even at low feed rates and pressures (Ref. 57). The WHV can operate with a smaller orifice because the air is accelerated by passing through the double down-tube and by the greater pressure differential between the intake manifold and the interior of the valve.
Turbulence appears to be essential to the high speed of vaporization in the WHV. The high friction produced by the passage of air and gasoline through these screens results in an irreversible thermodynamic process in the intake manifold of an operating engine (Ref. 59). Nonequilibrium thermodynamics (the extension of classical thermodynamics to transport systems) is an area of science that is not yet well understood. It is clear, however, that this kinetic energy is converted to latent heat by high friction flow, thereby increasing the internal energy of the air to the point where it can cause a change of state of the gasoline in an environment of low pressure and high turbulence.
Turbulence has long been known to be essential to rapid vaporization. It has long been known that turbulence can be effectively used to homogenize fluid mixtures and to accelerate chemical reactions (Ref. 59). The mass transfer takes place through the relatively fast process of eddy diffusion and can result in values 10,000 times that of normal molecular diffusivity (Ref. 60). A 5% increase in turbulence in the air can increase the rate of vaporization 20 to 30% (Ref. 61). The WHV creates turbulence that is apparently over 200% higher than standard and may yield potential increases in the vaporization rate of 800% or more.
Turbulent flows are always dissipative because viscous shear stresses perform deformation work on the gasoline which increases its internal energy at the expense of the kinetic energy of the turbulence (Ref. 62). In other words, the rapid movement of the air molecules and gasoline droplets are converted to internal heat energy upon deceleration after exiting the WHV. The intensity of the turbulence shears the main flow after it passes through the WH screens.
The most important characteristic of turbulence is its ability to transport or mix momentum, kinetic energy, and contaminants such as heat, particles, and fluids Ref. 63). The rate of transfer and mixing are several orders of magnitude greater, therefore, than the rate s due to molecular diffusion (Ref. 64). The diffusivity of turbulence is the single most important feature as far as automotive applications are concerned because it increases the transfer rates of heat and momentum, both of which result in vaporization at temperatures well below the normal boiling point (Ref. 65). In the case of the WHV, this takes place at a temperature slightly above 125° F (the dew point of gasoline in an intake manifold). The ambient intake manifold temperature is about 130° F if the exhaust heat recirculation normally used in standard engines is removed and about 240° F or more if it is employed.
The fact that turbulence has not been used to better effect in automobile engines illustrated the degree to which turbulence theories are not understood. As Tennekes and Lumley point out (Ref. 66):
"Randomness and nonlinearity combine to make the equations of turbulence nearly intractable; turbulence theory suffers from the absence of sufficiently powerful mathematical methods. This lack of tools makes all theoretical approaches to problems in turbulence trial-and-error affairs. Nonlinear concepts and mathematical tools have to be developed along the way; one cannot rely on the equations alone to obtain answers to problems. This situation makes turbulence research both frustrating and challenging; it is one of the principle unsolved problems in physics today."
Perhaps the most important point made by Tennekes and Lumley is the following (Ref. 67):
"This book has been designed to get this point across. In turbulence, the equations do not give the entire story. One must be willing to use (and capable of using) simple physical concepts based on experience to bridge the gaps between the equations and actual flows. We do not want too imply that the equations are of little use; we merely want to make it unmistakably clear that turbulence needs spirited inventors just as badly as dedicated analysts."
Screens have been used in other applications where similar conditions exist (but which do not involve combustion) and where similar effects were desired (Ref. 68):
"...At large pressure-drop coefficients, turbulence may be caused by screens and it is recommended that several low-pressure-drop screens are preferable to a single high-pressure drop screen. This is the practice normally followed in blowdown tunnel design."
The intake manifold equipped with a WHV is essentially a minute wind tunnel with a one-inch pressure differential or more and an ambient temperature of about 130° F. It has been observed that (Ref. 69):
"Uniform fluid distribution is essential for efficient operation of chemical processing equipment such as contactors and reactors, mixers, burners, heat exchangers, extrusion dies, and textile-spinning chimneys. To obtain optimum distribution, proper consideration must be given to flow behavior in the distributor, flow conditions upstream of the distributor, and flow conditions downstream of the distributor...
"A non-uniform velocity profile of turbulent flow through channels or process equipment can be smoothed out to any desired degree by adding sufficient uniform resistance, such as perforated plates or screens across the flow channel."
Droplet breakup is accomplished by the WHV through three separate but related processes (Ref. 70):
1. The impingement of the fuel and the air, after accelerating across the radial jump space, on the screens;
2. A pressure drop due to the presence of high-friction nozzles (the combined screens);
3. The high intensity of turbulence caused by the micronozzle effect of the adjacent nonuniform screen openings.
In all of these, the density of the air involved is an important consideration. Perry and Chilton found that when a solid jet of liquid (in this case gasoline) is being impinged upon a gas (air), its relative velocity assumes a much greater importance than it does without impingement (Ref. 71) Generally, there is a strong decrease in droplet size with increasing gas density. It is important to note that in an engine modified with a WHV, the intake manifold temperature may be as low as only 130° F versus the 240° F that is normal in standard engines (the extra heat recirculation can be removed because it is no longer necessary for the vaporization of gasoline). Thus, the cool air is much denser and capable of greater droplet size reduction because of its greater dynamic force (Ref. 72)
There are four flow regions associated with a valve of this type (Ref. 73):
1. Region of flow establishment -- a short region whose length is about 5 nozzle diameters or slot heights (for a slot of infinite width). The fluid within the cone or core of same length has a velocity of about the same as the initial discharge velocity;
2. A transition region that extends to about 8 nozzle diameters, slightly less for slots;
3. Region of established flow -- the principal region of the jet, extending to about 100 nozzles diameter or about 2000 slot heights;
4. A terminal region where the residual center-line or maximum velocity reduces rapidly within a short distance. For air jets, the residual velocity will reduce to less than 1 ft./sec., usually regarded as still air.
Virtually all of the droplet breakup (and much of the vaporization) probably occurs within the first half-inch after exiting the outer screen (113 nozzle diameters times a nozzle diameter of 0.0046 inch).The time required for this phase dispersion is about 13 milliseconds (Ref. 74).
The rapid rate of vaporization in the WHV is highly dependent upon the unusually high internal energy of the air and the rate at which it can be transferred to the fuel droplets which also are subject to an increase in their own level of internal energy (Ref. 75).
Pierce has shown that (Ref. 76):
"For an adiabatic flow with zero shaft work, the changes in system directed kinetic energy will result in system temperature changes with changes in the internal energy content and distributions in the various degrees of freedom...
"As the gas is decelerated, there is an energy exchange between the directed kinetic energy of the gas stream and the internal energy of the gas itself. Assuming an adiabatic stagnation, then all of the directed kinetic energy must manifest itself as an increase in internal energy. The stagnation can occur so rapidly, however, that the vibrational degree of freedom may not accept its share of this directed kinetic energy as rapidly as necessary to insure that equilibrium exists. Then since the total energy is constant, the active degrees of freedom must accept more energy than equilibrium requires to compensate for the deficiency in energy content of the lagging degree of freedom...
"In the flow through the shock front there is a sharp reduction in the directed kinetic energy of the gas. Assuming adiabatic flow along stream tubes, then this directed kinetic energy must be transferred to the gas molecules and manifest itself as intrinsic or internal energy of the gas. Since the vibrational degree of freedom is slow in acquiring its share of this new energy, a non-equilibrium situation can occur where initially virtually all this transformed directed kinetic energy is absorbed by the active translational and rotational degrees of freedom, and almost none is absorbed by the inert vibrational degree of freedom."
Another very important mechanism acting concurrently with the turbulence the WHV is differential vapor recoil. This principle, despite its dramatic effects on vaporization (especially at low temperatures), has not been well understood outside of research laboratories (Ref. 77). The WHV is apparently the first application of this principle to automotive use. Palmer has described the effects of vapor recoil on the evaporation rate (Ref. 78):
"A mechanism for inducing spontaneous convection, that of instability induced by differential vapor recoil, was first noted and correctly interpreted by Hickman in 1952 but has since attracted surprisingly little attention despite its dramatic effect on the evaporation of liquids at pressures above 1 Torr.
"Hickman (1952) has shown that an increase as high as 20-fold in the liquid evaporation rate may be enjoyed if the interface is interrupted by differential vapor recoil. The onset of such convection appears as a sharp transition fro a relatively quiescent evaporating liquid surface as the pressure above the liquid is decreased.
"Because mass must be conserved, the change in fluid density during evaporation results in a discontinuity in both the fluid velocity normal to the interface and the rate of transport of linear momentum across it. Momentum must also be conserved. Therefore the discontinuity in velocity results in a downward force on the interface (vapor recoil) which increases with the evaporation rate and with an increase in the density ratio of the liquid and gas phase. Since the density of the gas phase is linearly proportional to the pressure, the magnitude and, thus, the importance of this vapor recoil force increase markedly as the pressure is reduced."
If a liquid in contact with its own vapor is evaporated at low pressure, interfacial heat transfer and evaporation rates can be increased dramatically by the onset of interfacial convection by differential vapor recoil (Ref. 79). The resulting discontinuity produces a downward force on the interface (vapor recoil) which increases with the increasing evaporation and the decreasing pressure. Palmer has demonstrated that this mechanism dominates interfacial behavior for evaporation into a partial vacuum (Ref. 80). This same research provides the explanation for the likelihood that the Webster-Heise vaporization rate is probably on the order of 95% rather than 100% (compared to approximately 60% prior to combustion in warmed-up standard engines). Experiments and stability analyses have shown the extreme sensitivity of the vapor recoil mechanisms to interfacial contamination such as engine oil and water (from condensation) (Ref. 81). As a result, the interface (between the liquid and vapor phases) will still develop some "torpid" patches about 5% of the time because evaporative fluxes are suppressed wherever the less volatile material accumulates on the surface (Ref. 82).
Phase changes of fluids in pipelines have been observed for many years. In pipelines, this is an unwanted effect, but it serves as a useful analogy in explaining the way in which gasoline flashes to vapor on exiting the WHV and entering the intake manifold, which is very much like a pipeline in that it is a tube transporting a liquid which is subjected to pressure changes. In a pipeline, differential vaporization occurs because of the rapid relative velocity of the gas phase (Ref. 83). As the saturated liquid flows through the pipe, friction (as in the Webster-Heise screens) causes a pressure drop in the direction of flow followed by vaporization (Ref. 84). The mass transfer rate from the gas back to the liquid phase is zero (or very close to it)(Ref. 85). The vapor is swept away without contributing to the equilibrium composition, promoting further vaporization. The cooling effect accompanying the pressure drop is much less than would be predicted in an equilibrium model and apparently has the effect of increasing the volatility of the liquid (Ref. 86).
2. Mixing
Mixing is a critical part of the pre-combustion phase of the operation of a gasoline-powered engine. It has been somewhat neglected in current engine design in which only limited mixing occurs due to the presence of baffles and ridges in the intake manifolds. This not only is a relatively ineffective way to mix air and gasoline, it often has a negative effect in that it provides numerous opportunities for the liquid gasoline issuing from the carburetor to collect on the walls of the intake manifold.
Harrow has described the compromises that current engines represent because of imperfect mixing (Refs. 87, 88):
"Traditionally, at full throttle, part of the latent heat of vaporization of the fuel us used to chill the mixture entering the engine and thus to increase the maximum power. In the absence of a perfect mixture preparation system which will provide both a chilled mixture and a fully atomized fuel, engine inlet manifolds have to cope with mixtures of liquid and vapor. A consequence of this need to combine several functions is that an engine mixture preparation system is a package that represents the manufacturer’s best compromise between the conflicting requirements of power, emissions and economy. The result is invariably sensitive to minor changes in the carburetion and inlet system geometry, which can give an improvement for one type of engine condition at the expense of performance in another.
"The engine manufacturer tries to ensure uniform distribution of fuel between the cylinders by providing a hot spot to transmit exhaust heat to the mixture and by drawing some of the inlet air to the engine from around the exhaust manifold. The inlet manifold is also designed to permit a certain amount of liquid flow to flow freely to the cylinders. These procedures are not normally very effective, and almost invariably some fuel maldistribution does occur."
A homogeneous mixture of gasoline vapor and air is the ideal condition for the inlet charge of a gasoline engine (Ref. 89). Vaporization greatly improves the mixing rate but, even in high-heat engines, the charge is still far from homogeneous by the time it reaches the entrance of the cylinders. This is because insufficient turbulence is available to cause the gasoline vapor molecules and air molecules to associate completely. It is compounded by the fact that many intake manifolds are not sufficiently large to permit good mixing.
The WHV, by design, forces the gasoline vapor and air to mix thoroughly within a short distance from the outer screen. The high turbulence and shearing forces subject both components to an intense mixing process. Because the gasoline is in a vaporized or finely atomized state, it is far less likely to impinge and recondense downstream in the intake manifold. To reduce this possibility, all baffles and other obstructions are removed to provide a straight and clear path to the intake valve for each cylinder.
The mixing principles employed in the WHV have been known for some time. As Goulburn and Hughes, based on work by Ashley, have observed (Ref. 90):
"There are two important principles: break the stream down into a series of smaller streams to speed up mixing and break down stratification patterns; then pass the streams across and through one another to redistribute major stratification patterns. Multiple jets tend to overcome two undesirable characteristics of all of the natural mixing means, which are a relatively unmixed core inside a well mixed outer envelope, and insufficient turbulence.
A design of mixer was produced which met the above criteria and had the added advantage that the heat transfer fins in the vaporizer could, with some modification, also be used to produce a series of jets of mixture directed so that adjacent layers of jets have an interface where fluid shearing will occur. Consider a vaporizer with horizontal fins, and consider one flow passage between the fins. At the end of the passage the flow encounters vertical deflector tabs positioned at 45 degrees to the flow turning the flow to the left (say). The flow passages immediately above and below will have deflector tabs set at 45 degrees to the flow, but turning the flow to the right. The two layers of flow will then, at their interface, be shearing across each other at 90 degrees."
Virtually the same conditions exist in the WHV.
The benefits of improved mixing have been described by Harrow and are well worth considering in the case of the WHV (Ref. 91):
1. The effect of mixture quality on fuel economy is significant and increases in importance the weaker the mixture strength becomes. It is of particular importance for smooth engine operation under transient conditions;
2. In multi-cylinder engines, good mixture quality eliminates inter-cylinder fuel maldistribution and allows the carburetor to be tuned to the maximum-economy mixture strength;
3. Under cold-running conditions, good mixture quality allows minimal use of the choke, thereby reducing short-trip fuel consumption;
4.With a perfect fuel mixture preparation system, at least part of the engine power could be controlled on mixture strength alone. This would significantly improve part-load fuel economy;
5. Many devices exist for improving mixture quality. They improve fuel economy, however, only if the mixture strength and spark timing are adjusted correctly to exploit the benefits that they confer.
3. Distribution
Fuel distribution in conventional engines is a problem that is widely recognized. For efficient combustion and smooth engine operation, the air and fuel must be thoroughly and uniformly mixed, delivered in equal quantities to each cylinder, and evenly distributed within each cylinder (Ref. 92). It is generally acknowledged that the3 best way to achieve this is through a homogeneous charge (Ref. 93):
"The process whereby the gasoline fuel and air are metered in the desired proportions and then mixed has been isolated as one of the key elements in making improvements in engine combustion, hence in efficiency and products of combustion. It has been conclusively established that a homogeneous mixture of gasoline and air is the best condition for the inlet charge of a gasoline engine..."
In actual practice, however, the mixture in the intake manifold (even with the addition of heat) is not completely vaporized. As a result, the quality and quantity of the fuel charge reaching the cylinders vary considerably (Ref. 94). Because this distribution is unequal, some cylinders produce more power than others and some will have a greater tendency to detonate than others (Ref. 95). If the carburetor is adjusted to provide an adequate mixture to the weak cylinders, then the other cylinders will receive a mixture that is far too rich. The industry has compromised, therefore, by accepting weak and rich cylinders in order to obtain sufficient performance from the other cylinders (Figure 9). Unfortunately, these extreme cylinders produce a number of undesirable effects. The rich cylinders receive too much fuel which results in incomplete combustion and high hydrocarbon emissions because there is insufficient air available to burn all of it. The lean cylinders, in contrast, receive sufficient air but are relatively starved for fuel, and the resulting combustion pressures are so low that little useful work is done on the crankshaft. NOx formation is likely in these cylinders because of the relative abundance of oxygen and because the nitrogen is subjected to high heat levels for a comparatively long time due to the long duration of the primary flame.
Figure 9 Variation in the air/fuel ratio from cylinder to cylinder (Ref. 96)

Figure 10 Maldistribution resulting from deflection by the throttle plate (Ref. 100)

Figure 11 Maldistribution results from heavy components of gasoline flowing to end of manifold rather than entering branches (Ref. 101)

There are several reasons for these variations in distribution (Ref. 96). When the throttle valve of the carburetor is partly open (as it almost always is except at full throttle), the flow of air and fuel is deflected to one side (Figure 10). The choke valve can also have a similar effect. Even more important is the fact that the heavy components of the air/fuel mixture have greater inertia than the lighter ones. Consequently, the heavy components move past the branches of the manifold. The cylinders supplied by those branches, therefore, receive too many light components (which burn rapidly) and too few heavy components (which contain most of the energy). The problem is shown in Figure 11. For the same reasons, some cylinders will receive more tetraethyl lead (or other negative catalysts) than others. Those cylinders receiving the leanest mixture, the lightest gasoline components, and the least amount of negative catalyst (such as tetraethyl lead) will have the greatest tendency to knock (Ref.97). The variation between minimum and maximum cylinder pressure on the power stroke in a typical 6-cylinder engine is on the order of 185 psi, with two cylinders delivering the minimum (125 psi) and only one delivering the maximum (310 psi)(Ref. 98). The addition of multiple barrels to the carburetor reduces to some extent the variations in cylinder pressure due to poor distribution but does not eliminate them.
The distribution problem has been described by Toboldt and Johnson (Refs. 102, 103):
"The purpose of a carburetor is to deliver a metered amount of atomized fuel mixed with air, to the manifold. However, regardless of how well mixed and vaporized the fuel mixture is as it leaves the carburetor, its characteristics are unchanged as it passes through the manifold. Cold surfaces in the manifold will cause some of the vaporized fuel to condense, and changed in direction of flow will, through inertia, cause some portions of the mixture to settle out. These conditions have been observed using glass manifolds.
"The problem is further complicated by the characteristics of the fuel itself. Formerly, when the fuel was highly volatile, the problem was not so difficult. With today’s fuels, which are relatively non-volatile, it is necessary to supply heat to obtain better vaporization and more equal distribution of the fuel to each cylinder."
One of the ways the industry has used to avoid quantity distribution problems is fuel injection. This approach delivers a precise amount of fuel to each cylinder and injects it into the cylinder separately from the air flowing through the intake manifold. This system eliminates the need for manifold heat and permits lower intake temperatures, hence more volumetric efficiency. Fuel injection has its own associated problems, however, including higher emissions, lower fuel economy, and more difficult tuning. Because the gasoline is injected directly into the cylinder as a liquid rather than as a vapor, the quality of distribution is very low and more time is needed for the heat in the cylinder to partially vaporize the gasoline. As a result, the spark must be retarded severely with a considerable loss of fuel economy (the power stroke is partly over before the combustion begins)(Ref. 104). Retarding the spark does, however, reduce NOx emissions but not HC or CO emissions. The WHV has been shown to reduce all three simultaneously.
The effect of quality distribution on HC emissions has been described by Marsee and Olree (Ref. 105):
"...Quality is indirectly measured by measuring its effect on HC emissions and HC turn-up point as the mixture is leaned. Poor mixture quality at lean mixture leads to slow flame propagation and the extinguishing of combustion as the pressure and temperature decrease with the descending piston. Poor quality also leads directly to increased HC emissions as some portions of the charge may be too lean to burn."
The WHV is designed to address, through charge conditioning, the distribution shortcomings of both the standard carburetion and fuel injection approaches. The advantage over standard carburetion is apparently a result of the elimination of two long-associated problems: (1) incomplete vaporization of the fuel, and (2) incomplete mixing with the intake air.
The controlled turbulence created by the WHV (but lacking in a standard engine) forces the air and vaporized fuel to mix to a much greater extent than would otherwise be possible. Quantity distribution at idle is an especially difficult problem in a standard engine because of the low rate of flow (the throttle valve is almost completely closed)(Ref. 106). With the WHV, however, the combination of more complete vaporization and better mixing apparently permit idle speeds to be halved.
The advantage of the WHV over fuel injection is a direct result of the elimination of liquid fuel in the cylinders. By providing a central injection point for a substantially vaporized and thoroughly mixed fuel-air mixture, the quantity distribution problem that fuel injection addresses is effectively eliminated, as measured in the test at Ethyl (See Test 1, Summary of Tests). Just as importantly, the quality distribution is much greater with the WHV because only completely mixed fuel vapor and air reach the cylinder, as opposed to the liquid fuel that is injected separately by fuel injection. With the WHV, the spark can be advanced considerably to obtain maximum fuel economy (including lower pumping losses) and lower emissions without sacrificing the power gains available through fuel injection.
4. Volumetric Efficiency
One of the reasons that no engine is ever 100% efficient is the difficulty of getting a full charge of combustion mixture into each cylinder. A theoretical full charge does not reach the cylinder because of restrictions in the intake manifold, atmospheric temperature, valve timing, and other factors (Ref. 107). Volumetric efficiency is the ratio of the charge actually taken in per cycle to a complete charge. This ratio is very sensitive to heat (because it lowers the density of the incoming air) and atmospheric pressure (because the pressure difference between the air inside the cylinder and the air outside will determine the volume of air admitted to the cylinder).
The importance of additional air in raising the power output of an engine has been understood since 1902 when Louis Renault patented the supercharger. This device compressed the air, using a pump driven by the engine, to force more air into the cylinders. Even though superchargers are effective in raising power levels, they do so at a considerable loss in fuel economy due to the additional demands on the engine to drive the pump. They have generally been abandoned in favor of turbochargers, which use the pressure of exhaust gases to compress the intake air. While they have proven to be more satisfactory than superchargers, they are an expensive and relatively fuel inefficient means of boosting power outputs on gasoline-powered engines.
The WHV accomplished much of the same effect as superchargers and turbochargers without increasing engine loads or pumping losses. Torque gains of as much as 20% were recorded in Test 5, Summary of Tests. This is accomplished by removing the high intake manifold temperatures, which is currently used by automobile manufacturers to increase vaporization rates but which appears to be unnecessary when a WHV is used. Because incoming air reaches the intake valve at 130° F rather than 240° F or more, its density is much greater. In practical terms, this means that more air is available to complete combustion and to expand upon combustion, creating more pressure on the pistons immediately after top-dead-center. This results in substantially more torque. Because there is more air mass to expand after combustion, pressure is also exerted for a longer duration during the power stroke when the crank angle is most advanced (between 0° and 90° ). It should be noted, however, that substantial torque gains may be possible even if the heat is not removed on the Webster-Heise system. Torque increases of up to 40% may be obtainable (as shown in Test 6, Summary of Tests), and these gains are very likely due to the improved distribution and the greater pressures that are created in what would otherwise be the weak cylinders.
The benefits of lower intake manifold temperatures are readily apparent. Brake specific fuel consumption and engine coolant temperatures are both reduced (Ref. 108). Based solely on fuel consumption considerations, the optimum intake manifold temperature at rated power conditions is 130° F for gasoline (Ref. 109) (5° F over the dew point temperature of 125° F). This is the same approximate manifold temperature that ios maintained with a WHV.
B. Combustion Effects
1. Combustion Stoichometry
The ideal combustion is stoichometric, meaning that there is no excess gasoline or excess air. This condition, in which the ratio of air to gasoline is 14.7 to 1 by weight, produces the hottest flame and the lowest emissions (Ref. 110). The maximum temperature peaks and expansion pressures occur when the combustion air is 100% of stoichometric (neither too rich or too lean), giving further evidence that stoichometric combustion is the most effective condition for the utilization of thermal energy (Figure 12)(Ref. 111).
Figure 12 Influence of air/fuel ratio on power, fuel consumption and emissions (Ref. 112)

In this ideal situation, the gasoline is fully vaporized and distributed evenly to and within the cylinders, where blue-flame combustion converts to heat energy of the gasoline to mechanical energy. The gasoline must first be vaporized to individual molecules before oxidation to carbon dioxide and water can occur (Ref. 113). As discussed earlier, the incomplete vaporization and uneven distribution greatly affect the air/fuel ratio in individual cylinders even in the most modern automobile engines. In the rich cylinders, there is insufficient air to burn the relatively large amount of gasoline present, creating most of the HC and CO emissions. In the lean cylinders, the heat and pressure in the primary flame force some of the excess oxygen to combine with the nitrogen in the air rather than oxidizing the hydrogen and carbon for which the oxygen normally has a greater chemical affinity. To reduce NOx emissions, engine manufacturers currently recirculate some of the exhaust gases back into the primary intake air. Because these gases are less reactive, the formation of NOx is inhibited but the combustion process is deprived of oxygen that could be used to produce more energy from the unburned hydrocarbons.
Excess oxygen is not desirable either. Although it can decrease HC and CO emissions within certain limits, it does reduce power because the full amount of the work available from the fuel is reduced by the lower flame temperature resulting from the heating of the excess air to the temperature of the burned gasoline (Ref. 114). The flame temperature can be increased to its maximum, therefore, by using a stoichometric air/fuel ration (when possible) and low levels of residuals or recirculated gas (Ref. 115). The flame stability afforded by the use of a vaporized charge is very high under these conditions (Ref. 116).
Despite the recent interest in lean-burn combustion systems, it may be more desirable to use an air/fuel mixture that is closer to stoichometric. If the combustion conditions can be brought closer to the ideal stoichometry, then the optimum mixture is probably also very close to stoichometric. In an internal combustion engine, it is impossible to get something for nothing as some lean-burn techniques attempt to do. The best that can be achieved is the conversion of as much chemical energy in the fuel charge as possible into mechanical energy that can perform useful work with a minimum of harmful emissions.
2. Octane Requirements
One of the most unexpected and potentially most important gains shown by the WHV is its lowering of engine octane requirements by 10 points or more. To understand the reasons for this reduction, which was not an original objective of the inventors, it is necessary to examine first the combustion problems identifiable in standard engines and then to analyze the effects that the WHV has on them.
When the fuel charge is burned in a combustion chamber, the gasoline vapor and oxygen (which comprises approximately one-fifth of the air) combine and form new compounds. This reaction releases a large amount of heat which drives temperatures up to about 3500° F. This creates pressure on the gases in the cylinder which expand to push on the piston, forcing it downward on its power stroke. Normal combustion consists of three phases (Ref. 117, Toboldt and Johnson):
"As soon as the ignition spark jumps the gap of the spark plug, a small ball of blue flame develops in the gap. This ball is the first stage or nucleus of the flame. It enlarges with relative slowness and, during its growth, there is no measurable pressure created by the heat.
"As the nucleus enlarges, it develops into the hatching-out stage. The nucleus is torn apart, so that it sends fingers of flame into the mixture in the combustion chamber. This causes enough heat to give a slight rise in temperature and pressure in the entire air/fuel mixture. Consequently a lag still exists in the attempt to raise pressure in the entire cylinder.
"It is during the third stage, or propagation, that the effective burning of the fuel takes place. The flame burns in a front which sweeps across the combustion chamber, burning rapidly and causing great heat with its accompanying rise in pressure. It is this pressure which causes the piston to move downward.
"During normal combustion, the burning is progressive. It increases gradually during the first two stages. But, during the third stage, the flame is extremely strong as it sweeps through the combustion chamber. However, there is no violent or explosive action such as when detonation (ordinarily responsible for pinging and knocking) occurs.
"If detonation takes place, it occurs during the third stage of combustion."
When the flame is first propagated, it sweeps from the area around the spark plug toward the walls of the combustion chamber. Because the air/fuel mixtures used in all production gasoline engines contain a substantial amount of liquid fuel droplets, especially on the wetted surface of the cylinders and the piston heads, fuel-rich pockets (due to incomplete vaporization and poor mixing) tend to develop in the extremities of the combustion chamber. These pockets are compressed and heated far past the self-ignition point (even for high-octane fuels) by the combustion emanating from the spark plug. As the flame front advances, these unburned gases and droplets are further heated and compressed. This extreme heating of the unburned mixture can cause spontaneous ignition and explosion independent of the flame. It is this rapid, uncontrolled burning that is known as detonation and is perceived as knock or pinging when the rapidly burning flame front, expanding outward from the spark plug, collides with the secondary wave front produced by the detonation of the fuel-rich pockets ahead of the primary front (Ref. 118).
Detonation is more than an annoyance or an indication of inefficient engine operation. It can do great harm to an engine and can considerably impair its performance and expected life. It can shatter pistons, burst cylinders, melt pistons, overheat engines, break spark plugs, overload bearings, increase fuel consumption, and decrease power (Ref. 119). The problems caused by detonation have been observed by Khovahk (Ref. 120):
"An engine should never be allowed to operate for a long time with knocking, since the shock waves sharply increase heat transfer from the combustion products to the walls. This leads to overheating of the engine and destruction of some of the components in the combustion chambers (piston edges, gaskets between the cylinder and its head, electrodes and insulators of spark plugs). The vibrational nature of the load on the piston with detonation may destroy the antifriction layer in the connecting rod bearings and intensify the wear of the upper part of the cylinder liners, since the shock waves destroy the oil film on the surface of the metal, causing dry friction and also corrosive wear by the active substances, particularly nitrogen oxides, contained in the combustion products."
There are several factors that contribute to detonation (Ref. 121). These include lean air/fuel mixtures (which are more likely to contain uneven distribution of the gasoline with each cylinder), fuel of too low an octane rating (especially under poor vaporization and mixing conditions), high compression ratios, over-advance of ignition timing, engine lugging, and buildup of carbon deposits in the combustion chamber.
A separate but related problem is pre-ignition, the burning of the fuel charge before the ignition spark. If this premature combustion is completed before the spark-induced ignition begins there is no knock. Pre-ignition can cause detonation which, in turn, can cause more pre-ignition. The principal cause of pre-ignition is excess heat, particularly that retained by hot carbon deposits which act as impromptu spark plugs. This can raise the octane requirements by several numbers and, if excessive, can break the crankshaft.
The negative effects of detonation and pre-ignition have led gasoline refiners to increase the octane ratings of their fuels. The rating of a fuel is made by comparing it to mixtures of normal heptane and iso-octane (hence the term) in a test engine under certain test conditions. The combination of these pure hydrocarbons which provide the same knocking in the engine as the gasoline being tested is considered to have that "octane". The octane number of the fuel is the percent of iso-octane in the matching mixture of iso-octane and normal heptane (Ref 122).
The gasoline additive used as the most common knock suppressant for the past 50 years is tetraethyl lead. Other additives are also used to reduce the buildup of carbon deposits, to absorb water in the fuel, and to lubricate the valve stems and upper cylinder walls. Most of these additives have high associated toxicity, and some are being phased out by law. Fuel economy was not a principal objective in the development of these additives (Ref. 123). The major concern was in avoiding engine destruction due to knock.
Both detonation and pre-ignition are serious combustion problems. Both are uncontrollable explosions which generate their own flame fronts. Because they normally occur early in the process of compression and ignition, both violations reduce engine power because work is required to compress the combustion products (Ref. 124). They also transfer additional heat to the cylinder walls because burnt gases with high temperatures remain longer in the cylinders.
Even with the addition of knock suppressants, carbon deposits in high-compression engines using high-octane gasoline sometimes produce foci of self-ignition independent of the primary flame front. Known as "rumble", this violation occurs after periods when the engine has been operating for long periods under light loads (forming carbon deposits in the process) and then run at full throttle (Ref. 125). Often the engine will continue to run at very inefficient levels after the ignition is turned off because the compression and the hot carbon deposits are sufficient to cause self-ignition. This is known as "dieseling" or "engine run-on".
Tetraethyl lead and other anti-knock additives are negative catalysts which slow down the rate of flame travel and combustion so that extreme heat and pressure waves will not be created. This reduces the detonation tendency of the air/fuel charge and lowers the combustibility of the fuel-rich pockets in which detonation takes place.
Reducing the volatility of the fuel (low-octane gasoline is more reactive than high-octane) through the use of negative catalysts hinders detonation beyond the primary flame front. It does, however, have numerous associated disadvantages. Most anti-knock additives, particularly tetraethyl lead and aromatics, are highly toxic, and some are carcinogenic. Because of this, Federal laws have been enacted to reduce and eventually phase out their use. At the same time the demand for fuel economy has led manufacturers to increase compression ratios, which increase the octane requirements of their engines. At the same time, the refining industry is severely depressed and only the largest companies can afford the expensive reconfigurations necessary to increase octane of their gasolines. Those that can increase them do so at a great expense in extra crude oil consumed through more severe processing.
The alternatives to chemical octane have never been attractive because there appeared to be no effective way to improve the conditions in the combustion chamber. The fuel-rich pockets, carbon deposits, and low-density air inherent in automobile engines strictly limited the options. The Webster-Heise Corporation, however, claims that processing the charge (as described earlier) through a valve eliminates or greatly reduces those factors that artificially increase octane requirements. The highly vaporized and fully mixed charge prevents the formation of fuel-rich pockets, the low-temperature vaporization reduces the charge temperature, and because combustion is more complete, carbon deposits do not form.
The rate of combustion is of critical importance in determining the efficiency of an engine. As Khovahk has observed (Ref. 126):
"Combustion takes place in the gaseous phase. For the reactions of oxidation to develop at sufficiently high velocities, the liquid fuel should be vaporized and its vapors mixed with the air. The process of combustion takes place at the maximum velocity in homogeneous mixtures when the fuel molecules are uniformly distributed among the molecules of oxygen. In heterogeneous gas mixtures, the rate of combustion is mainly determined by the velocities of mutual diffusion of the fuel vapors and air, while the rates of the chemical reactions are of a secondary importance. The rate of liquid fuel consumption is limited by the rates with which it evaporates and the vapors formed mix with air."
Because of the conditions that apply in a conventional engine, slowing the reaction rate of the fuel is the only way that detonation can be avoided when it would otherwise occur. If those conditions are made more benign, as is possible through improved charge preparation and distribution, then a fuel of substantially lower octane can be used without detonation. Khovahk has noted that, "Detonation is hampered by the factors that accelerate combustion of the last portion of the charge in the flame front" (Ref. 127). If the flame travel is faster and less time is required to develop the pre-flame reactions in the unburned part of the charge, then the tendency to detonate is reduced (Ref. 128). If the air/fuel mixture, therefore, is highly vaporized and homogeneous, is relatively low in temperature (to accelerate the propagation of the flame front), then fuel with higher reactivity (lower octane) can be used. The size of the octane reduction varies but, based on laboratory tests, appears to be at least 10 octane points lower and may be as much as 15.
Flame speed is known to increase with decreasing initial droplet size, with the fastest speed for vapor (Ref. 129). The presence of droplets produces local variations in the air/fuel mixture which can cause the formation of a secondary diffusion flame in the wake of the propagating flame and produce local variations in the combustion temperature. This unsteadiness is due to the time-varying stratification of the droplets and their transient heating (Ref.130). These variations work against efficient engine operations and tend to increase the formation of undesirable emissions. In a premixed homogeneous charge, the secondary flame does not develop and the primary flame tends to propagate with a steady velocity (Ref. 131).
The ability of the Webster-Heise system to run on low octane gasolines with high spark advances and without detonation (as shown in the Summary of Tests) tends to suggest that vaporization of the gasoline is responsible for the likely absence of a secondary flame and consequent knock.
Another apparent factor in reducing the octane requirement with a WHV is the increased turbulence of combustion. It can be inferred from internal combustion theory that, because of the homogeneity if the charge, the intensity of the turbulence of the charge upon admission to the cylinder is probably higher than in a standard engine. This turbulence greatly increases the velocity of the flame front (Ref. 132). Experiments independent of the Webster-Heise development have shown that these intensive vortices and turbulent pulsations distort the flame front and break it into separately burning foci which greatly increase the actual surface of combustion by several orders of magnitude (Ref. 133). The turbulent pulsations that are smaller than the thickness of te laminar flame front do not produce distortions, but they do intensify the processes of heat transfer and diffusion in the flame front thereby increasing the combustion rate in the foci. These two factors combine to cause the velocity of the turbulent flame to increase in approximate proportion to the intensity of the turbulence (Ref. 134). These factors may also apply in the Webster-Heise system.
Another important factor in octane requirements is the manner in which the charge burns. As Khovahk has shown (Ref. 135):
"Heterogeneous mixtures such as jets of gas flowing into air or droplets of atomized liquid fuel suspended in air burn in a quite different manner than homogeneous ones. The rate of combustion of a heterogeneous gas mixture is determined in practice by the velocity of diffusive mixing of the fuel with the oxidant, since the chemical reactions of combustion have much higher rates than the mixing processes. This kind of burning is therefore known as diffusive combustion.
"The rate of combustion of liquid fuel droplets is mainly determined by the rate of vaporization (the vapors of fuel formed on the droplet surface diffuse into the air surrounding the droplet and form a combustible mixture at some distance from it)."
The heterogeneous mixtures always develop fuel-rich local zones which serve as centers for ignition of the surrounding leaner mixture. This permits the use of a very lean mixture but greatly increases the potential for harmful detonation and NOx formation.
In a series of tests at the Environmental Testing Corporation in Denver CO (an EPA-recognized testing laboratory), the use of the WHV was shown to reduce the octane requirements of the test car (a 1979 Chevrolet Monte Carlo) by 10 or more points (see Summary of Tests).
In real terms, the octane gains are greater than indicated because they were not offset by negative effects in other areas. Results in all other categories were favorable (low emissions, high torque, high fuel economy, and good driveability). He ability to use 75-octane fuel with no offsetting losses could eliminate the need to boost the octane of gasoline, a practice that is expensive in terms of crude oil requirements, of cost to the end-user, and of deterioration of air quality. The lowest octane fuel that can legally be sold is 85 (available only at high-altitude location). The test data shows that the WHV permits improved performance on fuel 10 octane points lower than that on the cars tested so far; wider applicability will need to be determined by testing on a wide range of vehicles.
As a practical mater, refiners probably would not wish to push octane as low as 75 but would probably prefer to sell the gasoline (once a large enough fleet were established) directly as it comes from the fractionating towers. The octane of this raw gasoline is normally about 82 or 83 (R+M/2), and it would need no further octane enhancement. It is the current effort to boost octane from these levels to the upper 80s and low 90s that consumes so much crude oil, and that adds significantly to the cost of the gasoline at the pump (as much as 20cents/gallon).
The octane reduction observed in the test of the WHV has also been observed in other tests. In its work on fast-burn engines, the General Motors Research Laboratories compared the octane requirements of a fast-burn engine and a slow-burn engine (Ref. 136):
"Comparing these two chambers shows that the fast-burn chamber requires a fuel octane number of only 80 to operate at MBT (minimum advance for best torque). In contrast, the slow-burn chamber needs a fuel with 97.5 octane rating for optimum timing. Alternatively, retarding spark timing to allow an 80 octane number fuel to be used in the slow-burn chamber would reduce efficiency significantly.
"A diagnostic study of combustion in the two chambers identified shorter combustion time in the fast-burn chamber as the dominant factor contributing to its lower octane needs. Specifically, analysis of pressure movement in both chambers showed end-gas temperature and pressure to reach higher values in the fast-burn case, primarily due to its higher energy release rate. Despite this higher temperature, more rapid flame front propagation into the end gas more than compensates."
Mattavi observed that fast-burning of the charge is a fundamental approach for reducing octane requirements. Most of the industry efforts, such as the May "Fireball" concept, have been concentrated on new combustion chamber designs that increase the swirl to reduce the opportunity for detonation pockets to form. All of these systems introduce liquid rather than vaporized gas into the chamber. None of them have yet to be used in large-scale production. The WHV may prove to be a far less expensive and possibly more efficient means of accomplishing the same objective as the fast-burn chambers: reducing octane requirements, improving fuel economy, and lowering emissions of pollutants.
3. Torque
The conditions for maximum efficiency in an automobile engine were established in Paris in 1862 by the originator of the operating cycle theory for the internal combustion engine, Alphonse Beau de Rochas. He determined that the engine should have the following characteristics (Ref. 137):
1. maximum cylinder volume with a minimum cooling surface;
2. maximum rapidity of expansion;
3. maximum ratio of expansion, and
4. maximum pressure of the ignited charge.
The two conditions that are of primary interest here are (2) and (4). Tests to date suggest the likelihood that the WHV, condition the charge so that combustion can occur much more quickly (once ignited by the spark) than in a conventional engine. Because it reduces the octane requirements of the engine (as indicated by its ability to run on low octane fuels with high spark advance and without detonation, it can use lower-octane fuels which burn faster than those with additives such as lead. Because of the faster burn, the expansion of the combustion products is more rapid and exerts more force on the pistons. The high density of the intake charge also increases the available torque because there is a greater mass to expand upon combustion.
Obtaining the maximum pressure of the ignited charge is crucial to achieving the maximum operating efficiency. The maximum work of a cycle and the maximum power and efficiency of an engine can be obtained when the main combustion phase begins and ends almost symmetrically with respect to top-dead-center (actually about 10° after TDC to get leverage on the crank). Because standard engines are not octane tolerant below certain limits, either the octane must be increased or the timing retarded. If the octane is increased, the combustion starts late and takes place over a range of 30 to 40 crank angle degrees. During that time, the volume of the combustion chamber increases as the piston moves downward on its power stroke. The effective compression ratio during most of the combustion, therefore, is lower than it is at TDC. If the timing is retarded (to decrease the pressure on the detonation-prone fuel-rich pockets ahead of the flame front) then the expansion of the gases does not occur until after the volume of the combustion chamber begins to increase as the piston moves away from it.
In both cases, the engine is not operating with maximum efficiency because the maximum available pressure is not being exerted due either to the slow reaction rate or to the late expansion. This is the principal reason that most modern automobile engines have relatively low torque, especially at low engine speeds. At the very beginning of the development of the four-cycle engine, Beau de Rochas theorized that maximum efficiency could be achieved only of the maximum pressure were exerted just after TDC, followed by expansion during the power stroke. Because the WHV permits combustion to occur rapidly without detonation, the timing can be advanced to provide the maximum pressure at a point just past TDC, thus increasing both torque and fuel economy.
The faster heat release and the greater volumetric efficiency made possible through the use of the WHV means that the peak pressures can be higher and can be made to occur at the moment they will be most effective (just after TDC). As a result, the torque has been shown to be as much as 40% higher than standard on 75 octane gasoline, as measured in tests at the Environmental Testing Corporation (see Test 6, Summary of Tests). This was accomplished with better fuel economy (lower brake-specific fuel consumption) than the standard production system.
A gain of that size over standard is substantial, but even more remarkable is that the torque increase began at low rpm. In the Ford tests the torque gain was largest at low engine speeds (1200 rpm) and was maintained throughout most of the test (through 3500 rpm). The difference was greatest at low rpm, however, where torque is needed most.
As Toboldt and Johnson have noted (Ref. 138):
"In the case of the automotive engine, torque is low at low engine speeds and increases rapidly with the speed. Automotive engineers make every effort to increase the torque at low speeds and to remain as nearly constant as possible."
In addition, the WHV apparently eliminates the problem of "manifold lag". Burtner and Morris have described this phenomenon Ref. 139):
"It is well known that octane distribution over the gasoline boiling range affects road octane numbers of loaded gasolines in manual transmission cars. At full throttle and low engine speed, some of the unvaporized gasoline flows along the walls of the intake manifold so that at the beginning of an acceleration, the cylinders are receiving more than the normal share of low-boiling components, Because of this phenomenon, known as "manifold lag", such cars respond to octane quality in the low-boiling "front end" of the gasoline.
"The higher boiling aromatics tend to remain in the "heavy end" of the gasoline and therefore do not contribute to octane quality of the "front end" while increasing the octane number of the whole gasoline."
These octane distribution effects can be important in limiting early torque. In conventional engines the components with the greatest heat content (and the highest boiling points) are the least likely to reach the cylinders as burnable vapor when additional torque is needed. As a result, only the lightest, most volatile components are burned, and these are the ones most likely to cause detonation. Because their heat content is so low they cannot generate as much pressure as a charge with the full range of gasoline components, particularly the heavy ones which are less volatile but which contain most of the heat energy (Ref. 140).
4. Cycle-By-Cycle Combustion Variations
Cycle-by-cycle combustion variations (CBC) have always been a problem in four-cycle engines, but the problem has become even more vexing with the advent of emission controls. CBC occurs under all operating conditions and increases as the air/fuel ratio moves away from a stoichometric value in either a lean or very rich direction (Ref. 141). It occurs to the greatest degree when excessive amounts of exhaust gases are recirculated to reduce the oxygen available for NOx formation (Ref. 142). As exhaust gas recirculation (EGR) increases, the combustion rate slows and the flame stability deteriorates. If CBC variations are too extreme, partial burn occurs and the unburned charge is carried over to the next cycle as a residual gas. Consequently, unburned HC and fuel consumption skyrocket. CBC variation is the random variation in the time between the spark and the establishment of a stable flame front in successive firings (Ref. 143). In an engine running at 2500 rpm and having a typical spark duration of a 20 degree crank angle, the limiting effect of CBC variations is the equivalent of randomly retarding timing between 0 degrees and 20 degrees (Ref. 144). This causes considerable variations in the rate of pressure rise and peak cylinder pressure and greatly reduces efficiency. This degradation of the combustion process limits the maximum air/fuel ratio (currently used to reduce emissions and to lower fuel consumption).
Traditionally, the method used for reducing CBC variations was to operate the engine with a richer mixture but, because of restrictions on exhaust emissions and the need for better fuel economy, this method is no longer practical. The ideal approach is to consistently increase the average combustion rate, as the WHV appears to do. This was not done by the auto industry, however, because faster combustion promoted detonation of the incompletely vaporized and poorly mixed charge. The effect of the fast-burn on CBC variations has been described by Rashidi (Ref. 145):
"Reducing the delay period in the second phase of ignition or reducing the flame stabilization time decreases the cyclic variation. This can be achieved by selecting a mixture with a high reaction rate. In this respect the high temperature of the flame nucleus is important."
With a fast burn, combustion duration declines sharply. With 20% EGR, the combustion duration of a fast burn is virtually the same as that of a conventional engine with no EGR. An improvement in the quality and rate of combustion, therefore, can greatly decrease the CBC variations in cylinder pressure (Ref. 146). The shorter combustion duration also reduces pumping losses (the piston does not have to compress the burning charge as much as the crank approaches TDC) which substantially reduces fuel consumption and lowers NOx emissions. The WHV’s inventors claim that because it conditions the charge to accept fast-burn rates and reduces the need for EGR, it is very effective at improving combustion stability and eliminating CBC variations.
5. Fuel Economy
The thermal efficiency of conventional gasoline-powered engines is very low. Because of te high internal friction in four-cycle engines, the theoretical maximum is only about 25% conversion of the thermal energy mechanical energy (Ref. 147). Virtually no engines achieve these levels because of incomplete combustion, poor mixture distribution, detonation, pumping losses, engine deposits, and other inherent problems. A relatively new car (after break-in) might get as much as 13%, but about 10% is more typical as wear and deposits accumulate (Ref. 148). In an engine far out of tune or badly worn, the efficiency can drop to as low as 6% or less.
The WHV appears to increase the thermal efficiency of the engine in several ways. The better mixture preparation and equal distribution result in more complete combustion and more effective work on the crankshaft. The vaporized state of the fuel and the cooler air temperature reduce detonation (which lowers fuel economy) and improves torque (less fuel is consumed to do the same amount of work). The WHV apparently facilitates the greater use of the heavy components of gasoline which contain most of the energy (Figure 5). When this fuel is burned, much of which would otherwise be lost as part of the exhaust in the form of unburned or partially oxidized hydrocarbons, it provides additional energy that reduces overall fuel requirements. This is consistent with experiments which have shown that a simultaneous reduction in droplet evaporation and entrainment reduces the combustion efficiency (Ref. 149).
Perhaps the most important factor, however, is the increased rate of combustion (fast-burn) which has been shown to improve fuel efficiency by about 10% compared to conventional engines under equal NOx conditions (Ref. 150). Fast burn greatly reduces the heat cycle rejection, which has a direct and significant effect on fuel consumption. The energy within the charge that is transferred to the coolant during the compression, combustion, and expansion phases of the engine cycle when the valves are closed is particularly important because it represents a direct depletion of the energy available for useful work (Ref. 151). The heat losses and related effects reduce the thermal efficiency of a standard engine by about 40% (Ref. 152). If greater use is made of the chemical energy available in the gasoline, then the overall engine efficiency and fuel economy are also increased. The application of this principle, plus the factors already noted, allow the modified engine to operate at a level much closer to the theoretical maximum efficiency.
The high heat losses in conventional engines not only increase fuel consumption directly but also indirectly through increased detonation. This necessitates retardation of the combustion timing to maintain the optimal pressure-versus-crank angle history in each cylinder (Ref. 153). The elimination of cycle heat losses would increase fuel economy by about 12% in most engines, and advancing the timing where possible) would add another 1% for each angle of advance (Ref. 154). Webster-Heise claims that the valve greatly reduces, but does not completely eliminate, these heat losses. This probably accounts, however, for at least some of the fuel economy gains observed in all of the tests,
As Novak and Blumberg point out (Ref. 155):
"The high wall and charge temperatures associated with reduced heat losses would undoubtedly aggravate the already serious problem of engine knock. Hence, any design alternative that reduces in-cylinder heat losses without addressing the knock limitation would be impractical."
They also observed that to prevent deterioration of the combustion process, especially with charge dilution, "would require major advances in fuel preparation, ignition, and controlled turbulence for its implementation" (Ref. 156).
In EPA-recognized laboratory tests, the WHV has shown substantial gains in fuel economy. In the EPA cold-start test and using an early prototype valve, the Webster-Heise modified automobile showed a 9.2% increase in fuel economy (with lower emissions) over baseline. The gains in other official tests were somewhat higher, ranging up to 20% in some. In unofficial road tests and under favorable conditions, even higher gains on the highway have been observed.
It is particularly important to note that in the EPA highway economy tests on October 15,1980, the WH car (1979 Monte Carlo) achieved 25.9 mpg with 75 octane fuel and a timing advance of 13 degrees before TDC (See Summary of Tests). The same car, without the valve, would barely run on that fuel at that timing and had severe knock. Even with 97 indolene (a high octane research gasoline) and the same spark advance, the fuel economy of the baseline car was only 23.8 mpg, 8.1% lower. In the July 1982 tests at ETC on a 1982 Oldsmobile Cutlass Supreme (See Test 6, Summary of Tests), the fuel economy on 75 octane gasoline was 35.614 mpg versus 31.414 mpg for the same car without the valve on 97 octane, an increase of 13.4 %. The EPA highway fuel economy for that model is 30 mpg.
As a general rule, each one degree of spark retardation causes a 1% reduction in fuel economy (Ref. 157). For this reason, the actual fuel economy obtained with the WHV tends to be greater than baseline because of the additional timing advance made possible by the lower octane requirements of the modified engine. The fuel economy of most new cars is less than the EPA numbers would suggest because the timing has to be retarded in order to prevent detonation when using 87-octane gasoline (EPA tests are run on 97-octane gasoline). This reduction in fuel economy can be as much as 8%, but spark retardation is widely practiced by new car dealers to eliminate knock when their customers complain about it in their engines (Ref. 158).
Figure 13 Engine and Fuel Utilization Patterns found for typical road service (Ref. 160)

The greatest potential for fuel savings is probably at low rpm in urban stop-and-go driving (Figure 13). Tests indicate that the greatest fuel economy (possibly as much as 30%) could be expected in that type of driving because it is the mode in which combustion in conventional engines is least efficient. As Knovahk has reported (Ref. 159):
"When the power of an engine is reduced by throttling, the initial and final compression pressure decrease and the dilution of the working mixture with the residual gases increases. First of all, this appreciably impairs ignition of the mixture by a spark and the development of the initial focus of combustion. This prolongs the initial phase, the process of combustion loses its stability and frequently cannot be resumed in some cycles.
"This difficulty can be overcome to a certain extent by so enriching the mixture with considerable throttling as to make spark ignition more dependable (less than 0.8-0.9). But here, too, it is very difficult to avoid combustion continuing during a substantial portion of the expansion stroke, since it is impossible to ensure uninterrupted ignition at large advance angles when the compression pressures are still very low.
"Poor combustion at low loads and the necessity for mixture enrichment are among the principal shortcomings of spark-ignition petrol engines which cause a waste of fuel and the ejection of a large amount of the products of incomplete combustion (including carbon monoxide another poisonous substances) into the atmosphere together with the exhaust gases."
There are two principal reasons for the low combustion efficiency of conventional engines at low engine speeds. The first is the longer time available during each cycle for combustion to occur. When it does take place, it develops unevenly and produces limited torque. The second is the highly restricted volume of air that manages to get by the throttle plate (it is nearly a total restriction when closed at idle). In unmodified engines, the limited air flow at low engine speeds is even less effective than usual in atomizing and partially vaporizing the gasoline. As a result, the standard engine has difficulty idling at speeds below 500 rpm. The WHV, on the other hand, is claimed to maintain a high degree of turbulence, even with heavy throttling, because the fuel is still substantially vaporized rather than atomized in large droplets. This permits a faster combustion rate, even at low engine speeds. Webster-Heise reports that in its test the valve allowed engine speeds as low as 300 rpm without significant deterioration of combustion quality. This means not only that less fuel will be used at complete idle but also that far fewer engine deposits (particularly carbon), which could impair performance later, are likely to form.
This aspect takes on added significance when considered in the context of the EPA tests. The EPA certification test consists of 55% city driving and 45% highway driving. According to the Dept. of Transportation, however, these percentages are not representative of actual driving patterns. DOT reports that 90% of the driving is done in the city and rush-hour driving and only 10% is done at high speeds on highways (Ref. 161). That 90% is where most of the improvement in fuel economy can come because it is the least fuel-efficient mode of driving.
This weighting of the EPA tests inflates the fuel economy numbers to the high side and consequently they are seldom matched in actual experience. One of the main reasons that new car buyers so not obtain the fuel economy promised in the automakers’ advertisements is that the stop-and-go mode of rush-hour driving (over 35% of all driving) is far more important than its representation in the tests. Because this is also the most efficient engine speed at which to operate, the potential gains over standard is much greater than in other modes of driving.
C. Post-Combustion Effects
1. Emissions
The presence of harmful emissions in the exhaust of internal combustion engines has been a source of concern for over two decades. Limited progress has been made in reducing them, but a substantial air quality problem still remains, particularly in urban areas. The efforts of the automobile industry have been directed at devices such as the catalytic converter (which is sensitive to contamination). Efforts to reduce NOx often result in increases in HC and vice versa. The WHV is unusual in that it apparently reduces all three pollutants simultaneously (especially if the manifold is cooled), although not all are reduced to the same extent.
HC emissions, as indicated earlier, are principally the result of incomplete combustion. This is especially true if the engine is run rich (air/fuel ratio less than 14.7:1). Enrichment assures maximum power and stable operation at low loads and idle. The lack of oxygen, however, prevents complete combustion of all of the charge and reduces the evolution of heat upon combustion (Ref. 162). As a result, the products of incomplete combustion (CO, free hydrogen, and others) appear.
A direct relationship exists between knock and HC emissions. For this reason, the reactions that take place ahead of the flame and those that occur behind it are most important. As Bracco observes, "The pre-flame reactions are responsible for the process of engine knock and determine the nature of the unburned hydrocarbons that are emitted when the flame fails to process all of the combustible mixture ahead of it" (Ref. 163).
A major reason for the high concentrations of liquid gasoline in the extremities of the cylinders, which lead to detonation and contribute to high emissions, is the admission of the gasoline to the cylinders in the form of liquid droplets. These droplets, especially the larger ones, tend to collect on the metal surfaces of the cylinders, the cylinder heads, and the pistons. This wetted surface is very difficult to ignite properly because of its thickness and because oxygen can reach it from only one side. Because of this surface wetting, the fuel concentration in the extremities of the cylinders can be quite high even in very lean air/fuel mixtures. When the heat and pressure from the advancing flame front emanating from the spark reaches these areas, the fuel-rich pockets detonate before the flame itself actually reaches them. The WHV, because it apparently delivers a basically homogeneous vaporized charge to the cylinders, greatly limits the potential for the formation of these knock-inducing concentrations.
An effect known as "wall quenching" is also a factor in HC emissions (Ref. 164). This refers to the fact that the temperatures of the metal surfaces of the combustion chamber are lower than that of the combustion itself and tend to extinguish the flame when it reaches them. This normally results in some fuel not being completely burned and in its exhaustion as a pollutant. If quenching occurs early in a cycle, fewer HC molecules are retained by the wall than if it occurs late in the cycle due to the longer post-oxidation time available (Ref. 165). The greater timing advances that have been recorded with the WHV and low-octane gasoline tend to minimize the wall-quenching effect to the extent that it occurs, according to Webster-Heise. Because the charge is apparently vaporized, however, wall-quenching is a relatively unlikely source of exhaust HC, in contrast to standard engines which receive heterogeneous charges that are more prone to this effect (Ref. 166). Higher cylinder pressures, such as those produced in the Webster-Heise modified engines, also reduce the quench distance (Ref. 167).
In the EPA-recognized laboratory tests, the Webster-Heise modified car obtained HC levels that were as much as 13.4% lower than standard (Test 6, Summary of Tests). This pollutant was reduced the least of those tested. It should be noted, however, that the baseline engine had been thoroughly cleaned prior to testing. Therefore, there were no carbon deposits present which probably would have increased the HC levels in the standard engine under normal operating conditions. Conversely, the Webster-Heise test car reportedly did not form carbon deposits even after 80,000 miles, including severe dynamometer tests (See Summary of Tests, Informal Testing). If this proves to be characteristic in general use, the difference in HC levels could be expected to increase over time in favor of the Webster-Heise modification.
Carbon monoxide (CO) emissions are subject to many of the same influences as HC, and they tend to rise and fall inversely, although at somewhat different levels. Retarding the spark increases CO emissions because the airflow must increase to maintain power; advancing the spark lowers CO levels (Ref. 168). CO is a measure of incomplete combustion and is indicative of low droplet vaporization rates within the cylinder, assuming a constant air/fuel ratio. It is also one of the most difficult emissions to control in modern catalytic converters (Ref. 169). If the turbulence of the charge is increased, as it is believed to be in the Webster-Heise system, then the oxygen concentration will be sufficiently high for the chemical reaction to proceed with a high intensity (Ref. 170). The CO is then oxidized to CO2 to a greater degree than in a standard engine. In the ETC tests (Tests 3 and 6), the Webster-Heise modification lowered CO by 19.4 % in the Monte Carlo and 15.4 % in the Oldsmobile Cutlass Supreme, although in one test the decrease was 54 %.
Another difficult emission to control is nitrogen oxide (NOx), which (together with oxidants, HC, and sunlight) is responsible for smog. Because NOx rises when HC is lowered, virtually all emission controls on standard cars are compromises. Because NOx peaks on the lean side of stoichometric (about 16:1 air/fuel ratio), it has proven to be a barrier to improved fuel economy (Ref. 171).
The automobile industry has dealt with the problem of NOx by lowering the spark advance and by using exhaust gas recirculation (EGR) and most recently three-way catalytic converters. A metered amount of exhaust gas is recirculated into the air/fuel mixture on the intake stroke. Because these exhaust gases are deficient in oxygen, they slow down the combustion and absorb heat (Ref. 172). This retarded combustion and shortage of oxygen then reduces temperatures and pressures which, in turn, reduce NOx emissions. Unfortunately, these reductions also increase the HC and CO levels because less oxygen is available for their oxidation and for flame propagation. This can cause misfiring or inhibition of flame development, increase of cycle-by-cycle variations in combustion, deterioration of driveability, increase in HC and CO emissions, and other disadvantages (Ref. 173).
The combustion rate is a very important factor inreducing NOx. As Thring has observed (Ref. 174):
"For given engine operating conditions, there is an optimum rate of combustion for minimum NOx emissions from spark ignited engines. In essence, this is because if the combustion rate is too slow, there is plenty of time for NOx formation, while if it is too fast, charge temperature becomes very high and more NOx is formed. It was considered that rates of combustion higher than those attained in current production spark ignition engines might be desirable, firstly because current rates of combustion were believed to be slower than the optimum for minimum NOx emissions and secondly because previous research had indicated that fast burn improved the lean mixture and EGR tolerances. Also, there were indications that fast burn improved economy."
In conventional engines, a shorter burn time would increase NOx emissions. Most of the total NOx comes from the first part of the charge to be burned, due to its continued heating by the combustion of the rest of the charge (Ref. 175). High turbulence combined with a homogeneous vaporized charge, however, should theoretically provide better distribution within the cylinder and should increase the opportunities for the oxygen to combine with the hydrogen and carbon in preference to the nitrogen. Combined with the faster flame travel, this tends to reduce the time-temperature histories of each element of the gas in the charge (Ref. 176). The faster combustion reduces the time during which the heating takes place and during which NOx can form. Because the temperatures are most intense in the primary flame, NOx can be greatly reduced by accelerating the combustion rate and by lowering the charge temperature. The addition of a slight amount of EGR to the chamber permits the highest efficiency possible while lowering NOx levels (Ref. 177).
The WHV has a greater effect on NOx than on any of the other automobile emissions. The test car showed a 48.6% reduction in NOx over baseline on the Monte Carlo (cold manifold) and 45% on the Oldsmobile Cutlass Supreme (hot manifold) (Tests 3 and 6, Summary of Tests). Most significantly, these NOx reductions were achieved despite a large increase in the spark advance. The NOx reduction probably is due to two factors: (1) the faster combustion rate and (2) the shorter spray flame. Mellor has shown that faster evaporation leads to a shorter spray flame and results in less NOx production (Ref. 178). The faster vaporization observed by Mellor was achieved by increasing the pressure drop through air-assist and pressure atomizing injectors.
2. Driveability
Driveability is a subjective consideration not amenable to quantification. There are, however, several qualitative observations that can be made. As would be expected in an essentially vaporized charge, the cold-start characteristics of an engine are greatly improved with the use of the WHV. Because the automobile uses a weak choke to obtain satisfactory HC emission levels in the cold-start tests, most new cars are very difficult to start when cold (the low vaporization rate of the gasoline is not sufficiently offset by fuel enrichment because it would increase HC emissions). As a result, the standard engine is provided with a choke to restrict the amount of air going into the cold engine so that the small amount of fuel that does evaporate will comprise a large enough part of the total air/fuel mixture to allow combustion to occur.
Harrow has pointed out that all of the mixture problems normally present in a fully warmed-up engine are exaggerated during a cold start for the following reasons (Ref. 179):
1. The manifold has to handle relatively large amounts of liquid;
2. Because of the low engine speed, the gas velocities through the carburetor are low so that mixing is even poorer than usual;
3. Again because of the low cranking speed, there is little manifold depression to encourage fuel evaporation. A consequence of this is that very rich mixtures must be used for starting and cold running, and these rich mixtures have a serious effect on the engine’s fuel economy.
Repeated tests of the Webster-Heise equipped engines, however, have shown no cold-start problems. Webster-Heise claims that this is because the vaporization rate is very high initially, even at ambient temperatures. Because enrichment is unnecessary with the WHV, cold-start emissions are very low (see Summary of Tests).
Because the torque is greater than standard, as discussed in the section on torque, performance is improved over baseline. One of the most chronic complaints about new domestic cars being built today is that they have very limited power. Although some specialty cars forego fuel economy in favor of high compression ratios and lower gearing to regain better performance, most manufacturers seem to have accepted the loss of performance as a necessity to retain fuel economy gains. The WHV makes possible increases in both torque and fuel economy. During the ETC tests, the Webster-Heise car (on 75-octane fuel) was noted by the certified test driver as having consistently better driveability than the baseline car (see Summary of Tests).
Another characteristic observed in the tests is the resistance of the WHV to engine stalling and flooding. Because the WHV forces the gasoline to change state at low manifold temperatures, the charge is in a more combustible state for ignition in the combustion chamber. As a result, the air/fuel ratio does not have to be as rich as standard (because much more of the gasoline is in vapor form). Air/fuel ratios of 1:1 or 2:1 are normally needed to provide a reliable cold start in conventional engines (Ref. 180). The reason most accelerators have to be pumped to start conventional cars is to force so much liquid into the manifold that enough gasoline vapor will evolve to permit stable combustion, although this greatly increases HC emissions.
Improvement in combustion stability at idle has been observed as a characteristic of the WHV. Because the time available for combustion increases with a decrease in engine speed, the already slow combustion in standard engines is reduced to the point (around 500 rpm) where flame instability causes roughness and eventual stalling. Because the WHV appears to accelerate combustion, the idle limit can be lowered to a much lower speed (around 300 rpm) before instability appears. This could be especially important in reducing HC emissions and in increasing fuel economy in city and stop-and-go driving.
3. Engine Maintenance
There are a variety of problems which plaque the standard engine and which can interfere with continued operation or, if severe enough, can cause engine failure. As discussed earlier, detonation is a most serious problem and is a common one in modern automobiles. Knock can cause serious damage to engine parts as well as limiting performance. Use of the WHV, as indicated in the test data in the Summary of Tests, appears to be even more effective than chemical additives in reducing this problem.
Removal of the additives from gasoline, especially tetraethyl lead (which is not combustible), can greatly reduce the amount of engine deposits formed and the contamination of catalytic converters. The use of lead-free gasoline, for example, greatly reduces combustion chamber deposits which, if not removed, can double the HC emissions with mileage (Ref. 181). Spark-plug fouling from lead can also sharply reduce fuel economy. Removing lead, however, can cause other deposits to form in a standard engine. The aromatics and other octane enhancers that are substituted for lead tend to form carbon deposits more readily than leaded gasolines. This eventually causes engine octane requirements to increase. Webster-Heise claims that its valve eliminates the need for either type of additive and greatly reduces the formation of engine deposits when additives are used.
The vaporization of the fuel itself is an important factor in reducing engine deposits. The part of the fuel that is not vaporized and burned will accumulate and form sludge, varnish, and other harmful deposits in the engine (Ref. 182). These carbonaceous deposits are abrasive and greatly contribute to engine wear, especially if the unvaporized gasoline dilutes the lubricating oils. It was observed during testing of the Webster-Heise modified car that the crankcase oil stayed clean, even after extensive operation, indicating an absence of carbon deposits in the cylinders. Webster-Heise also reported that no oil was consumed or needed to be added, other than for infrequent oil changes. When the engine was disassembled after 80,000 miles of testing, not only were there no engine deposits found but the expected cylinder wear of 0.0100 to 0.0250 inch was found to be only 0.0025 which is within the normal factory tolerance (See Informal Testing, Summary of Tests. This represents a reduction in normal wear of between 75 and 83 %,. There was no appreciable crankshaft wear.
4. Catalytic Converter Operation
A long-standing concern in automotive pollution control has been the need to maintain exhaust temperatures high enough to "light off" unburned HCs in the exhaust. The WHV reduces the amount of HC left after combustion but, because of its higher thermal efficiency (less heat is transferred to the coolant because of the faster burn), it also produces exhaust temperatures that are somewhat higher (about 4%) than standard. This makes the catalytic converter more effective and extends its useful life because the volume of pollutants that it must treat is lower. This is also supported by the fact that the engine coolant temperature is lower than standard and exhaust temperatures are higher, indicating greater thermal efficiencies.