
rexresearch.com
The Technology of Low Temperature Carbonization
by
Frank M. Gentry
Table of Contents
Preface & Table of
Contents
Chapter I ~ Fundamentals
Chapter II ~ Low Temperature
Coal Gas
Chapter III ~ Low Temperature
Coal Tar
Chapter IV ~ Low Temperature Coke
Chapter V ~ Nitrogenous &
Other By-Products
Chapter VI ~ Processes of Low
Temperature Carbonization
Chapter VII ~ Operation, Design,
& Materials of Construction
Chapter VIII ~ Economics &
Conclusion
Bibliography
Name Index
Subject Index
High & Low Temperature Coke
Semi-Coke
Porosity
Coherency
Swelling
Preheating & Oxidation
Coke Reactivity
Coke Combustibility
Coke Strength
Temperature Effect
Semi-Coke Uses
Domestic Fuel
Power Char
[ Note: The quality of the scanned graphics (tables & figures) are "uneven" at best despite repeated efforts to scan and tweak the images. Enough is, so here it is anyway: I've had enough and done enough for the time being. ~ R.N. ]
Agitation for a smokeless fuel, which would eradicate the civil nuisance of smoke, together with the shortage of oil in some countries of the world have been largely responsible for the development of low temperature carbonization. Added to this incentive was the shortage of hard coals during the Great War and the possibility of obtaining a substitute in the form of an easily burning coke. Although some small amount of information was in hand at that time, it was soon realized that insufficient data were available for undertaking large-scale operations in coking at low temperatures. Research was the obvious remedy and, in recent years, a great many investigations have been made in this field both in the USA and abroad.
In high temperature coking such as occurs in the metallurgical and gas industries, only a very small percentage of volatile matter remains in the coal after carbonization, so that from 64% to 74% by weight of the charge is converted into coke. Low temperature distillation, on the other hand, does not entirely remove the volatile constituents from the coal, and therefore the percentage yield by weight of coke may be expected to be somewhat larger than in the case of gas and metallurgical cokes. Low temperature ovens yield about 66% to 76% of the charge as coke. The proximate and ultimate analyses of the three types of coke given in Table 73 furnish an excellent basis for comparison.
The outstanding peculiarity of the low temperature carbonization residuum, from a chemical standpoint, is the large proportion of volatile matter remaining in the coke. To this fact must be attributed many, though not all, of the desirable properties of this material as a fuel. As a consequence of the high volatile content, we find a somewhat greater thermal value in the coke which has been subjected to less extreme measures of carbonization. The semi-coke, or low-temperature coke, whose analysis is given in Table 73, represents the average of that obtained from 6 full-scale retorts. The large percentage of hydrogen recorded for the semi-coke, as compared with the cokes from other sources, is present in the form of volatile hydrocarbons. Low temperature coke also is characterized by the presence of considerable oxygen. The experimenters make no comment on this peculiarity in their reports, but doubtlessly it arises form the existence of volatile oxygenated hydrocarbons such as the organic alcohols and possibly the aldehydes and ketones in small quantities. It might be pointed out at this time that the nitrogen content of low temperature coke is greater than that of the high temperature varieties. This fact will be taken up under the subject of ammonia and nitrogen recovery. How nearly the problem of anthracizing bituminous coal is solved by the low temperature process of carbonization is demonstrated by comparing Table 1 and Table 2 with Table 73. It will be seen that semi-coke has a proximate analysis almost identical with that of anthracite and that the ultimate analyses are not greatly at variance.

In their experiments on the coking of Illinois coal, Parr and Olin (31) obtained a coke yield of 77.5% for 3-hour periods of distillation and 72.5% for 6-hour periods. This represents 1,550 pounds and 1450 pounds of semi-coke per net ton of coal, respectively. By recalling that at a given temperature of carbonization the volume of gas evolved is a function of the duration of distillation, as shown in Figure 14, it will be seen that the decrease in coke yield, resulting from extension of the retorting period arises from the removal of more of the volatile matter.
Pennsylvania coke, coked by Davis and Parry (97), produced an average of 72.2% ash, or 1,444 pounds, of semi-coke per net ton of coal. Their yields ranged from 70.4% to 74.8%. On the other hand, Parr and Layng (101), from 12 experiments on Utah coals, similar in analysis to that of bituminous coal given in Table 1 and Table 2, obtained an average of 61.2%, or 1,223 pounds of semi-coke per net ton of charge. For the average yield of semi-coke, however, this figure is exceedingly low.
Table 74 gives the average proximate analyses of semi-cokes obtained from various bituminous coals of the USA. While it is recognized that these tests are for different coals, certain deductions can be made with regard to the influence of temperature. It will be observed that the percentage of fixed carbon, for example, increases as the temperature of distillation advances. Such a change is to be expected since the higher the temperature, the less volatile matte remains in the residuum. Monett (100) also distilled Utah coals at 550° C. His yields of coke ranged from 1464 pounds to 1250 pounds per net ton, representing from 73.2% of the charge to 62.5%. The semi-coke had a thermal value ranging from 13,56 BTU to 12,227 BTU per pound and it contained from 0.86% to 1.93% moisture; 19.84% to 10.46% volatile matter; 81.67% to 64.35% fixed carbon; 14.81% to 5.94% ash; and 0.40% to 1.09% sulfur. Analyses of these Utah coals have been given in Table 15 and Table 16, while the composition of the low temperature gas obtained from them was discussed under the subject of American coals in Chapter II.


Table 75 shows the temperature variation of the constituents of Farmville NC coal which originally contained 37.08% volatile matter, 52.95% fixed carbon, 9.03% ash, and which had a heating value of 14,336 BTU/lb as determined by Giles and Vilbrandt (99). We have already seen in Figure 12 the manner in which temperature influences the composition of gas evolved from this coal and in Table 58 its influence on the volatility of the low temperature tar. It must be said that this particular coal is an exception and the percentage of volatiles remaining in the coke at a given temperature is of the order of that to be found in an average semi-coke at approximately 200° C higher temperature. There is a decided change at 540° C, accompanied by a voluminous evolution of gas. Below that temperature, the change in the coke structure is uniform but slight, while above 540° C to as far as 660° C there is little apparent change in the residuum. The heating value of the coke is high. It has been pointed out that Farmville NC coal decomposes very easily, so that only 8.95% volatile matter is left at 300° C. Above 540° C, where the major decomposition occurs, less than 1% volatile matter remains in the coke.
In Table 29 and Table 59, the composition of the gas and the volatility of the tar from Washington lignite have already been discussed. Benson and Canfield (108) also determined in Table 76 the effect of temperature variation on the proximate analysis of the char. Originally the air-dried lignite contained 39.4% volatile matter, which was reduced to 4.1% at 600° C. The percentage ash varies inversely as the percentage of volatile matter remaining within the residuum. Up to 500° C, the lignite char was dull black and it more or less retained its original form. From 500° C to 600° C the lumps had a metallic luster and swelled slightly larger than the initial size.



It has been pointed out before that the Fuel Research Board (106) carbonized peat in vertical retorts, obtaining gas of the composition shown in Table 27 and tar of the character given in Table 60. Both proximate and ultimate analyses of the peat char, obtained from these experiments, are given in Table 77. The principal point to be noted in the carbonization of peat is the increase in calorific value by removal of the large amount of moisture present. North and Garbe (121) report a test on carbonizing Shetland peat at 500° C in Tozer retorts. The raw peat contained initially 21.4% moisture and 73.8% volatile matter, yielding on distillation char to the extent of 33.6% of the original charge and containing 18.6% volatile matter, 71.4% fixed carbon, and 9.9% ash.
In Table 78 are given the analyses of 6 cokes form full-scale low temperature processes. The experiments of the Fuel Research Board (114) were carried out in their horizontal retorts, while those on Coalite (187) were in that company's vertical retorts. Coke from the Maclaurin (188) retort was carbonized by internal heating through the agency of the sensible heat of producer gas. Carbocoal (189) was produced through primary distillation, briquetting, and subsequent secondary carbonization in an inclined retort. The semi-coke from the Nielsen (19) process was produced by carbonizing the coal in a rotary retort by means of the sensible heat from producer gas. The Tozer (190) process utilized a static cast-iron vertical retort.
A great deal of the ease in combustibility of semi-coke depends on its high volatile content, but the coke structure also has a great influence. One of the main factors, from a physical viewpoint, is the area available for oxidation. Some of the desirable features, however, of low temperature coke can be attained only by allowing a certain proportion of the volatile matter to remain; notable among these is a low ignition temperature.
When coal is coked, it first reaches a fusion temperature and becomes plastic. Then, as the temperature is raised, gases are evolved and form bubbles throughout the mass until it becomes a foam. These bubbles then break, allowing the gases to escape. Rigidity finally sets in upon cooling and leaves the cellular structure of the coke. If the plastic material is not confined by walls, the bubbles swell the mass and cause it to become light and porous. But the pressure developed against retaining walls compresses the material to form a dense coke. Upon consideration of combustibility, it would appear that the more numerous and hence the smaller the pores, the greater is the area exposed for oxidation, and therefore the more easily the coke should burn. A point is reached, however, where the outside surface of the lump exposed to oxidation is so reduced by the pores as to neutralize the result. Consequently a point of apparent porosity is reached where there is maximum combustibility. We shall see later that combustibility depends more on the microscopic cells of the coke than on the large pores.
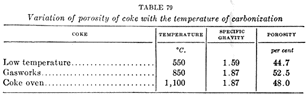
Table 79 shows that different temperatures of carbonization produce coke of different porosities. The determinations were made by Cobb and Greenwood (191), who used a coal with 1.27 initial specific gravity. From this table, it is evident that low temperature coke is less porous than either gasworks or coke oven coke. We shall see later that nevertheless, semi-coke is far more reactive and combustible than either of the others for several reasons, but primarily because of a microscopic cellular structure.
Coherency depends upon the presence of sufficient resinous bodies to act as binding material. In their absence, various methods have been resorted to in order to secure a coherent carbonization residuum. One method of frequent use is the blending of coking and non-coking coals so that the resins present in the former will serve to bind the entire mass. Non-coking bituminous coals give a coke which is very friable and unsuitable for transportation, while coking coals cause trouble in the design of low temperature retorts. A blended charge yields a residuum upon distillation that is sufficiently strong, from the mechanical standpoint, and in a large part free of the retort design annoyances. It occasionally happens that a good coke is obtained without blending, solely by using the product of the seam as a whole, but as this situation is not a common occurrence, efforts have been made to provide means for coking the non-coking coals without resort to binding, in order to save transportation charges.
No indication whatever of a coal's coking qualities can be determined from its proximate analysis, for it depends entirely on the proportion of resinous material present. There are two types of coals, the swelling and the non-swelling, depending on whether or not the volume of the resultant coke is greater than or equal to the initial volume of the coal. The coke residuum of non-swelling coals is usually non-coherent and friable, while the swelling coals form a good coke upon carbonization. For this reason, these two classes of coals are often classed as non-coking and coking.
If the resins are in excess, other difficulties arise in the possibility of foaming, and here again a number of remedies have been proposed. Cruickshanks in 1841 is said to have been the first to propose the blending of coking and non-coking coals, so that the resinous deficiencies of the one can be balanced against the excesses of the other. This method is successful, but suffers the economic disadvantage of transportation. To avoid this objection, Roberts (192) suggested that coke breeze be blended with the retort charge. This proposal, however, reduces the quantity of virgin coal that is carbonized, and hence the output of the plant.
Parr and Olin (31) found that the texture of the coke could be altered by means of exerting pressure during carbonization. When pistons were used for this purpose, however, the outer layers of the charge, which had undergone fusion, hardened and formed a shell surrounding the inner mass, which later became excessively porous due to the inability of the gases to escape. Efforts to overcome this by use of additional pressure resulted in fracture of the hardened shell and the coke was withdrawn from the retort in small broken pieces, They then resorted to ramming the charge into the retort and applying the pressure entirely before carbonization with decidedly more success. The semi-coke thus produced has a very good crushing strength and fine texture.
Taylor and Porter (96) found that bituminous coal, which gave a friable coke at atmospheric pressures, even when slowly heated, gave a dense coke when carbonized in vacuo at a pressure of about 3 cm Hg. This is probably due to the fact that the gases evolved are sucked away before large bubbles can form in the fused mass.
When coals contain a large proportion of resinous bodies, swelling occurs during coking. When first heated, these coals slightly contract. This probably results from the melting of certain constituents between 350° C and 450° C. A secondary contraction usually develops later at much higher temperatures, during coking proper. This is attributed to the liberation of gaseous products. Between the two contraction periods, the coke expands as the result of entrapped bubbles in the plastic mass. Expansion is rather slow at first until the center of the charge reaches a given temperature, at which point expansion increases enormously. This swelling is usually greater than the initial contraction and develops a pressure which is liable to injure the retort, or at least to render discharge difficult. A great many of the past failures encountered in low temperature retorts may be attributed directly or indirectly to this phenomenon.
Audibert and Delmas (193) studied the intumescence of coal under various conditions of time, temperature, and rate of heating. Their method of determination was to place the finely ground coal in a heated metal cylinder equipped with a counterpoised piston to transmit expansion and contraction of the charge to an indicating pointer. Figure 31 shows the swelling of Loire coal as a function of the time of heating, for various constant temperatures. This is a most interesting diagram. It discloses that at such low temperatures as 390° C there is no swelling of the charge at all, but an actual shrinkage amounting to about 11%. At 400° C there is an initial contraction during fusion, amounting to as much as 20%, at the end of 35 minutes heating, after which the coal expands and finally, after 75 minutes heating, reaches a steady volume about 6% greater than its initial condition. As the temperature advances, the initial contraction, which always takes place at fusion, reaches a maximum at some temperature which is characteristic of the given coal. In the case of Loire coal, the initial contraction amounts to only about 6% at the end of approximately 10 minutes, after which the coal swells rapidly to an increase of 75% over its initial volume in 25 minutes, thereafter continuing to swell at a very slow rate. In this particular coal the swelling amounted to 95% at the end of 150 minutes.
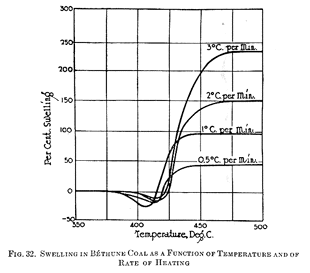
Figure 32 shows for Bethume coal the variations of swelling as a function of temperature for several different rates of heating. It is interesting to note how rapidly the swelling increases with the rate of heating when the same final temperatures are attained. This family of curves has a common characteristic, in that there is an initial period of contraction during fusion amounting to as much as 20% of the original volume at 400° C to 425° C, following which there is a rapid expansion at 425° C to 450° C which ceases rather abruptly and leaves the charge at constant volume. The final swelling of Bethume coal amounts to about 47% at 440° C, when heated at 0.5° C per minute; 95% at 440° C, when heated at 1° C per minute; 150% at 475° C when heated at 2° C per minute; and about 225% at 475° C when heated at the rate of 3° C per minute. Audibert and Delmas (193) concluded that for each coal there existed a rate of heating below which intumescence is not observed, whatever the final temperature reached may be. However, the rate of heating may be so low as to render it impractical in practice to sue this means of avoiding excessive swelling. Swelling is observed only at rates of heating in which fusion of the coal particles is complete, i.e., when the coal becomes fused into a homogenous mass, and intumescence never occurs when the rate of heating is so low that the charge retains the heterogenous structure of coal particles in juxtaposition.
Figure 33 shows the loss of weight of the charge, due to the evolution of volatile matter at various temperatures for a Durham coal, together with the intumescence at the same temperature. It is quite apparent that swelling, at the same rate of heating, always occurs at a higher temperature than that of initial softening and initial evolution of volatile matter. The presence of microscopic cavities in the charge after heating led Audibert and Delmas (193) to conclude that swelling was only an apparent change and one which arose, not through changes in the condition of the solid material, but through the imprisonment of bubbles of volatile matter trapped within the plastic mass during thermal decomposition.

Some coals, which in their neutral state will not coke, can be made to do so by special thermal treatment to remove some of their volatile matter, according to Charpy and Godchot (194). That is, by subjecting the coal to preliminary distillation at 450° C and by subsequent carbonization at 700° C, it can be made to coke. These experiments were made on Durham coal, which would not coke in its natural state under ordinary conditions. From Table 80, it is seen that a good coke was obtained after 2 hours of preliminary carbonization at 450° C when the volatile matter of the coke had been reduced to 18%. Primary distillation, as a means of inducing coking, has also been studied by Illingworth (195), whose results confirm what has already been said.


Audibert and Delmas (193) investigated the effect of preheating and partial oxidation on intumescence in coal during carbonization. Figure 34 shows the swelling of Varennes coal as a function of the temperature for various periods of preheating in vacuo at 350° C, and when finally heated to the required temperature at the rate of 1° C per minute. This temperature of preheating is within 15° C of the softening temperature of this coal at the particular rate of heating used. In every case, no matter how long the preheating, the initial contraction of the charge during fusion apparently remains. Varennes coal, when untreated, swelled about 65% at 465° C, but when heated for 4 hours in vacuo the swelling property was entirely destroyed and the residuum at 525° C had practically the same volume as the original coal. When preheating was prolonged to 4.5 hours, there was a resultant contraction of about 22% at the final temperature. The influence of preheating is attributed to the fact that, even at temperatures below the fusion point, carbonization reactions must have an appreciable velocity and that such chemical changes as occur within the coal, upon preheating under the above conditions, are not usually accompanied by loss in weight to any extent.
Destruction of a portion of the excess resin by oxidation is another means which may be employed to effect coking in certain coals. Bone (195) showed that the resins could be completely destroyed by prolonged oxygenation, even at as low a temperature as 108° C. Roberts (196) found that by heating a non-coking coal for a very long time at this temperature it could be made to coke. Here it is not clear whether the effect is one of oxidation or evolution of the undesirable constituents. At any rate, this method does not promise great commercial favor because of the length of time required in the operation, unless carried out at higher temperatures.
Figure 35 shows the swelling, according to Audibert and Delmas (193) in Varennes coal as a function of the temperature for various periods of oxidation. The oxidation was carried out at 120° C in air and subsequently heated to the required temperature at the rate of 1 C per minute. Preoxidation, even for short periods, exerts a considerable effect. The swelling at 500° C is reduced from about 65% in the unoxidized coal to 50% after 10 minutes oxidation, and to about 10% after 40 minutes oxidation. Oxidation for 4 hours causes a contraction to the extent of about 38%, while 5 hours oxidation entirely destroys the fusing property of the coal. The influence of oxidation on the intumescence of coal is attributed to the fact that oxygen fixation transforms some of the coal constituents into more refractory substances.


Parr (87) separated the lignitic and bituminic portions of the coal by extracting the latter with xylene and then heated both the fresh and oxidized materials at various temperatures to determine the volume and character of the gas yield. In Figure 36 is shown the volume of carbon dioxide evolved from both the fresh and the oxidized lignitic residuum when heated at various temperatures up to 350° C. the same data for the bituminic extract are given in Figure 37. Table 81 gives the composition for all four gases collected up to 300° C. A study of this table, in connection with the two preceding diagrams, shows the striking difference between the behavior of the fresh and the oxidized lignitic material, as compared with the fresh and oxidized bituminic material, when all are heated to the same temperature. Up to 350° C, little carbon dioxide is ordinarily evolved from the fresh lignitic residuum, but this amount is increased over 6-fold when the material has been oxidized. On the other hand, oxidation has very little effect on the quantity of carbon dioxide evolved from the bituminic extract up to 350° C. The gas analyses show a reduction in hydrogen content after oxidation of the material in both cases, but a much greater reduction in the case of the lignitic residuum than in the sample of a bituminic extract. The same observation applies also to methane. It is quite evident from these experiments that the reduction of coking power by oxidation can be attributed primarily to changes in the constitution of the lignitic material of the coal.


A number of different experimenters have sought to devise a means for measuring the reactivity of coke, thus Koppers (197, 198) sought to measure its reactivity by the rate at which a particular coke reduced carbon dioxide to the monoxide; Bunte (199, 200) assumed coke reactivity to be proportional to the temperature at which it begins to react with oxygen; Fischer, Breuer, and Broche (201) considered the reactivity to be inversely proportional to the temperature at which the coke begins to reduce dioxide to the monoxide of carbon; while Bahr (202) determined the reactivity of coke by measuring both the kindling point of the coke and temperature of reduction in an atmosphere of carbon dioxide. Bahr (202) observed that the kindling temperature of carbonaceous materials ranged from 230° C, with low temperature coke, to 140° C with electrode carbon; that the reaction temperature varied from 480° C with charcoal to 820° C with graphite; and that the reactivity ranged from 99% for charcoal to 4.4% with graphite. The addition of 3% iron oxide was found to increase the reactivity of 950° C coke from 17.5% to 58.4%, at the same time affecting neither the kindling temperature nor the reaction temperature to any extent.
Davis and Greene (203) investigated the reactivity of pulverized cokes in air, carbon dioxide, and water vapor at 950° C. Table 82 gives analyses of the cokes which were tested, together with the experimental determinations. The low temperature coke was made by heating coal for 30 minutes at 750° C with superheated steam. The beehive coke was dense and hard in structure, whereas the low temperature coke was soft and porous. Before submitting the coke samples to test, the specimens were crushed and sized to pass a 10 mesh and to remain on a 20 mesh sieve. In Table 82 the coke reactivity in air is expressed as the percentage of the theoretical maximum amount of carbon gasified, the reactivity in carbon dioxide is reported as the percentage of carbon dioxide reduced to the monoxide, and the reactivity in steam is given as the sum of the percentages of hydrogen and carbon monoxide. The kindling temperature was taken as that at which carbon dioxide was first formed. The results indicate that the kindling temperature of low temperature coke is less than half of that of the other cokes, that the reactivity of low temperature coke is 11% to 14% greater than the other cokes in an atmosphere of air, nearly 950% greater in carbon dioxide, and finally 110% to 190% greater in water vapor.

Coke may burn either from the external surface or externally and partially internally, depending on its structure. Semi-coke, obtained from horizontal retorts at 600° C, burns on the surface and for some distance into the interior and makes a very attractive fire. On the other hand, low temperature coke made by fusion, briquetting, and secondary carbonization burns mainly at the surface and deposits thereon a fine ash which must be brushed away frequently to maintain a nice looking fire. These are factors to be considered when semi-coke is used as a domestic fuel.
Kreisinger, Ovitz, and Augustine (204) investigated the combustibility of various fuels in hand-fired furnaces when burned at approximately 25 lb/sq ft/hr, by determining the mean percentages of carbon monoxide and of free oxygen for various distances above the grate. Some of their results are shown in Figure 38, from which it is apparent that metallurgical coke is the least combustible and lignite char the most combustible of the fuels studied. Low temperature coke strongly resembles lignite char, from which we may conclude that its combustibility in furnaces will greatly exceed that of bituminous coal and in no way resemble the behavior of ordinary cokes.
Thau (205) has pointed out the common error of assuming that combustibility increases with porosity, having in mind principally those pores which are visible to the naked eye and not those of microscopic character, which have been shown elsewhere to contribute greatly to combustion. Combustibility depends on two characteristics of the coke, structure and volatile matter. While in ordinary coking practice it is true, within limits, that the denser coke is the more combustible, at the same time, when judging the reactivity of a coke on this property, its purity must also be considered. The ash content of the coke plays a much greater part in reducing combustibility than is ordinarily believed. It is quite impossible to judge a coke by its structural density without a knowledge of its chemical composition. It is common experience that often a spongy coke is less combustible despite its apparent porosity, than a more consolidated sample; the reason for this peculiar situation being that combustion is a surface phenomenon and it is the reactivity of the multitude of microscopic cells that is of importance rather than the large pores.
Thau (205) also points out that the general belief that silvery appearance of coke, caused by a graphitic deposit, is wholly a desirable condition is erroneous. While the graphitic deposit adds greater strength to the coke, when used for some purposes where it has to sustain a great burden, at the same time the vitreous carbon coating is less reactive in two ways, first by its own inactivity, and second because it plugs the highly reactive surface cells. Thau (205) is quite convinced that the combustibility of low temperature coke is wholly due to its cell structure, which is maintained by the presence of a small amount of volatile, which volatile is driven off as combustion proceeds to leave additional minute cells which are free from vitreous carbon and are highly reactive. The volatile matter present in low temperature coke serves principally to reduce the coke's ignition temperature.
On account of its high combustibility, should it be possible to strengthen low temperature coke in some manner, so that it could withstand the burden in metallurgical operations, it would be of great industrial importance. A number of attempts have been made to do this, with more or less indifferent success, by applying pressure to the charge during or after carbonization.

The strength required of a coke varies greatly with the purpose for which it is produced. For use in blast furnaces and cupolas, where the weight of the burden which it must sustain is tremendous, a high crushing strength is necessary. The strength of a coke depends upon its structure and a strong structure usually requires high temperatures with the resultant deposition of graphitic carbon. Thus, despite the greater reactivity of low temperature coke, the superior strength of the high temperature product gives the latter favor in metallurgical processes.
A variety of different schemes have been tried to increase the strength of semi-coke, all of which are variations or combinations of three methods, application of pressure, blending of the charge, and temperature regulation. In the use of pressure to this end, recourse has been made to its application during carbonization by mechanical means or by the weight of the supernatant charge, and to briquetting before, after, or between stages of carbonization. Some little success has been attained by blending different coals to reduce foaming in the fusing stage, but a more fruitful effort has been made in blending a certain amount of coke breeze with the raw coal, Record has been made of a case where the crushing load on a piece of coke, less than one square inch in cross-section, was increased from 250 lb when 20% coke breeze was mixed, to 425 lb when 35% coke breeze was blended. The addition of breeze, however, is open to the objection that it reduces the daily throughput of raw coal.
Audibert and Delmas (193) determined the influence of rate of heating on the crushing strength of coke produced from a number of different coals, the data regarding two of them being reproduced in Figure 39. These cokes were made at 900° C. Experiments by Audibert and Delmas (103) indicated that all coals examined by them attained a maximum strength at a given rate of heating, rising rapidly to this maximum as the rate of heating increases and declining slowly from the maximum as the rate of heating is further increased.

Davis and Parry (97) found that the introduction of steam during carbonization at 550° C has no appreciable influence on the coke yield. They obtained 70.7% of the coal as coke when it was dry distilled and 70.8% when large quantities of steam were admitted. Variation of temperature, however, under steam distillation does have considerable influence on the amount of coke obtained. Figure 40 shows the coke yield obtained from steam carbonization at various temperatures, together with the proximate analyses of the cokes produced from Pennsylvania coal of the Freeport bed. The ultimate analyses and calorific value of this low temperature coke, carbonized at three different temperatures, are given in Table 83. The yield of coke gradually decreases with rise of temperature for two reasons. In the first place, a greater percentage of the volatile constituents is removed, which reduces the weight of the yield. This is confirmed by the decrease in the proportion of volatile matter remaining in the coke. In the second place, it has been pointed out that steam reacts with the coal to form the dioxide of carbon at approximately 550° C. Consequently, the yield of coke would be decreased by the percentage of carbon dioxide generated. That this is a factor in reducing the amount of coke obtained is verified by the fact that the percentage fixed carbon in the coke attains a maximum at about 550° C, after which it decreases on account of gasification.
From Table 83, temperature is seen to have no great effect on the ultimate analysis of the coke, other than its influence on the percentage of carbon. The reduction in oxygen and hydrogen content is due to removal of the phenolic derivatives and volatile hydrocarbons. The coke produced under steam distillation at 475° C was black and spongy. Considerable swelling had occurred and the residuum was filled with blowholes. The carbonized product from distillation at 550° C was of much denser structure. The layer next to the retort wall was rather porous, but of a grayish color, indicating more complete carbonization. The action of the steam was evidently responsible for the external porosity. The coke obtained at 650° C was practically the same as that produced at 550° C, except that it was slightly lighter in color.

As a whole, semi-coke is fairly dense and of good texture, although some of it is quite friable. Samples tested y the US Bureau of Mines (101) indicate that it is adapted to the usual screening and sizing processes for preparation as a domestic fuel. The coke produced, however, varies very greatly with the raw coal used, but there is no doubt that a few of the low temperature carbonization residuums are suitable for metallurgical purposes. The high percentage of volatile hydrocarbons remaining in the low temperature coke supplies a free-burning constituent, which is readily ignited and which will maintain fire without special attention. In this respect, the semi-coke is superior to anthracite. The percentage of smoke-producing elements, consisting of tar and heavy hydrocarbons, which can be removed from American coals at various temperatures of carbonization, as determined by Porter and Ovitz (96), is given in Table 84. It has been pointed out that one of the purposes of low temperature processes was to produce a smokeless fuel, and how nearly this purpose has been attained is made evident by the table. The semi-coke obtained as a carbonization residuum is an ideal smokeless fuel.

Parr and Olin (31) conducted some experiments on semi-coke gasification using a small Otto gas producer equipped with a wet scrubber, the results of which are given in Table 85. This producer was designed to operate on pea size anthracite coal. Using anthracite, the normal capacity was 4500 cubic feet of gas per hour. The semi-coke which they used as a fuel was obtained from carbonizing of Illinois coal at 550° C. It was light and porous and showed no tendency to pack on the fuel bed. Analysis showed it to contain 18% volatile matter and 71% fixed carbon. The residuary gases evolved from the secondary carbonization of this primary coke were remarkably free from heavy condensation products. This suggested that producer gas made by the gasification of semi-coke would be sufficiently free from tar to render it suitable as a fuel for internal combustion engines. For comparative purposes, data obtained from the operation of the same producer, using a Pennsylvania anthracite, as determined by Garland and Kratz (206) are also given in Table 85.

The great increase in efficiency attending the use of low temperature coke as a gas producer fuel is the notable feature of Table 85. The higher terminal value of the gas from semi-coke is shown by the analysis to be the result of a low percentage of diluents, such as carbon dioxide, nitrogen, and oxygen, in the gas. Little tar was produced during the operation of the producer with semi-coke as a fuel. As a whole, the tests were entirely successful and little attention was required during the runs. The lightness of the low temperature coke and its slight tendency to arch made it necessary to poke the charge about once an hour and, in this respect, it required somewhat more attention than anthracite. However, this semi-coke contained more volatile matter than the usual product, which fully accounts for such minor difficulties as were experienced. There was remarkable freedom from ash and clinker trouble.
Lander and McKay (186) reported some tests conducted at the Fuel Research Station in a small gas producer to determine the suitability of low temperature coke for complete gasification, as compared with high temperature coke from vertical retorts. Since the tar has, for the most part, been removed from the semi-coke, it is reasonable to anticipate the success of using this fuel as a charge for gas producers. The low temperature coke gave 23% higher consumption of carbon than the high temperature product and yielded under the same conditions twice the percentage of carbon monoxide and less dioxide than the high temperature gas coke. The high velocity of consumption is indicative of high reaction velocity, which accounts for the increase of 25% in temperature of the producer hot zone.
One of the greatest potential markets for low temperature coke is that of a household fuel. Semi-coke is ideal for this purpose since it is clean, ignites easily, and burns readily with little attention. Fireplaces and other domestic heating appliances emit a large portion of their heat by radiation. Fishenden (207) has investigated the radiation efficiency of low temperature coke, as compared with coal ordinarily used. Three tests were made on various types of open grates. In the first test, a radiation efficiency of 19.7% was obtained for low temperature coke, as compared with 17.3% for bituminous coal; the second test showed the semi-coke to have a radiation efficiency of 24% and the bituminous coal 19.5%. The third test gave corresponding figures of 30.8% and 24.2%. This shows that in open grates the radiation efficiency of low temperature coke is 20% to 25% greater than that of bituminous coal.
In kitchen boilers, Fishenden (207) also found low temperature coke to be more effective than bituminous coal. Three different tested were conducted likewise. In the first test, the radiation efficiency of semi-coke was 17.3%, as compared with 13.1% for bituminous coal; the corresponding figures for the second test were 20.7% and 14.5%; while in the third trial the low temperature coke gave 41% radiation efficiency and the bituminous coal 31%. In ordinary kitchen ovens, however, the coke does not show any increase in efficiency over coal, but probably in an oven designed to take better advantage of the increased radiation this could be improved. The advantage of low temperature coke over coal, aside from its smokeless quality, lies in its superior radiation, which is most apparent when the distance from fuel bed to boiler or oven is small. When they are far apart, the advantage of the coke diminishes and the longer luminous flame of coal gives better results.
An ideal fuel for pulverization should contain about 10% volatile matte and be easy to grind. One of the principal difficulties in installations utilizing powdered fuel is the wear on the pulverization mills. Coke residuum from the low temperature process is comparatively easy to crush and prepare for the furnace. For these reasons, low temperature coke is peculiarly suitable for pulverized fuel. Gentry (208) has stressed the importance of semi-coke as a pulverized fuel for central electric stations and pointed out that the fuel can be prepared by grinding before or after carbonization or between operations of a multi-stage process. A number of methods have been devised, notably by McEwen and Runge, in which the coal is carbonized in finely divided particles, thus entirely eliminating or reducing the pulverization ordinarily required after distillation.
It is well to consider the specific case of a bituminous coal which contains 32.2% volatile matter, 57.8% fixed carbon, 2.8% moisture, and 7.2% ash, with a heating value of 13,800 BTU/lb. Such a coal ordinarily contains about 5.4% hydrogen, which causes a loss on combustion of 628 BTU/lb. Before pulverization, it is considered expedient to dry the coal to a moisture content of about 1.7% water in order to reduce the power required for grinding. The 1.7% moisture in the fuel after drying causes a further loss of 22 BTU/lb on burning. These losses reduce the heat available in the boiler furnace to 13,150 BTU/lb. A modern large pulverized fuel boiler has an overall efficiency of about 88%, calculated on the gross heating value of the coal, or 92.4%, figured on the net heating value as delivered to the furnace. A year's operating experience on the Columbia Power Station (209) showed that on the average 0.94% of the coal was required to dry the fuel, with 3.9% average initial moisture, to an average of 1.7% moisture before grinding. Proportioned for a coal with 2.8% initial moisture, 65 BTU/lb must be deducted for drying. At the same power plant 162 kwh/ton were required to operate the mills which, since the plant operated on 12,495 BTU/kwh, amounts to 102 BTU/lb required for pulverization. These additional deductions for preparation of the fuel give a new figure of 12.007 BTU/lb, or 87% of the calorific value of the raw fuel, usefully absorbed by the boiler.
An average externally heated low temperature coke made from the bituminous coal considered above will have a thermal value of 13,400 BTU/lb. Such a semi-coke will contain no moisture, as discharged from the retorts, but may absorb as much as 1% upon standing, thereby causing a combustion loss of 13 BTU/lb. The residual hydrogen in the low temperature coke amounts to about 2%, which represents a combustion loss of 233 BTU/lb. Deduction of the hydrogen and moisture losses gives 12,168 BTU/lb, available in the furnace when the same boiler efficiency as used for the raw coal is applied. Semi-coke needs no drying before pulverization, so that no deduction need be made for that item, while the heat required for carbonizing the coal will be charged to the cost of processing for by-products. The consensus of opinion of those who have had experience in grinding semi-coke is that it will require 20% less power for pulverization than the raw coal. On this basis a deduction of 81 BTU/lb must be made for grinding, giving finally 12,087 BTU/lb usefully absorbed in the boiler. This amounts to 87.6% of the heat in the semi-coke usefully absorbed, as compared with 87% for the coal from which the semi-coke was made. Similar calculations made for a semi-coke obtained from an average partial gasification low temperature process, with 13,200 BTU/lb calorific value and 1.5% hydrogen, show about the same results. It may be concluded, therefore, that low temperature coke, pound for pound, has a furnace efficiency in the pulverized form slightly exceeding that of the raw coal from which it was made.
Moreover, there are further economies that may be expected from the use of pulverized low temperature coke as a boiler fuel, which may be of importance exceeding that of importance exceeding that of its increased furnace efficiency. In the first place, semi-coke can be stored indefinitely in the powdered form without danger of spontaneous combustion, so that mill house operating costs can be reduced somewhat by allowing the mills to run at a much better load factor. Furthermore, the friability and softness of semi-coke will require less mill maintenance than raw coal. Here record must be made of the fact, commonly misunderstood, that low temperature coke is in no way comparable to high temperature coke, which is known to be brittle and excessively abrasive. The firing of pulverized coal in boilers requires an excessively large furnace volume to secure complete combustion, because of the long flame. The length of a pulverized coal flame depends solely depends upon rate of combustion, which in turn depends on three things, temperature of the furnace, turbulence to bring fresh air for oxidation, and combustibility of the fuel. The high combustibility of low temperature coke has already been pointed out, so that it is quite apparent that, for given furnace temperatures and given conditions of mixing, the rate of combustion of semi-coke is greater than raw coal and its pulverized fuel flame correspondingly shorter. In fact, certain experiments have shown that the flame of pulverized semi-coke was so short that it was necessary to ix a certain proportion of raw coal to lengthen the flame in boilers designed with large furnace volume. These results demonstrate that by using pulverized semi-coke in place of the raw coal, considerably smaller furnaces can be used for boilers of the same capacity, or vice versa, for a given furnace volume, increased combustion rates will permit the boilers to be designed for greater capacity. Either increased capacity for a given furnace size or reduction in furnace size for a given boiler capacity will effect material savings in capital expenditure, which will ultimately be reflected in the cost of power. A further discussion of this subject will be given in Chapter VIII under the subject of by-product recovery in central stations.
The safety with which powdered semi-coke can be stored without fear of explosion is of importance in marine power plants. The limited space on shipboard and on locomotives also makes the short flame of low temperature pulverized coke and its associated small furnace volume of considerable importance to the transportation industry.
Low temperature coke briquets, made by the Carbocoal process, have been tested as a fuel in both locomotives and marine installations. It was demonstrated that the fuel was smokeless and that it evaporated from 8.5 to 12.8 pounds of water per pound of fuel per hour, from and at 212° F, when fired at rates ranging from 100 pounds to 27 pounds of fuel per hour per square foot of grate surface, respectively. A maximum rate of 166 pounds of low temperature briquets per square foot of grate surface per hour was fired for short periods with no greater draft than was required by ordinary bituminous coal.