Eduction of Primary Tar ~
Designation of the condensable products from low temperature
carbonization, taken collectively, as a tar is most unfortunate,
for they more nearly resemble oils or petroleums than they do
tars. Since it has become well established by prior usage, the
name tar will be retained in this volume and the term primary
tar will be used synonymously with low temperature oil. This
confusion in terminology has caused a great deal of
misunderstanding and many persons have thereby been led to
evaluate low temperature crude oil in terms of high temperature
coal tar, to which it bears very little relation.
Like any industry in the early stages of its development, the
products of low temperature carbonization have had no
established market in the past. This applies in particular to
the primary oil. The chief assured industrial uses of low
temperature tar, at present, is as a crude stock for the
preparation of creosote oil and as a liquid fuel. While the
valuable by-products, which may be obtained by fractionation of
the tar will doubtlessly find industrial favor eventually, no
such outlets can at this time (1928) be guaranteed. There are
but few plants operating on a large scale, and consequently no
standard method of industrial analysis has been developed,
despite the quality and quantity of work that has been done in
the study of low temperature tar by numerous investigators. For
the most part, these workers were interested more from the
chemical than from the engineering standpoint, and therefore
planned their investigations more from the standpoint of science
than of technology.
The attitude of the tar distillers, in regard to low
temperature tar at the present time (1928), appears to be that
its refinement will require extensive alterations in their
plants, due to the manner in which primary tar differs from the
high temperature tar, which they are accustomed to handle. The
absence of regularly operating large scale low temperature
carbonization plants of sufficient capacity to assure them of
dependable quantities of raw material has thus far rendered the
necessary changes impractical. This situation is rapidly being
remedied and will be overcome in proportion as sources of raw
material increase and as markets for the refined products
develop.
According to Parrish (117), whatever coal may be used in the
same high temperature coke oven, the resultant tars resemble
each other to a great extent. Primary tar, on the other hand, is
much more closely allied to the coal from which it originated
and variations in the composition and quantity of the tar
yielded follow more closely variations in the character of the
coal from which it was produced. It will be observed later that,
in general, the younger the coal and the greater its oxygen
content, the greater is the quantity of low temperature tar
yielded per unit of weight.
The influence of the conditions of carbonization can almost
wholly be attributed to the action of heat upon the tar vapors.
The effect of these conditions on the low temperature tar has
already been alluded to in the definition of low temperature
carbonization. Aside from the bearing which the chemical nature
of the coal and the conditions of retorting have upon the
character of the tar, there remain certain influences, resulting
from the physical properties of the coal. If the coal is a
readily fusing one, the formation of a plastic layer may so
retard the passage of the primary tar vapor from the
carbonization chamber as to cause it to remain for a
comparatively long time in contact with hot surfaces. The effect
of hot surfaces in catalyzing secondary decomposition has
already been demonstrated.
Low temperature tars differ materially from high temperature
tars. The former are brownish black, fluid at ordinary
temperatures, and more viscous than crude Pennsylvania
petroleum. In general, the low temperature tars consist of
hydrocarbons of the alipathic series, a few aromatics, and a
high proportion of the tar acids. This tar is not greatly
different from shale oil and crude petroleum, and for that
reason some of the methods used in the petroleum industry may be
applied to the evaluation of low temperature tar.
Table 44 gives a representative comparison of the fractions
obtained from high and low temperature tars. The high
temperature tar yields are the average of 6 coke oven tars of
the United States, while the low temperature yields are the
average of 8 British experiments at 600° C. In the high
temperature product, only about 25% of the tar is volatile up to
315° C, while low temperature tar yields nearly 55% of its
weight in oils. High temperature pitch is almost three-quarters
of the entire yield of tar and contains a high percentage of
free carbon, whereas the primary tar is characterized by less
than 50% pitch of a low free carbon content. The decrease in the
yield of oils at high temperatures is complementary to the high
gas yield. It has been demonstrated that, in this case, the
primary volatile products are cracked and decomposed at higher
temperatures, with the formation of gas and the deposition of
carbon.

===45tab===
Experiments conducted in England, in especially constructed
cast-iron retorts, on 5 high class Barnsley coals whose
proximate analyses were almost identical with that of
Pennsylvania bituminous coal, showed that in low temperature
carbonization the average yield of tar was 23.5 gallons per net
ton of coal. The average of 6 Pennsylvania coals distilled at
550° C by Davis and Parry (97) gave 27.8 gallons of dehydrated
tar, with a specific gravity of 1.039, per net ton of coal
carbonized. This is equivalent to 12.2% of the coal by weight.
The Fuel Research Board, in large-scale horizontal retorts,
obtained a tar yield of from 17.9 gallons to 19.6 gallons per
net ton. They recommend, as a result of their extensive
investigations, that 19 gallons of tar per net ton is the
average yield to be counted upon in low temperature
distillation. The present author, however, believes this figure
to be unduly low for other processes and for high volatile
American coals. The fuel value of this primary tar is very high,
the average of 8 British experiments giving 16,630 BTU/lb.
Porter and Ovitz (96) in their investigation on the volatile
matter of coals of the United States, obtained from 12.4 gallons
to 21.2 gallons of tar per net ton of coal in small-scale
experiments, depending to a great extent on the geologic age of
the coal examined. The experiments were conducted over a period
of one hour in a furnace adjusted so that the interior of the
charge remained at 800° C. Monett (100) observed, from a study
of 11 Utah coals, that the tar yield ranged from 16.1 gallons to
29.4 gallons per net ton. It is interesting to note in Table 45
the general decrease in yield of tar accompanying the
consolidation of the fuel.
The results of Table 45 have been observed also by Berry (118),
who found a distinct relation between the amount of tar
distilled and the geologic formation of the coal, or more
specifically of the carbon-hydrogen ration of the fuel. The
curve in Figure 18 is due to him. According to this curve, the
maximum amount of thick tar is to be expected from fuels having
a carbon-hydrogen ration ranging from 13.5 to 18.0.
Berry (118) investigated the tar-forming temperatures of
American coals and found that the first trace of tar appeared,
in all the coals examined by him, at about 300° C and the last
traces disappeared at about 550° C, although in a few instances
tar formation did not cease until over 600° C was reached. Tar
was formed in quantities in the temperature range from 375° C to
475° C. he found no evident relation between the volatile matter
in the coal and the amount of tar evolved. He also concluded
that neither geologic age, carbon-hydrogen ration, nor
percentage volatile matter in the coal had any relation to the
temperature of tar evolution.
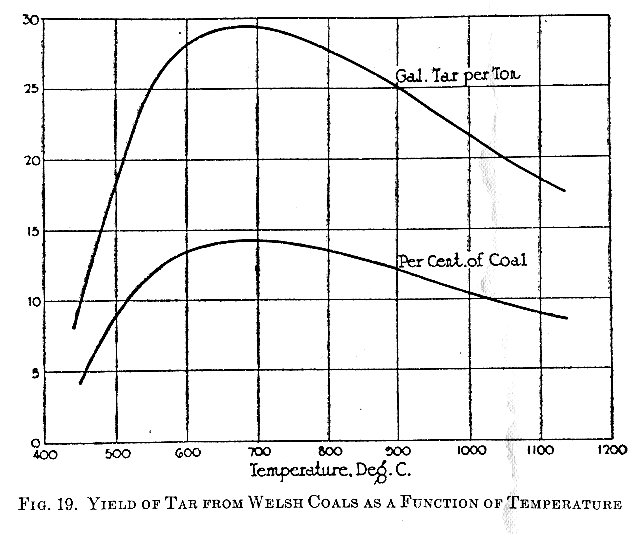
Burgess and Wheeler (57), in examining Welsh coals, found
certain variations in the yield of tarry matter when the
temperature of carbonization ranged form below 500° C to above
1000° C. Their data are plotted in Figure 19, where it is seen
that the yield of tar per gross ton of coal rises to a maximum
at about 700° C. The experiments of Burgess and Wheeler (57) are
not in agreement with the figures given by Lewes (17), whose
maximum yield occurred about 300° C lower. No information was
given by the latter authority regarding the composition of the
coal used to secure his data, so that the disagreement cannot be
explained. The specific gravity of the tar gradually increases
with the temperature increase. This is to be expected, since the
lighter hydrocarbons are destroyed through decomposition at the
higher temperatures to form fixed gas, leaving the heavier,
higher-boiling hydrocarbons and the carbon freed in the
cracking.
Porter and Ovitz (96) investigated the tar distilled at various
temperatures from several coals of the United States. Their
figures, computed from a percentage basis to gallons of tar per
net ton, are plotted in Figure 20. Comparison of this
illustration with Figure 19 will disclose that, qualitatively,
the data are in agreement with the results obtained by Burgess
and Wheeler (57) on Welsh coals. In Figure 20 it appears that,
from the standpoint of securing maximum tar yield, the optimum
temperature of carbonization is in the neighborhood of 800° C,
although in commercial plants this figure appears somewhat high.
Too much confidence cannot be placed in the quantitative value
of these curves for they seem low for high volatile coals.


According to Fischer (119), the solid paraffins in primary tar
represent 1% or 2% of the tar and in this respect we find one
striking difference between the tar obtained from brown coals
and that obtained from bituminous coals. Fischer (120) found the
tar from Saxon brown coal to contain as much as 29% solid
paraffins, while the tar from Rhenish brown coal yielded 13%. In
other respects, the two primary tars are quite similar.
Garbe (121) distilled a Shetland peat containing 21.4% moisture
and 73.8% volatile matter, at 500° C in a Tozer retort and
obtained a yield per gross ton of 33.6% peat char, 4500 cu ft of
500 BTU/cu ft gas, 1.44 gallons of 0.7885 specific gravity motor
spirit, 23.4 gallons of crude oil, and 13.6 lb of ammonium
sulfate. The peat tar contained 35.2% moisture, 2.2% light oil
up to 170 C, 19.8% medium oil in the fraction 170° C to 230° C,
and 37.8% residuum above 230° C. The middle oil contained 22%
tar acids.
Garvin (112) reports that, on the average, distillation of
Scottish shale yields 24.5 gallon of 0.860 sp gr tar, 9800 cu ft
of 270 BTU/cu ft gas, 35.7 lb of ammonium sulfate, and 1550 lb
of waste spent shale from each ton of raw material. Oil shales
of the United States have tested all the way from less than 7.5
gal of tar/ton, in the case of a Nevada sample, to over 60
gal/ton for a specimen from Colorado. The specific gravity of
these tars varies also from 0.881 to 0.924. The average test
yield of tar from 11 US shales from 6 states was 37.1 gal/ton.

Time Effect ~
The time element deserves important consideration in regard to
the tar as well as with respect to the gas. If the distillation
is slow and the tar vapors are removed slowly, secondary
decomposition may occur with a corresponding decrease in the
yield of oils and increase in the evolution of gas. But the tar
yield is not the only outcome of slow carbonization. The
structure of the tar is vastly changed. A tar produced in slow
distillation contains a larger proportion of the aromatic
derivatives and fewer members of the alipathic series. The curve
of tar yield against time is somewhat similar to that of gas
evolution, shown in Figure 14. We know, of course, from the
differential calculus, that the derivative of this curve is the
curve of the rate of yield of tar. Inspection of the slope shows
that the rate of yield in gallons/minute, as plotted against
time, should reach a maximum and then gradually fall off until a
point may be reached where it will be uneconomical to prolong
the process for the tar alone. In other words, the rate of yield
curve for tar has been found by experiment to be similar to that
for gas shown in Figure 17. It should not be overlooked that,
although the production of tar after a certain period may not be
sufficient to warrant continuation of carbonization, the gas
evolved may entirely justify prolongation.
The effect of bringing the charge up to maximum temperature in
5 hours, as compared with attaining the same temperature within
a fraction of an hour, is shown by the results of Table 46,
after Taylor and Porter (98). These experiments were conducted
on Pennsylvania coal. It is seen that in every case the yield
changed from about 16.0 gal/gross ton of coal to about 20.5
gal/gross ton when the period of attaining the maximum
carbonization temperature was decreased from 5 hours to a much
shorter period.
Pressure & Vacuum Effect ~
Some 30 years ago, Dewar and Redwood patented the distillation
of petroleum under pressure to crack the heavy distillates into
low-boiling and more valuable fractions. In 1910, Burton
developed the pressure cracking system and now it is a
well-established industry. Attempts have been made to reduce the
deposition of carbon by the introduction of catalyzers and
Bergius has proposed the hydrogenation of the hydrocarbons by
carbonization under pressure in an atmosphere of hydrogen.
Cracking, however, in low temperature carbonization process, is
ordinarily avoided in order to preserve the primary products of
distillation. Hence, the retorts are usually operated at
atmospheric pressure under slight vacuum, of hardly more than 2
inches of water, to overcome the resistance of the piping
system. A discussion of the possibility of increasing the yield
of lighter hydrocarbons, suitable as a fuel for internal
combustion engines, by cracking the primary tar will be taken up
under the subject of motor spirit.
A vacuum system assists in the removal of the vapors and
prevents condensation of the heavy tars in the retort. It also
removed the air which is present, thus preventing oxidation, and
preserves the primary tar by avoiding cracking. In 1905 an 1906,
Simpson obtained patents which were afterwards incorporated in
the Tozer retort for distilling in partial and complete vacuum.
Little work has been done by way of investigation in this field,
but Taylor and Porter (98) have examined certain coals of the US
when distilled at pressures below 4 cm Hg. A comparison of the
tar yields from atmospheric and vacuum carbonization is
instructive. The atmospheric distillation in Table 47 was
carried out at 475° C. The figures, for convenience, have been
converted from a percentage basis to gallons per gross ton.
The effect of temperature on the yield of vacuum tar also was
investigated by the same authorities. In this case, different
periods of distillation were used at different temperatures, so
that the data are not strictly comparable, but they are given in
Figure 21 because no other similar figures are available. The
data are computed to gallons per gross ton. It will be noted
that the yields in this illustration are higher than those
indicated by Table 47. This is due, doubtlessly, to the
systematic error pointed out above, as well as to an additional
cause. In this case, the percentage tar obtained was determined
by subtraction, rather than by direct observation, and
consequently the cumulative errors of analyses fall entirely on
the tar content. Figure 21 should be compared with Figure 20,
where the corresponding results of atmospheric distillation are
shown. The main difference in the characteristic temperature
versus yield curves of atmospheric and vacuum carbonization lies
in the absence of a maximum on the vacuum system. The yield of
tar, in the latter case, quickly rises until about 450° C is
reached, when little change in the quantity of tar produced is
noticed during the next 150° C temperature increase. Thereafter,
the yield of tar increases with temperature rise even up to
1050° C.


Atmospheric & Moisture Effect ~
Taylor and Porter (98) investigated the effect of inert
atmospheres on the quantity of tar produced. The presence of
coal gas or nitrogen did not exert any decided influence on the
yield. Thus, over a 5-hour period of distillation, 17.1 gallons
of tar per gross ton were obtained in an atmosphere of coal gas
at 950° C and 16.0 gallons when pure nitrogen was introduced
into the retort during carbonization at 1040° C.
Weathering of the coal before coking appears to have an effect
on the quantity of tar evolved in carbonization. Thus, it was
found that a Wyoming coal yielded 9.1% of its weight as tar,
when distilled as received, but only 6.2% after air-drying. It
is a well known fact that weathering reduces the volatile
content of the coal, both through escape of the occluded gases
and through oxidation. It is not surprising, therefore, to find
a larger evolution of tar immediately upon reception of the
sample than after the coal has been exposed to the action of the
air.
The moisture content of the coal does not have any influence on
the amount of tar produced, when computed upon a dry coal basis,
according to Porter and Ovitz. (96). Thus the yield of tar from
a dry Pennsylvania coal, containing but 1.2% moisture, as
compared with another sample containing 9.6% moisture, is given
in Table 48. The decrease in tar, when figured as a percentage
of the coal as charged, is a fictitious decrease only, arising
entirely from the selection of a base for computation which
contains a smaller quantity of the tar-producing element.

Davis and Parry (97), it will be recalled, conducted
experiments to determine the effect of superheated steam upon
the carbonization products. It has been shown that probably the
temperature is not sufficiently high in the low temperature
distillation process to cause any interaction between the steam
and the primary volatile products, and consequently the
principal effect of steam will be in preventing secondary
decomposition by assisting the quick removal of the products.
With Pennsylvania coal from the Upper Freeport bed, Davis and
Parry (97) found that 33.0 gallons of tar per gross ton were
obtained without the use of steam, whereas the following yields
were obtained when 88% steam was passed through the retort: 30.8
gal/gross ton at 475° C, 33.3 gal/gross ton at 550° C, and 50.9
gal/gross ton at 650° C. At 550° C, which is the only
temperature at which experiments were made on the same coal
without the use of steam, apparently there was little effect on
the yield. It is evident that an enormous quantity of tar was
evolved under steam distillation at 650° C. Unfortunately, no
information is at hand to determine whether this large yield is
due to the presence of steam or is a peculiarity of the coal.
The tar produced in steam distillation at low temperatures is a
little more viscous but otherwise it does not appear essentially
different from the tar obtained without the use of steam. Its
fractionation, however, shows it to have a marked difference in
volatile content. It contains a smaller percentage of light
oils, but more of the heavier hydrocarbons. This topic will be
taken up again under a discussion of the fractionation of low
temperature tar.
Constitution of Primary Tar ~
Although low temperature tar obtained from lignite and brown
coal may be solid at ordinary temperatures, due to a large
proportion of solid paraffins, primary tar, in general, is
lighter and more fluid than that from high temperature
processes. The specific gravity of primary tar usually ranges
from 0.95 to 1.06, as compared with 1.2 sp gr for high
temperature tars, according to Fischer (119). The comparatively
low density is due mainly to the presence of large proportions
of liquid hydrocarbons and phenols. Low temperature tar usually
has a strong odor of phenols and of hydrogen sulfide, but it
never has the naphthalene odor of secondary tars. In transparent
layers, its color ranges from orange to red. Fischer and Gluud
(60) have pointed out another way in which low temperature tar
resembles petroleum and differs from high temperature aromatic
tars. They found that some of the hydrocarbons from primary tar
exhibit a small but distinct optical activity. In a tar from
Lohberg coal, freed of phenols, the optical rotation with sodium
light through a 20 cm tube varied from +0.05 for the fraction
175° C to 185° C, to the value +0.30 for the fraction from 225°
C to 235° C. Low temperature tars are usually soluble in the
ordinary organic solvents, such as benzene, ether, acetone,
chloroform, carbon tetrachloride, and petroleum ether, but its
solution is usually accompanied by precipitation of resins.

It has been shown conclusively that distillations of the
primary tar, during analytical procedures, distinctly alters its
composition, especially that of the higher boiling compounds.
Fischer (49) states that primary tar cannot be distilled without
the part above 300° C decomposing, the viscous lubricating oils
suffering the greatest decomposition and the higher phenols
being affected to a lesser degree. A specific case of this for
low temperature brown coal tar has been given by Fischer (85)
and is reproduced in Table 49. The distillation was carried out
slowly for 8 hours, yielding 85.6% oil, 4.7% gas, 9.2% coke, and
a small amount of water. It will be observed that distillation
so altered the constitution of the fraction from 70° C to 250° C
as to increase its proportion from 18.5% to 31.7% of the
original tar. The proportion of acids was simultaneously greatly
decreased. Schneider (122) confirmed these results with tar from
a bituminous coal.
Decomposition of primary tar during distillation is further
indicated by the evolution of gas. Windel (123) found that gas
first was evolved at 270° C, reaching a maximum at about 335° C.
The gas contained 7% hydrogen sulfide, 4% carbon dioxide, about
75% methane and its homologues, and about 10% ethylene and
acetylene hydrocarbons. Edwards (124), and also Brittain, Rowe,
and Sinnatt (125) detected the evolution of sulfur dioxide at
the beginning of distillation and hydrogen sulfide at 170° C,
when studying tar from the Coalite process.
It is quite evident that the evaluation of low temperature tars
is a difficult task and that the method of separation by
solvents must be used to avoid distillation as much as possible,
but even this method of approach is not without its difficulty
of solvent reaction. These troubles are augmented by the fact
that most low temperature tars change color and character on
standing. This has been attributed by Jaeger (126) to oxidation
by the atmosphere through the action of light, but the presence
of bases are important factors. Parr and Olin (37) found, also,
that the viscosity of the tar increased with standing,
apparently the result of oxidation, for Brittain, Rowe, and
Sinnatt (125) found no change when the tar was kept in darkness
and out of contact with air.
As early as 1862, Williams (127) and Schorlemmer (128) seem to
have made the first chemical examination of low temperature tar
obtained from cannel coal. The latter of these investigators
identified paraffin hydrocarbons. However, the first noteworthy
research was conducted by Bornstein (95) in 1904. He
fractionated primary tar from Westphalian coals, from 50° C to
450° C, and found it conspicuously different from high
temperature tar, being a thin liquid with little free carbon and
containing no naphthalene or anthracene, but quantities of
phenols and up to 2% solid paraffins. With another Westphalian
low temperature tar he found no solid paraffins, but something
less than half of one percent methylanthracene.
Pictet and Bouvier (51, 129, 130 & 131), and also Pictet,
Kaiser, and Labouchere (52) conducted a series of
investigations, beginning in 1913, on primary tar obtained from
vacuum distillation of a French bituminous coal containing about
11% ash, 70% fixed carbon, and 19% volatile matter. The fresh
tar was composed of 68% unsaturated hydrocarbons, 30% saturated
hydrocarbons, 2% alcohols and 0.2% bases. They found no phenols
in the fresh tar, but detected some after the tar had remained
standing a long time. As other investigators have found over 15%
phenols in fresh tar examined by them, it appears that Pictet
and Bouvier must have had an exceptionally true primary tar.
They established the presence of toluidine, pentamethylphenol,
tetramethylphenol, xylenol, hydroguinoline, and isoquinoline.
Jones and Wheeler (42) examined the tar obtained from a
Scottish and a Durham coal, distilled at 450° C in vacuo. In the
oils below 300° C, they found about 40% unsaturated
hydrocarbons, about 7% aromatic compounds, probably homologues
of naphthalene, about 12% phenols, chiefly cresols and xylenols,
traces of pyridine bases, and a small amount of solid paraffins.
Identification of Compounds ~
Regarding particular chemical compounds that are found in low
temperature tars, some quantitative determinations by Schultz
and Buschmann (34, 35) and also by Fieldner (36) have been given
under the chemistry of low temperature carbonization, discussed
in Chapter I and compiled in Table 5 and Table 6 respectively. A
qualitative list of compounds, actually identified in low
temperature products, was given in Table 7, accompanied by
references to the authorities who isolated them.
Among the aromatic hydrocarbons, Whitaker and Crowell (132,
134), when investigating a Pennsylvania coal, observed that
benzene first formed at 500° C and toluene at 400° C, but that
maximum yields were not obtained until 800° C for benzene, 700°
C for toluene, and 600° C for xylene. Traces of benzene have
been found by a number of observers, but most investigators have
been unable to detect it in primary tars examined by them. Parr
and Olin (37) found benzene, as well as toluene, xylene, and
possibly mesitylene, in Illinois low temperature tar. Schultz
(43) is the only authority who has reported considerable
quantities of benzene hydrocarbons. He examined a tar obtained
by distillation of Furst-Hardenberg coal in a rotary retort at
about 480° C. Broche (53), however, examined tar from the same
coal and found less that 0.4% benzene, from which he concluded
that the tar examined by Schultz was abnormal. Fischer and Gluud
(39) detected a very small amount of benzene in tar from one
coal examined by them.
Besides Parr and Olin, toluene in minute quantities was found
by Jones and Wheeler (42) and its presence was confirmed by
Frank and Arnold (133). The latter observers also found three
xylenes in the tar fraction from 120° C to 180° C. Klein (135)
found that both toluene and xylene in the light oil from a large
rotary retort, while Weissberger and Moehrle (46), and also
Kruber (136) and Kaffer (137), found the higher homologues of
benzene, of which pseudocumene, durene, and a few others have
been identified.
Less than 1% naphthalene seems to be present in low temperature
tars, as compared with 4% or 5% in aromatic tars. According to
Ruhemann (138), brown coal low temperature tar contains some
naphthalene. Brittain, Rowe, and Sinnatt (125), Morgan and Soule
(45, 48), as well as Weissberger and Moehrle (46), found it in
tar from full-scale low temperature plants, while Parrish and
Rowe (139) found small quantities in tar carefully distilled
from coal at 600° C. Anthracene is present in even smaller
proportions than naphthalene, but it has been reported as
present by Britain, Rowe and Sinnatt (125) and by Weissberger
and Moehrle (46), the last of which also established the
presence of naphthalene homologues such as the
methylnapthalenes, confirming the observations of Fischer,
Schroeder, and Zerbe (47).
Among the saturated and unsaturated hydrocarbons, Fischer and
Gluud (39) found pentane, hexane, many of their homologues, and
possible terpenes, among the low-boiling fractions of primary
tar. Schultz, Buschmann, and Wissebach (38) established the
presence of ethylene, propylene, butylenes, pentene, butadiene,
and cyclopentadiene in the light oils, most of which were also
found by Fieldner (36) in certain American low temperature tars,
as shown in Table 6. Klein (135) also detected octylene and
nonylene in large-scale primary tar.
Among the oxygenated compounds, acetone has been observed by
Frank and Arnold (133), by Schultz (140), and by Broche (53).
Acetaldehyde and indications of ketones were obtained by
Brittain, Rowe and Sinnatt (125), while Schultz (140) also
identified acetaldehyde as well as methylethylketone and
acetonitrile.
Free sulfur is probably present to a small extent in low
temperature tar, although its presence is undesirable, but it
cannot be avoided if the raw coal is one of large sulfur
content. Schultz (43) detected methylmercaptan and
dimethylsulfide, in addition to the simple sulfur compounds
hitherto mentioned. Brittain, Rowe and Sinnatt (125) found over
1.5% combined sulfur in the crude full-scale low temperature
tars examined by them.
Tar Acids ~
Heretofore it has been pointed out that one of the outstanding
peculiarities of low temperature tar is its high acid content.
These acids do not consist so much of phenol itself as of
homologues of the series, such as the cresols, xylenols, and
more complex phenol derivatives.
There has been a great variation in the amount of acids present
in the tars examined by various authorities. Thus, Jones and
Wheeler (42) found about 7%; tests on the Toser retort gave
10.2%; Edwards (124) reported 10.8% to 15%; Morgan and Soule
(48) determined 14.7%; Davis and Gallway (141) found from 12% to
19%; the Fuel Research Board reported 17.8%; Morgan and Meigham
(142) 17.5%; Davis and Parry (97) 18.2%; tests on the Nielsen
retort yielded 20%; Parr and Olin (37) 27.9%; and Church and
Weiss (143) as much as 50%.
The work of Morgan and Soule was conducted on tar from the
Carbocoal process; Jones and Wheeler used vacuum tar; Church
examined a tar from an Illinois coal of high oxygen content;
Parr and Olin used Illinois coal; Davis and Parry used tar
derived from Pennsylvania coal; Edwards used English coals. The
high yield obtained by Church does not seem extraordinary, when
it is recalled that he used a coal of high oxygen content and
that the phenolic compounds contain combined oxygen. In fact, it
seems permissible to predict that, in general, coals which are
highly oxygenated may be expected to give a larger quantity of
the tar acids upon carbonization. The same may be said, perhaps,
of coals which have been weathered through exposure to the
atmosphere or retorted in the presence of a considerable amount
of oxygen. Unfortunately, no experimental data are at hand to
substantiate this belief.
The bulk of low temperature tar phenols consist of complex
hydrogenated and alkalated derivatives or ordinary high
temperature tar phenols. They are largely saturated compounds.
Morgan (144) states that on account of their long side chains
the primary tar phenols are less soluble in water than those
derived from aromatic tars.
Phenol itself usually is present in amounts of less than 0.5%
of the tar, as compared with twice that amount for high
temperature tars. Its presence in small quantities has been
established by Fischer and Breuer (145), by Brittain, Rowe, and
Sinnatt (125), by Morgan and Soule (48), by Parrish and Rowe
(139), and by Frank and Arnold (133). Schultz (43) and his
coworkers (38) are the only investigators reporting as much as
1.35% phenol I the crude tar. Pictet, who examined an
exceptionally true primary tar, found no phenol but up to 2%
methylcyclohexanol, an unstable alcohol which decomposed into
phenol upon standing.
All three cresols have been identified, but there seems to be
no uniformity in the way in which they predominate. Gluud and
Breuer (50) found that m-cresol predominated; Brittain, Rowe and
Sinnatt (125) found about 46% of-cresol, about 37% m-cresol, and
about 16% p-cresol; while Morgan and Soule (48) found 54%
p-cresol, 27% of-cresol, and 19% m-cresol. Schultz, Buschmann,
and Wissebach (38) have identified 4 of the zylenols, while
Avenarius (147) has isolated a fifth.
Catechol was detected qualitatively by Morgan and Soule (48),
by Edwards (124), and by Parrish and Rowe (139). Bornstein (95)
also detected it, while Gluud (148) estimated the quantity of
this compound in the tar from Lohberg coal, examined by him, to
amount to 0.02% of the coal. Brittain, Rowe, and Sinnatt (125)
found catechol present in tar from the Coalite process.
Among the higher-boiling phenols, Morgan and Soule (48)
reported the presence of napthols, while Weindel (123) observed
beta-napthol, and Edwards (124) reported both alpha- and
beta-napthol. Gluud and Breuer (50) found trimethylphenols, the
presence of which was confirmed by the researches of Fromm and
Eckard (40) and of Pictet, Kaiser, and Labouchere (52). The
latter experimenters also identified tetramehtylphenol and
pentamethylphenol. Marcusso and Picard (149) recorded the
presence of solid phenols in primary tar from Upper Silesian
coal, but Tropsch (150), who examined the low temperature tar of
a coal from the same region, was unable to confirm this.
Among the sulfonated phenols, Schultz, Buschmann, and Wissebach
(38) reported appreciable quantities of mercaptans and Avenarius
(147) concluded that thiophenols were present. Parrish and Rowe
(139) found 0.73% sulfur in their crude phenols, while Brittain,
Rowe and Sinnatt (125) estimated the sulfur content of the crude
phenols from Coalite tar to be less than 1%.
Some idea of the distribution of the various constituents may
be gained from a rough separation of the phenols made by Fischer
and others. They found from 1% to 6% cresols, bout the same
proportion of xylenols, 5% to 25% higher phenols, and 8% to 15%
acid resins. The tar from the Fuel Research Board horizontal
retorts yielded about half the acid content reported by Fischer,
about 5% cresols, 11% xylenols, and the remainder was composed
of the heavier derivatives of phenol. Parrish (117) has compiled
a list, shown in Table 50, which gives the distribution of
phenols among the tar acids in low temperature carbonization.



According to the results of Davis and Parry (97), introduction
of steam into the retort during distillation of the coal
considerably alters the quantity of tar acids produced in each
fraction of the tar. Their data are given in Table 51 as
percentages of the fraction. The tar examined was obtained in
both cases by carbonization of Pennsylvania coal from the
Freeport bed at 550° C. The yield is equivalent to 4.76 gallons
of tar acids per net ton of coal, or 14.5% of the crude tar, for
dry distillation and 4.04 gallons per net ton, or 12.2% of the
crude tar, under steam distillation. The average yield per net
ton from 5 Pennsylvania coals, tested by Davis and Parry (97)
was 5.45 gallons. The specific gravities of the acids separated
from the various fractions of tar were 1.021, 1.028, and 1.051
for the light, middle, and heavy oil cuts, respectively. The
middle oil contained a preponderance of cresols and xylenols.
Tar from the same coal, carbonized at different temperatures,
was examined by the same investigators with a view of
determining the effect of temperature variation on the
production of tar acids. The results are reproduced in Table 52,
which shows conclusively that increase of the temperature from
475° C to 650° C does not influence the yield to any extent. It
is apparent that the acids are stable up to beyond 650° C and
that no decomposition occurred at that temperature. This
experiment was carried out in the presence of steam, which to a
great extent reduced the susceptibility to thermal
decomposition.
Figure 22 shows the variation of the percentage of tar acids,
as a function of the average boiling point of the fraction, for
low temperature tar from Pennsylvania coal, as determined by
Davis and Parry (97), when compared with the same determinations
on tar from the Carbocoal process, as found by Morgan and Soule
(151). In the illustration, it will be noted that the maximum
yield of tar acids occurs in the fraction whose average boiling
point is 200° C, in the Freeport tar, whereas in the Carbocoal
tar this maximum occurs 60° C higher.
Tar Bases ~
The nitrogen bases are characterized usually by absence of the
simpler members, although pyridine and the simple alipathic
amines have been detected in small quantities. The tar bases
consist mostly of secondary and tertiary compounds and others of
unknown origin.
Davis and Parry (97) found 1.23% nitrogen bases in the low
temperature tar from Pennsylvania coal; Davis and Galloway (141)
found 2.8% to 5.7% in the tar distillate up to 275° C,
representing 1.1% to 2.3% of the total tar; Edwards 124) found
from 2.9% to 5.8% in the distillate up to 311° C, amounting to
1.3% and 2.5% respectively of the total tar; Jones and Wheeler
(42) found less than 1%; Parr and Olin (31) found 0.9%; Pictet,
Kaiser, and Labouchere (52) found as little as 0.2%; Fischer and
Gluud (152) found 0.46%; Parrish and Rowe (139) found 2.7%;
Morgan and Soule (48) found 0.6% in Carbocoal tar; and Brittain,
Rowe and Sinnatt (125) found from 2.45% to 4.1% in the total tar
from the Coalite process. These figures, however, can be
misleading, as most of the determinations were made on fractions
representing as little as 30% of the total tar and the quantity
of bases present in those distillates computed back on a total
tar basis, whereas it is quite apparent that a large percentage
of the bases remains in the tar residuum above 300° C.
Indications are that the tar bases amount to from 3% to 5% of
the total tar.
Gollmer (146) found among the primary amines in low temperature
tar, analine, the toluidines, and the xylidines; the primary
amines all amounting to 4.5% of the tar bases. Schultz,
Buschmann, and Wissebach (38) confirmed the findings of Gollmer.
Other investigators, however, found no primary amines, but
Morgan and Soule (48) found 20% secondary and 80% tertiary
amines; Brittain, Rowe and Sinnatt (125) fund 56% secondary and
44% tertiary amines; While Parrish and Rowe (149) found 15%
secondary and 85% tertiary amines.
It has been observed generally that a large proportion of
unsaturated compounds are found in the tar bases. Traces of
pyridine have been observed by Gollmer (146), by Brittain, Rowe
and Sinnatt (125) in the tar liquor, by Fromm and Eckard (40),
by Schultz, Buschmann, and Wissbach (38), and by Morgan and
Soule (48). Among the homologues of pyridine, Schultz and his
coworkers found methylpyridines, dimethylpyridines, and
trimethylpyridines; the presence of the latter being confirmed
by Gollmer (146), while Morgan and Soule (48) found
ethylpyridine and its hydrogenated derivatives. Brittain and
Soule (48) found ethylpyridine and its hydrogenated derivatives.
Brittain, Rowe and Sinnatt (125) also found small amount of
diethylamine and triethylamine in the tar liquor. Schultz (38)
and his associates established the presence of quinoline and its
derivative, methylquinoline, whiel Pictet and his coworkers
found a number of dihydroquinolines present.
The distribution of nitrogen bases in the fractions of low
temperature tar, as determined by Davis and Parry (97), is given
in Figure 23, where the percentage yield is plotted against the
average boiling point of the fraction. The determinations were
made by washing the oil fractions, after removal of the tar
acids, with 20% sulfuric acid to remove the bases. The results
shown in this curve agree with the fact discovered by Morgan and
Soule (48), namely, that the pitch contains a larger proportion
of bases than the lighter oil fractions.
Brittain, Rowe and Sinnatt (125) fractionated two samples of
Coalite tar under atmospheric and under reduced pressures and
determined the percentage of nitrogen bases present in each
fraction. The results are given in Table 53. It will be observed
that the quantity of bases, present in the various fractions, is
considerably greater than that shown in Figure 23, but the
general trend to large proportion of bases as the boiling point
of the fraction increases is in agreement with other
experimenters. It is quite apparent that the proportion of bases
in a given fraction is more than doubled by vacuum distillation.



Tar Solids, Sulfur, and Liquor ~
Many low temperature tars have been observed easily to form
emulsions with the tar liquor, thereby entailing a great deal of
trouble in bringing about demulsification. This tendency to
emulsify has ordinarily been attributed to the low specific
gravity of primary tar and to dust and other mineral matter
carried over with the tar during distillation. In the course of
their investigations, Brittain, Rowe and Sinnatt (125) isolated
a brown amorphous powder, precipitated from the tar fractions by
various organic solvents. After removal of this solid powder,
which amounted to as much as 0.94% of the tar in one sample and
9.9% of the tar in another, it was observed that emulsification
of the samples did not take place. It was concluded that this
solid material, present in the tar either as suspended powder or
partly in solution, was a contributing factor in emulsification.
The existence of such a brown powder was confirmed by Edwards
(124), who attributed its formation to changes in the tar vapor
prior to condensation or to changes in the tar itself during
storage, rather than to primary decomposition of the coal during
carbonization.
Ruhemann (138) distilled low temperature tar from lignite and
found sulfur in the various fractions to the amount of 3.5% in
the lower cuts and 1.9% in the higher cuts. The neutral oil of
the primary tar, examined by Marcusson and Picard (149),
contained from 5.4% to 6.3% sulfur. Avenarius (147) and Schultz,
Buschmann, and Wissebach (38) found sulfur in their tar acids.
The nature of these sulfur compounds has already been discussed
and need not be repeated here. Parrish (117) has compiled a
table from the experiments of Brittain, Rowe and Sinnatt (125)
and of Parrish and Rowe (139) to show the distribution of sulfur
in low temperature tars which were examined by them. This
compilation is given in Table 54. The notably high sulfur
contents of the tar solids suggests that it plays an important
part in these substances.
The aqueous distillate from low temperature tar is usually
straw yellow in color and has an acid reaction. According to
Jones and Wheeler (42), the tar liquor contains both
hydrochloric acid and ammonium chloride. Brittain, Rowe and
Sinnatt (125) made a careful examination of the tar liquor from
distillate of Coalite tars, and established the presence of
acetic acid, formic acid, acetaldehyde, and certain ketones
which could not be identified. They also found traces of
diethylamines, triethylamines, pyridine, and methylpyridine,
while phenol, cresol and xylenols were isolated.
Primary Tar Distillation ~
Following the procedure adopted in the evaluation of petroleum,
a rough separation of the constituents of low temperature tar
can be obtained by preliminary fractionation. Thus, the tar
obtained from the distillation of 5 high grade Barnsley coals
has been separated into 3 qualities of oil, according to the
boiling points. The first cut, up to 170° C, is given as light
oil, the second cut, from 170° C to 245° C, is called middle
oil, and the last cut, from 245° C to 315° C, is designated as
heavy oil. The viscous residuum is mostly pitch. The results of
the fractionation are given in Table 55. On the average 6.9% of
the tar by volume is light oil, 30.5% middle oil, and 18.4%
heavy oil. The proximate analyses of the coal, whose tar yield
is given in Table 55, from tests by Lewes as reported by
Wellington and Cooper (89), was very similar to that of
Pennsylvania coal containing about 60% fixed carbon and about
32% volatile matter.
A Warwickshire coal, containing 10.3% moisture, 35.3% volatile
matter, 40.8% fixed carbon, and 13.5% ash, was distilled at 600°
C in vertical retorts at the Fuel Research Station. The tar form
this experiment had a specific gravity at 15° C of 1.029,
contained less than 1% mineral matter and 2.29% water. It was
fractionated by Parrish and Rowe (139) with the results shown in
Table 56. The percentage acids and bases are also shown.
In regard to experiments made upon coals of the United States,
Parr and Olin (37) obtained a dark brown tar which had a
specific gravity of 1.069, from Illinois bituminous coal. Crude
fractionation gave the following results: 17.2% light oil up to
210° C, with a specific gravity of 0.966; 52.7% heavy oil from
210° C to 325° C, with a specific gravity of 1.032; and 30.1%
pitch over 325° C, with a specific gravity of 1.270. These
fractions were refined and the yield between certain points
computed as a percentage of the tar with the results shown in
Table 57.



Low temperature tars obtained from the Pittsburgh and Upper
Kittanning beds of Pennsylvania at 550° C to 650° C have been
fractionated by Davis and Parry (97). A comparison of the
boiling ranges of low temperature tar, crude shale oil, and
petroleum is given in Figure 24. Examination of these curves
discloses that below 250 C none of the low temperature tars is
as volatile as petroleum and that below 200° C shale oil
contains a larger percentage of volatile constituents than the
tar. Above 200° C, the percentage by volume of the tar which
distills exceeds that of the shale oil and, at a slightly higher
temperature, even that of petroleum. The low temperature tar
obtained from the Carbocoal process contains higher-boiling
hydrocarbons than the other tars, as demonstrated by the
position of the curve in the illustration.


Figure 25, after Davis and Parry (97), shows the distillation
range of tar from 5 Pennsylvania coals. All of these tars had
about the same volatility, except Pittsburgh rooster coal, which
contained more of the lighter oils with boiling points below
150° C. This particular tar gave a low percentage of phenol and
unsaturated compounds than the other samples. The specific
gravity of these tar fractions increased from 0.882 at 175° C to
0.992 at 275° C.
Parr and Layng (102) distilled low temperature tar from Utah
coal and found that 4.02% distilled from 0° C to 170° C; 37.1%
from 170° C to 300° C; 34.7% from 300° C to 3260° C, leaving
22.8% pitch and loss. The fraction from 170° C to 300° C was
composed of 28.6% tar acids, 4.6% amines, and 22.4% paraffins.
Giles and Villebrandt (99) made a careful study of the low
temperature carbonization products obtained at various
temperatures from Farmville, NC coal. The effect of temperature
upon the composition of the gas from this coal has already been
seen in Figure 12. The influence of temperature of carbonization
on the volatility of the tar is nicely illustrated in Table 58.
It will be observed that when the temperature of distillation is
advanced from 300° C to 660° C, the proportion of the lower
fractions as a percentage of the entire tar undergoes a decided
decrease, while the proportion of the heavier fractions have a
corresponding increase. Between the distillation temperatures of
300° C and 660° C, the percentage of the fraction 70° C to 170°
C is reduced at the higher temperature to almost one-fourth of
its initial value; the percentage of the fraction from 170° C to
230° C is reduced to about one-third of its former amount; and
the fraction 230° C to 270° C suffers a reduction to about
three-fifths of its value at the lowest temperature. On the
other hand, increase of the carbonization temperature from 200°
C to 660° C causes the amount of the fraction from 315° C to
355° C to about double, while the residuum above 355° C is
increased from 7.5% of the total tar to 54.9%. It is quite
evident that the higher the temperature of carbonization, the
less is the proportion of light oil and the greater is the
proportion of heavy oil and pitch in the tar.


We have already seen in Table 29 the effect of the temperature
of carbonization on the composition of the gas evolved from
distillation of Washington lignite. Benson and Canfield (108)
also fractionated the tar condensed from these experiments. The
results are given in Table 59. The findings in this case agree
with those of Table 58, except that here the decrease in light
oil fractions with increasing temperature of carbonization is
compensated by an increased proportion of coke. With Washington
lignite, the specific gravity of the light oil fraction advanced
from 0.807, when obtained at 250° C, to 0.821, when the coal was
distilled at 600° C. The specific gravity of the middle oil
fraction advanced from 0.905 to 0.955 at the same temperatures,
while that of the heavy or paraffin oil changed from 0.939 to
0.985.
In Table 27 there was shown the composition of gas obtained
from carbonizing peat in vertical retorts, as determined in an
investigation conducted by the Fuel Research Board (106), under
conditions of temperature somewhat exceeding the limits
ordinarily construed to fall within the category of low
temperature distillation, but which will be reproduced here, as
data on peat tar are decidedly lacking in the technical
literature. The results of these tests, as applied to the tar,
are given in Table 60.

The tar from peat was a thick and semi-solid mass which
contained a high proportion of paraffin wax and only a small
amount of ammoniacal liquor. The tar from the 843° C test
contained about 6.5% paraffin wax which melted at about 52° C.
The first fraction contained about 13.2% bases and unsaturated
compounds and 40.5% tar acids, while the second fraction was
composed of about 54% bases and unsaturated hydrocarbons and 8%
tar acids. The last two fractions were solid at 10° C and
contained practically all the paraffin wax. A separate
proportion of the 843° C tar was submitted to vacuum
distillation with the result that 0.45 gallons of purified and
refined light motor spirit was obtained per gross ton of peat.
The total peat tar, which was produced, represented about 20
gallons per gross ton. From a study of the effects of
temperature on the proportion of the tar fractions, as
demonstrated in Table 58 and Table 59, it would appear that
under true low temperature distillation, peat tar would
fractionate to about 7% in the cut up to 170° C, about 31% in
the cut from 170° C to 230° C, about 12% in the cut from 230° C
to 270° C, and about 24% in the cut from 270° C to 335° C, with
about 26% pitch and loss.
According to Garvin (112), to whom the data in Table 61 are
due, shale oils of the USA are less volatile than Pennsylvania
crude petroleum and somewhat more volatile than Scottish shale
oil. Up to 270° C, nearly 53% of Pennsylvania petroleum distills
over, as compared with about 48% for Indiana shale oil, 40% for
Utah shale oil, and 39% for Colorado shale oil. It is well to
compare the results of Table 61 with those of Figure 24, where
it is quite evident that low temperature tars from bituminous
coals are closely related to shale oil.


The data in Figure 26 are plotted from figures reported by Garvin
(112). The illustration gives a distillation analysis of the
various fractions of Scottish shale oil and two distillation
analyses of American gasolines for comparison. It is seen from the
illustration that light oil fractionated from crude shale oil is
considerably more volatile than ordinary gasoline, while the
naptha cut has a characteristic distillation curve very nearly
resembling that of gasoline, except that it has a slight
deficiency in oils boiling below 100° C and contains slightly more
of the oils boiling between 120° C and 220° C, The lubricating
oils, fractionated from shale oil, contain only about 25% oils
boiling below 340° C.
Steam Distilled Tar ~
It has been pointed out that, although the presence of steam in
the retort during low temperature carbonization appears to have
little effect on the quantity of tar produced, there is a
decided change in the constitution of the product, as indicated
by its fractionation. It has also been shown that the major
function of steam is the prevention of secondary reactions by
rapid removal of the products of distillation from the
carbonization chamber. Steam distilled tar should, therefore,
contain more of the primary products of coal. The curves in
Figure 27, after Davis and Parry (97), compare the distillation
range of steam distilled tars at various temperatures with that
obtained in atmospheric distillation at 550° C. The low
temperature tar was distilled from Pennsylvania coal. The
volatility curve for atmospheric tar obtained at 550° C lies
entirely below the curve for steam distillation beyond fractions
with a boiling point of 150° C. This indicates that steam
carbonization at low temperatures yields a tar with a larger
percentage of higher-boiling constituents than the ordinary
product. It will be noted that under steam distillation the
proportion by volume of lighter oils in the tar increases with
temperature rise in carbonization, due no doubt to cracking
effects.


Variations of the pressure under which fractionation is
accomplished is a factor in determining the percentage of tar
distilled at a given temperature, Figure 28, after Davis and
Parry (97), shows the effect of fractionation under 4 cm of
mercury pressure, when working with low temperature tar from
Pennsylvania coal. In the first place, it will be seen that the
steam tar is far more volatile than ordinary tar obtained at the
same temperature. This is the reverse of the case when
fractionation is carried on under atmospheric pressure, as shown
in Figure 27. Furthermore, above 275° C, the proportion of the
steam tar distilled increases far more rapidly than does
ordinary low temperature tar. The volatility of both steam and
regular low temperature tars, when fractionated in vacuo, is
decidedly less than when the distillation is carried out under
ordinary pressures. At first thought, this would not be
anticipated, because it might be supposed that reduction of
pressure would lower the boiling point and consequently increase
the volume of oil distilled at a given temperature. This would,
of course, be the case if the constituents of the tar existed in
the same form in both the crude and the redistilled state. By
comparing Figure 27 and Figure 28 it will be seen, however, that
at 225° C only about 2% of the tar is distilled in vacuum
fractionation, whereas about 6% is obtained at the same
temperature under ordinary distillation. This behavior finds
easy explanation in the fact that temperatures even as low as
225° C cause transformation of the tar constituents from primary
to secondary compounds.
The unsaturated hydrocarbons which are present in the tar must
be accounted part of the refining loss, judging from the present
standards of the petroleum industry. Pennsylvania low
temperature tar contains about 42% unsaturated material in the
light oil, 45% in the middle oil, and 63% in the heavy oil. This
is of importance from the standpoint of motor fuel, because the
unsaturated compounds must be removed before the oil is
considered satisfactory for use in internal combustion engines,
according to present standards [1928]. Therefore, a low
percentage of unsaturated material in the low boiling fraction
will greatly increase the yield of motor spirit.
Low temperature tar is usually examined by determining the
percent of each crude fraction soluble in 10% sodium hydroxide,
which represents the amount of tar acids present in the cut.
Then the tar is treated with 95% sulfuric acid, which removes
the unsaturated hydrocarbons. The residuum consists of the
saturated compounds which are present in the fraction. The
Pennsylvania coals, studied by Davis and Parry (97), have been
examined in this manner. Using a tar obtained at 550° C, they
found for ordinary low temperature tar the following percentages
of saturated compounds in the various fractions: 50.5% from 0° C
to 175° C; 30% from 175° C to 225%; and 26% from 225° C to 275°
C. With steam distilled tar the quantity of saturated
hydrocarbons in the various fractions was 53.1%, 37%, and 30.3%
respectively, showing a slight increase. The percentage of
unsaturated compounds in the corresponding fractions of the same
tars was 35%, 25%, and 44% respectively for the ordinary tar and
38.5%, 23%, and 43.4% respectively for the steam-distilled tar.
We see that no great change occurred in the proportion of
unsaturated material in the fractions of low temperature tar by
virtue of the presence of steam. Under the discussion of tar
acids, it was shown that the small increase in saturated
compounds, noted above in the case of steam tar, is accounted
form not through any change in the amount of unsaturated
material present, but through a reduction in the percentage of
tar acids evolved in steam distillation.
The results of Davis and Parry (97) on the examination of low
temperature tar from Pennsylvania coal carbonized at 550° C
showed that a larger amount of oil up to 275° C was present in
ordinary low temperature tar than in tar obtained by
carbonization in the presence of steam. This higher yield of oil
up to 275° C is doubtlessly the result of some secondary
reactions, prevented by the introduction of steam, as indicated
by the presence of considerably more pitch in the ordinary tar
than in the steam tar.
Full-Scale Retort Tar ~
Many additional factors enter into the situation when
large-scale operations are carried out, so that only rough
estimation of commercial retorts can be drawn from comparatively
small laboratory experiments it is important, therefore, to
study the products of large-scale carbonization processes when
such data are available. In this connection, it should be
pointed out that so much depends upon the coal used that it is
important that some method of small-scale distillation be
devised so that by the application of certain factors of
proportionality a fair idea may be gained as to what might be
expected in large-size retorts. A discussion of several
laboratory devices for this purpose has already been given in
Chapter I under the subject of assay. A more detailed
examination of low temperature tars will be undertaken in the
discussion of individual processes, but for the present purposes
a comparison of the low temperature tars, obtained by several
different processes, will suffice. This comparison is given in
Table 62. The various retorts, given in the table, represent
different types. All are eternally heated, except for the
Nielsen retort, which is a rotary kiln wherein the coal is
carbonized by the sensible heat of producer gas. The Fuel
Research board and Tozer processes are horizontal and vertical
static retorts, respectively. The Carbocoal system is a
two-stage process involving primary carbonization in a
horizontal internally agitated retort, briquetting, and finally
secondary carbonization in an inclined oven.


The Fuel Research Board (114) experiments were made with Dalton
Main #2 coal as the charge. It will be seen that not only is
there a variation between the small- and large-scale products,
but also among the large-scale processes themselves. Especial
attention is called to the low yield of the cut, 270° C to
pitch, obtained in the Fuel Research Board experiments, as
compared with that obtained in other processes. Furthermore, the
large amount of pitch obtained and the generally higher yield of
lighter hydrocarbons are all indicative of cracking and
secondary reactions in the Fuel Research Board horizontal
retorts. The fractions of tar obtained from Dalton Main #2 coal
in the Fuel Research Board horizontal retorts can be classified
according to their use in the trade. The classification in Table
63 gives in gallons per gross tone the quantity of oil that may
be expected in these cuts. In addition to the yields reported in
the table there was a refinery loss of 4.09 gallons per gross
ton, a yield of 2.54 gallons per gross ton of tar acids, and a
residuum of 67.5 pounds of pitch per gross ton. The temperature
of carbonization was 600° C.
Four British coals, carbonized at low temperatures by the Fuel
Research Board (114) in horizontal retorts, averaged 20.7
gallons of crude oil per gross ton of dry coal retorted. This
yield of crude oil was reduced to 5.8 gallons per gross ton upon
washing and refining, or a loss of about 44% in the refinement
process. It has been shown that low temperature tar contains 35%
to 40% unsaturated hydrocarbons which must be removed from the
oils to render them of commercial value according to the present
standards of the petroleum industry, which is the chief
competitor of low temperature carbonization as a source of
liquid fuels. Thus, the unsaturated material becomes the major
portion of the refining loss. Table 64 gives the original
fractional yields in gallons of dry tar per gross ton, as well
as the yields of washed and refined oils. The reduction in
refining values varies from about 25% in the first fraction to
about 50% in the last.


Low temperature tar has a high fuel value, ranging from 16,000
BTU to16,500 BTU per pound, but it has a low flash point.
Attempts to remedy this fault have been made through removal of
the light oils, but so far this procedure has rendered the tar
too viscous for use. The question of mixing low temperature tar
with mineral oils for use as a fuel has been raised. In a number
of instances this has been found impractical because of their
immiscibility. Thus, with American petroleums and shale oils,
low temperature tar has been found to be quite immiscible in all
proportions, their mixtures causing a separation of a thick
black gummy fluid. Burmese oil formed a stable mixture up to 25%
tar and Mexican oil did not give a separation until more than
50% low temperature tar was present. Trinidad was the only
petroleum which was miscible in all proportions and then only
upon heating during the mixing process. Table 65 shows the
results made on this problem by the Fuel Research Board (114).
A method of overcoming the immiscibility of low temperature tar
with crude petroleum has been proposed by Lessing (153). The tar
is mixed with petroleum, which causes a separation of the pitch,
and the mixture is then subjected to steam distillation, which
gives a sharp separation of the pitch and the oil. The
distillate is finally fractionated into liquor, neutral oil, tar
acids, bases and pitch. The crude oil is approximately 81% of
the mixture and yields on distillation: 56.1% neutral oil, 24.4%
tar acids, 0.2% tar bases, and 16.9% pitch.
Tar Cracking ~
Dalton (154) apparently made the first investigation of the
pyrogenic decomposition of hydrocarbons, but no systematic
effort was made to study this phenomenon until Berthelot (155,
156, 157) published the results of his researches, in which he
found that methane, ethane, and ethylene decomposed into
acetylene and other hydrocarbons. The polymerization of
acetylene to form aromatic hydrocarbons has been discussed in
Chapter I under the subject of chemistry.
According to Williams-Gardner (158), the views of Berthelot,
that acetylene plays an important part in the formation of
cyclic compounds, are obsolete and the mechanism of the
formation of aromatics must be modified to include either the
momentary existence of unsaturated residuums, which may by
transformed by polymerization, or broken down into carbon and
hydrogen, or else the direct formation of olefins and methane,
with or without the liberation of hydrogen and without the
necessity of forming acetylene. The opinion of Williams-Gardner
is substantiated by his own experiments, by the work of Auschutz
(159), who obtained aromatic compounds, ranging from benzene to
anthracene, from Russian oil residuums by condensation of the
complex hydrocarbons without elimination of hydrogen, and by
Haber (160), who found that n-heptane decomposed between 600° C
and 700° C to form methane and the next lowest olefin, that is,
amylene, without the formation of acetylene. This is also in
agreement with the results of Thorpe and Young (161), who
distilled solid paraffins under pressure and obtained amylene,
pentane, hexylene, hexane, heptylene, heptane, octylene, octane,
nonylene, nonane, undecylene, and undecane, all boiling below
200° C. The latter experimenters concluded, with others, that
the decomposition gives rise to olefin and lower paraffins with
or without the evolution of hydrogen.
Jones (162) observed, from his study of the influence of
temperature in cracking low temperature tars, that the mechanism
of the napthalenes, paraffins, and unsaturated hydrocarbons to
form olefins, which condense at higher temperatures to form
aromatic compounds. He found the proportion of higher olefins,
which appear in the gas from cracking, reached a maximum at 350°
C and almost disappeared at 750° C. At the latter temperature,
naphthalene appeared in the gas and shortly afterwards there was
a rapid increase in hydrogen evolution.
The effect of heat on the various constituents of primary tar
was studied by Fischer (49), who concluded that the amount of
phenol and its lower homologues, initially present in the tar,
is augmented by decomposition of the higher members of the
series at temperatures below 700° C, aromatic hydrocarbons of
the benzene series being formed from the phenols above 700° C.
According to Parrish (117) also, below 700° C the effect of heat
upon primary tar is to cause decomposition of the higher boiling
constituents into lower boiling compounds of a similar type. The
higher phenols in particular are sensitive to this cracking
effect. In general, the higher the temperature, the greater is
the proportion of lower members of a given series of compounds.
Decomposition becomes more radical above 700° C, producing
aromatic compounds, especially benzene, naphthalene, anthracene,
phenol, and pyridine. Most of the higher phenols are entirely
cracked into phenol, cresols, and xylenols. The tar bases also
decompose to form pyridine and quinoline together with their
homologues and derivatives.


The light and middle oils reported in Table 70 as fractionated
from Pennsylvania low temperature tar by Davis and Parry (97)
were vaporized in a retort and passed through a cracking chamber
which was heated to various temperatures at atmospheric
pressure. The raw stock constituted the neutral fraction from
175° C to 275° C. Table 66 shows the amount of motor spirit
obtained from the cracked distillate and scrubbed from the
cracked gas. A maximum yield of hydrocarbons boiling below 175°
C was obtained from the neutral oil at a cracking temperature of
750° C. the refining loss, due to the presence of unsaturated
material and tar acids in the fraction, amounted to 56%.
Analysis of the fraction showed the presence of aromatic
hydrocarbons, principally in the form of benzene.
The curves in Figure 29, after Davis and Parry (97), shows that
the amount of oil recovered after cracking is almost exactly
inversely proportional to the temperature and that the yield of
light oil and gas increased with temperature rise, but at a
decreasing rate. Comparison of Table 66 and Figure 29 points out
that, although the percentage of motor spirit contained in the
cracked oil reaches a maximum at 750° C, the yield of motor
spirit in the gas has no such maximum, but continues to increase
with the temperature.
When the anthracene oil fraction from 280° C to 350° C of Table
70 was subjected to similar cracking reactions at 800° C and
atmospheric pressure, about 50% of the cracked oil was
recovered, yielding per net ton of coal 0.12 gallons of crude
light oil boiling below 175° C. This was no increase from that
obtained in the Hempel distillation of the anthracene oil before
cracking, so that in cuts up to 200° C, at least, it may be said
that little secondary decomposition took place in the anthracene
oil fraction. About 47 cu ft of gas per gallon were evolved
during the heating at 800° C and this yielded upon scrubbing
2.2% of the fraction, or 0.09 gallons of crude light oil per net
ton of coal.
Curtis and Beekhuis (163) conducted a number of cracking tests
on the neutral oil of low temperature tar from the McIntire
process and made from West Virginia coal containing 3% moisture,
36% volatile, 50% fixed carbon, and 10.5% ash. The coal was
carbonized at 620° C. Pressures varying from 100 pounds to 500
pounds per square inch were used inc racking. They found that
decomposition was slow below 400° C, but that above 450° C the
formation of coke, accompanied by high gas yield, was rapid.
Within the range of their observations, the effect of pressure
variation of the rapidity and character of the cracking was
slight. By cracking the kerosene fraction of the neutral oil,
they found that the yield of oil boiling below 210° C could be
increased from 20% of the neutral oil initially present to about
35%.
Morrel and Egloff (164) cracked a typical low temperature tar
from a West Virginia bituminous coal, which yielded about 25
gallons of crude tar per net ton. The crude tar, with a specific
gravity of 1.074, was cracked at 100 pounds per square inch
pressure and 452° C temperature to yield 40% cracked distillate
with 0.8927 sp gr. The gasoline fractionated from the cracked
distillate amounted to 18.1% of the initial crude tar, and had a
specific gravity of 0.8299. The straight distillate from this
low temperature tar analyzed 26.4% tar acids, 3.3% tar bases,
44.3% unsaturated hydrocarbons, 9.5% aromatic hydrocarbons, and
16.4% napthenes and paraffin hydrocarbons, while the cracked
distillate from the straight distillate contained 21.3% tar
acids, 4.3% tar bases, 27% unsaturated hydrocarbons, 20.3%
aromatic hydrocarbons, and 27.3% napthalenes and paraffin
hydrocarbons.
The neutral oil from this same low temperature tar, having a
specific gravity of 0.9484, when cracked at 175 pounds per
square inch and 427° C, yielded 50% cracked distillate of 0.8514
sp gr, which fractionated to give 31.9% of the initial crude tar
as gasoline of 0.8090 sp gr. Analysis of the neutral oil before
cracking showed it to be composed of 2.7% tar acids, 4.5% tar
bases, 60.6% unsaturated hydrocarbons, 9.4% aromatic
hydrocarbons, and 22.8% napthene and paraffin hydrocarbons,
while the cracked distillate analyzed 4.8%, 4.8% tar bases,
38.4% unsaturated hydrocarbons, 19.4% aromatic hydrocarbons, and
32.7% napthalenes and paraffin hydrocarbons.
These data show a decided decrease upon cracking in the
unsaturated compounds and a small increase in the aromatic
hydrocarbons. The tar acids in the cracked distillate appear to
be mainly cresols. No naphthalene was present, either in the
initial crude tar or in the cracked distillate. While only a
trace of anthracene was found in the original low temperature
tar, considerable quantities were found after cracking. This was
thought remarkable inasmuch as the temperature of cracking was
only 452° C, whereas anthracene heretofore had been regarded as
a high temperature product.
Egloff and Morell (165) also cracked the low temperature tar
from an Ohio-Indiana bituminous coal at 100 pounds per square
inch and 426° C. The crude tar has a specific gravity of 1.0794
and yielded upon fractionation 29% neutral oil, 27.5% tar acids,
and 1.6% bases. When cracked, this crude low temperature tar
yielded 33.9% gasoline containing 35% tar acids, or an acid-free
yield of 22% gasoline. The neutral oil from this same crude
stock was cracked at 200 pounds per square inch and 455° C to
yield over 50% high grade antiknock motor fuel.
Shale oils from the USA, Australia, and France were cracked by
Morrell and Egloff (166) under pressures ranging from 120 pounds
per square inch and at temperatures ranging from 398° C to 433°
C to produce over 50% of the raw stock as gasoline. This cracked
gasoline was declared to be a motor fuel of high antiknock
value. The results of the cracking tests on shale oil are given
in Table 67.

Cracked Tar Gas ~
The fixed gas evolved during cracking is of an entirely
different nature from the gas which is evolved during
carbonization of the coal. The latter is the result of
destructive distillation, or primary modification of the fuel
constituents, while the former is the outcome of secondary
decomposition induced through the medium of heat. An analytical
study of the fixed gas from the neutral oil fraction from 175° C
to 275° C at various cracking temperatures was made by Davis and
Parry (97). This is the same oil fraction from Pennsylvania coal
already referred to in Table 66 and Figure 29, and which will be
further discussed in Table 70. Analyses of the fixed gas which
was evolved from cracking the neutral oil at various
temperatures are given in Table 68.
The permanent gas, obtained from cracking the anthracene oil
fraction from 275° C to 350° C at 800° C was almost identical in
composition with that liberated by the neutral oil when cracked
at 820° C. The large percentage of hydrogen is indicative of
much unsaturated material in the cracked oil and this was shown
to be the case. The proportion of ethylene and of ethane
decreased with rise in the cracking temperature, while the
percentage of methane greatly increased. The small percentage of
the oxides of carbon which was produced is worthy of notice,
especially in comparison with the compositions of the gas from
carbonization, as shown in Table 23 and in Figure 10.
If low temperature neutral oil is essentially the same as
petroleum gas oil, there is a possibility of finding a
commercial outlet in that market. A great deal of information is
thrown on that matter by an examination of the fixed gas
obtained in the cracking of petroleum gas oil. Such an
experiment has been conducted by Davis and Parry (97), the
results of which are given in Table 69.


The gas, evolved in cracking petroleum gas oil, has about the
same thermal value as that obtained from the low temperature tar
neutral oil, but a somewhat larger quantity of gas is produced.
Far less hydrogen and methane is evolved from the former, but a
very large percentage of nitrogen. It might be said, in general,
that the two oils are not greatly different and that low
temperature tar neutral oil is comparable with the petroleum gas
fraction.
Hydrogenation ~
Upon consideration of the facts hitherto presented, it appears
that primary tar normally consists of alipathic hydrocarbons and
phenolic compounds. Whatever small percentage of light aromatic
compounds that is found to be present in the tar must be derived
from reduction of these phenolic substances. This suggests the
possibility of entirely reducing the tar acids to low-boiling
oils for use as a motor fuel. This problem has been studied by
Fischer (167) and by Fischer and Schroeder (168), who passed
elementary hydrogen together with a mixture of phenols and
hydrocarbons through a tube heated to 750° C. A great deal was
found to depend upon the nature of the reaction tube, but they
discovered that a tinned iron tube was suitable for commercial
processing and avoided the excessive deposition of carbon that
sometimes plugged the tube. The deposition of carbon was
apparently accelerated by iron. However, if water gas was used
as a source of hydrogen for hydrogenation, the tinned coating
peeled away, as a result no doubt of the action of carbon
monoxide in the gas. An iron reaction tube, coated internally
with iron sulfide, was finally adopted by Fischer and Zerbe
(169) to overcome this last difficulty. With the improved form
of hydrogenating tube, little separation of carbon occurred and
the phenols were reduced to benzene homologues. With cresols and
xylenols, the conversion to motor fuel was up to 99% and 72%,
respectively, of the theoretical.
Whether or not hydrogenation at high pressures and high
temperatures, according to the Bergius process (170, 171, 172),
is applicable to low temperature tar acids has not yet been
demonstrated. According to Fischer (173), at the temperature of
400° C, employed in such circumstances, hydrogenation could be
successful only at hydrogen pressures exceeding the dissociation
pressure of hydrogen in the compounds to be produced. At lower
temperatures of 200° C, the reaction velocity would be entirely
too slow. The introduction of a catalyst to accelerate the
reaction rate at lower temperatures necessitates excessive and
expensive purification of the reaction gases to eliminate sulfur
and avoid poisoning the catalyst. Fischer and Schroeder (174)
obtained the conversion of the phenolic portion of primary tar
into oils under pressure at 400° C with nascent hydrogen derived
from sodium formate and from carbon monoxide and steam.
Sabatier and Senderens (175) showed that naphthalene vapor
mixed with and excess of hydrogen could be passed over finely
divided nickel at 175° C to produce decahydronapthalene with a
boiling point of 187° C, and at 200° C to from
tetrahydronapthalene with a boiling point of 205° C. Fischer
(176, 177) obtained the partial liquefaction of naphthalene to
the extent of 35% by treating it for about half an hour at 150
lb/sq in pressure in the presence of aluminum chloride. From
these experiments, it is apparent that naphthalene can be
considered as a raw material for the manufacture of motor fuel.
On account of the limited amount of naphthalene present in
primary tar, however, it is doubtful if this process is
important from the standpoint of low temperature carbonization,
unless it be in the secondary treatment of cracked tar products.
For the most part, cracked motor fuel contains a high
proportion of unsaturated compounds which, according to present
standards, are more or less undesirable, due principally to
unpleasant odors and darkening of the distillate. The method of
hydrogenation, developed by Bergius (172) and commonly known as
berginization, in which the distillate is cracked at the
required temperature under a high pressure of elementary
hydrogen, largely overcomes this difficulty and yields a
distillate containing only a small percentage of unsaturates.
Figure 30, after Kling (178), shows the pressure developed in
berginization and in cracking the same oil. It is obvious that,
as the heating period is prolonged, the pressure of the cracked
vapor becomes greater and greater. During berginization,
however, there is a rapid increase of pressure during initial
heating but finally, when equilibrium has been reached, the
pressure decreases at constant temperature, thereby indicating
hydrogenation. Berginization appears to require close pressure
and temperature control, as indicated by the negative
experiments of Waterman and Perquin (179), who were unable to
effect hydrogenation of paraffin wax at 435° C and initial
pressured of 600 psi, and of Skinner and Graham (180), who were
unable to secure hydrogen absorption by low temperature tar
obtained at 600° C from Fifeshire cannel coal and redistilled at
450° C, when the tar was treated under 1560 psi hydrogen
pressure at 411° C for 4 hours. Bergius used initial pressured
of as much as 1500 psi which rose in some cases to above 3000
psi when the autoclave was heated to the required temperature.

Motor Fuel ~
Motor fuel is obtained directly from two sources in low
temperature carbonization, the tar and the gas, and it can be
obtained from three subordinate sources by special processing,
such as cracking of the heavy tar fraction, hydrogenation of the
tar acids, and liquefaction of the gas by high pressures and low
temperatures.
The entire fraction up to 200° C, when freed of the tar acids,
bases, and unsaturated material, may be sued as an internal
combustion engine fuel. This fraction consists mostly of higher
paraffins, unsaturated hydrocarbons, and in some cases a small
quantity of benzene and toluene. This cut may yield anywhere
from one to 3 gallons of refined motor spirit per ton of coal.
Davis and Parry (97), in their work on the low temperature
carbonization of Pennsylvania coals paid special attention to
the yield of products valuable as motor fuel. Using the tar
obtained from retorting Freeport coal at 550° C in a stationary
vertical retort, they examined the light oil fraction obtained
from the tar and the possibility of cracking the higher-boiling
hydrocarbons. The fractionation of this tar is given in Table 70

=== Table 71[Not available] ===
The entire fraction boiling under 175° C was considered a crude
light oil suitable as a motor fuel. It was found that 42% of
this cut consisted of unsaturated hydrocarbons and 17% of tar
acids, which must be removed in refinement before the oil could
be sold to the trade. This means that the crude yield of 2.8
gallons must be reduced 59% to 1.15 gallons of refined motor
spirit obtained from this fraction.
The recovery of light oils in the gas is not a simple process.
It can be accomplished by condensation, scrubbing, or
adsorption. Fischer and Gluud (181) recovered the motor spirit
from the low temperature gas in their experiments by scrubbing
with paraffin oil and subsequent rectification. They obtained a
yield of 3.8 gallons to 2.0 gallons per net ton of coal
carbonized, representing 1.23% and 0.67% of the coal,
respectively. The Fuel Research Board (114) also carried out
scrubbing experiments on the gas from low temperature
carbonization in horizontal retorts, as shown in Table 71. They
recovered 1.68 gallons of refined spirit per gross ton of coal.
It has a specific gravity of 0.731 at 15° C. Dalton Main #2
coal, which gave 3200 cu ft of gas at the temperature of
distillation, was used in the work of the Fuel Research Board.
The duration of the scrubbing, as well as the rte of washing,
was varied in order to dtermine the influence of these factors
on the quantity of light oil recovered.
=== Table 72 [Not available]===
The refining loss of motor spirit, obtained from low
temperature gas, is less than that occurring in the light
fraction of the tar, Scrubbing greatly reduces the percentage of
hydrocarbons present in the gas and it has been pointed out,
notably in Figure 13 and elsewhere, that low temperature gas is
in part characterized by a low proportion of unsaturated
hydrocarbons. Consequently, it is anticipated that there should
be a low refining loss in the light oils scrubbed from the gas.
The rate of scrubbing with wash oil does not appear to have as
great an effect on the amount of spirit recovered as the
quantity of wash oil used per cubic foot of gas scrubbed.
Engelhardt (182) investigated the recovery of light oil from
illuminating gas by activated charcoal, the condensed vapors
being subsequently liberated by treatment with superheated
steam. This same process was used by Davis and Parry (97) in the
recovery of light oils from the gas evolved in the low
temperature carbonization of Pennsylvania coals. They found that
the efficiency of the charcoal was greatly reduced in the
presence of hydrogen sulfide and water vapor. Thus, while up to
3 gallons of crude light oil per net ton of coal were recovered
in ordinary distillation, only one gallon was obtained under
steam distillation. By this method they recovered an average of
2.75 gallons of crude motor spirit per net ton of coal from 6
different coals. This light oil had a specific gravity of 0.740
and represented 0.838% of the coal charged. It contained 23%
unsaturated hydrocarbons, so that the net yield of refined
spirit was reduced to 2.23 gallons. A recent development
utilizing this same principle of adsorption has been the
introduction of silica gel in place of activated charcoal.
Table 72 shows the yield of light oils reported by a number of
different investigators in low temperature carbonization. All of
these experiments were made in externally heated retorts, except
those of Gluud (23) and a few runs by Parr and Olin (37), in
which cases internal heating was used. Lewis (17) used a gas
coal; Runge (183) used a by-product coal; Parr and Olin (37)
used Illinois coal; Davis and Berger (184) used Freeport coal;
Morgan and Soule (48) used Pittsburgh Terminal coal; Davis and
Parry (97) used coal from the Freeport bed; and the Fuel
Research Board (114) used Dalton Main #2. Attention must be
called to the fact that the yields in Table 72 are crude light
oil and large reductions occur in refining, as has been pointed
out, so that the yield of refined motor spirit will be
considerably less. It is interesting to note the wide variation
in the yields reported and caution must be sounded that some
observers report their tar yields before hydrogenation and their
crude tar yields are swelled by moisture content. The high
yields of Parr and Olin, and also of Gluud, are due in part to
the fact that they included higher boiling hydrocarbons in their
light oil fraction. The figure given by Davis and Parry includes
the light oil obtained from both the tar and the gas.
The explosive range of low temperature motor spirit is over 20%
greater than that of petrol, while a mixture of equal parts of
motor spirit and petrol has an explosive range 30% greater than
petrol alone and 35% greater than benzol, according to the Fuel
Research Board (114). The explosive range, expressed as cubic
centimeters per cubic foot of air at 15° C, was 2.7 to 6.7 for
benzol of 0.881 sp gr; 2.5 to 7.3 for petrol of 0.723 sp gr; 2.3
to 9.5 for low temperature motor spirit of 0.731 sp gr; and 2.5
to 10.5 for a mixture of equal parts of petrol and motor spirit.
The motor spirit obtained from low temperature distillation of
coal is miscible in all proportions with benzol and petrol for
use as a fuel in internal combustion engines. With the growing
use of alcohol in the field of motor fuels, something should be
said concerning the miscibility of low temperature motor spirit
with alcohol. The amount of low temperature motor spirit held in
solution by alcohol depends largely upon the strength of the
latter; thus at 10° C the Fuel Research Board (114) found 95%
alcohol held as much as 74.2% motor spirit in solution, while
90% alcohol held but 16.3% and 85% alcohol retained only 7.1%.
With a mixture of benzol and 95% alcohol, in equal proportions,
low temperature motor spirit is dissolved up to 60% at 10° C.
Even with refined spirit, there is a slight tendency for
resinous matter to deposit on standing, thus darkening the fuel,
but alcohol prevents deposit by dissolving the resins which
separate.
It has been pointed out that Davis and Parry (97) recovered
1.15 gallons per net ton of refined motor spirit from the light
oil fraction of low temperature tar which they obtained from
Pennsylvania coal and 2.23 gallons per net ton of refined motor
fuel from the low temperature gas, netting 3.38 gallons per net
ton which were obtained directly without additional processing.
It has also been pointed out that they obtained an additional
0.66 gallons per net ton by cracking the higher-boiling tar
fractions. We have already seen that other Pennsylvania coals
yielded 5.45 gallons per net ton of tar acids, 56.5% of which
can be converted to motor fuel by hydrogenation at an efficiency
of 72% to 99% of the theoretical maximum conversion, which
ranges from 64% to 87% depending on the initial and final
products. On this basis, an additional 1.5 gallons to 2.5
gallons of motor fuel per net ton could be obtained. By
employing these additional means, there appears the possibility
of increasing the total motor fuel yield to 5.5 gallons or 7.5
gallons or even more per net ton of coal. The desirability of
associating hydrogenation with low temperature carbonization, to
effect an increase of motor fuel by hydrogenating the tar acids,
has been stressed by Gentry (185), but Egloff and Morrell (165)
see no reason at all why the tar acids should be removed from
the motor fuel, as demanded by present standards.
Fuel Oil ~
Crude low temperature tar has a viscosity quite suitable for
fuel oil, but according to Lander and McKay (186) its flash
point is far too low. On the other hand, if sufficient low
volatile constituents are removed by distillation to increase
its flash point, the crude tar becomes too viscous for use as a
motor fuel. However, the tar may be fractionated in such a way
as to obtain a distillate which meets all the requirements of a
fuel oil, both from the standpoints of flash point and of
viscosity. Furthermore, there is no evident reason why the fuel
oil, if too viscous, cannot be heated before firing to increase
its fluidity. The immiscibility of low temperature tar with
other fuel oils has already been dealt with.






































